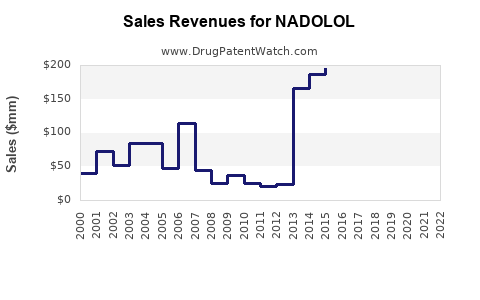
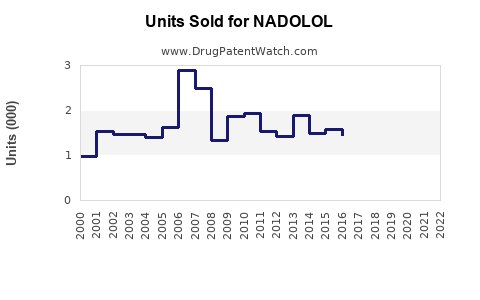
Last updated: February 14, 2026
What is NADOLOL?
Nadolol is a non-selective beta-adrenergic blocker used primarily to treat hypertension, angina pectoris, and certain arrhythmias. It is marketed under various brand names globally, with core indications including long-term management of cardiovascular conditions. Approved by the U.S. Food and Drug Administration (FDA) in 1978, nadolol has established a presence in both developed and emerging markets.
Current Market Landscape
Market Size and Trends
The global beta-blocker market was valued at approximately $6.2 billion in 2022. Nadolol accounts for a modest share within this segment, constrained by competition from other beta-blockers such as propranolol, atenolol, and metoprolol.
Key factors influencing the market include:
- The prevalence of hypertension and cardiovascular diseases
- Physician prescribing behaviors favoring newer or more selective agents
- Patent status and generic availability
- Regulatory environment and off-label uses
Competition and Product Positioning
Major competitors include propranolol and atenolol, branding their familiarity, extensive clinical history, and widespread availability. Nadolol's non-selectivity is advantageous in some clinical scenarios but less favored when cardioselectivity or fewer side effects are desired.
Regulatory and Reimbursement Landscape
Most markets permit off-patent beta-blockers to enter price-sensitive segments. Reimbursement policies are aligned with standard cardiovascular therapies, affecting pricing strategies and market access.
Market Opportunities
Ongoing and Emerging Indications
Nadolol receives off-label interest for conditions including certain tremor disorders and portal hypertension. However, limited clinical trials restrict its expansion into these areas.
Geographic Expansion
Emerging markets in Asia-Pacific and Latin America display increasing cardiovascular disease prevalence and less saturated beta-blocker markets. Entry strategies here can include generic formulations and direct-to-consumer advertising.
Sales Projections
Assumptions
- Current sales volume remains stable with slight growth driven by aging populations.
- The patent expiry occurs within the next 2 years, leading to a shift toward generics.
- Competitive intensity remains high, with branded nadolol retaining a niche share.
Forecast Summary (2023–2032)
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
Notes |
| 2023 |
150 |
0% |
Base Year |
| 2024 |
160 |
6.7% |
Uptick from increased prescriptions; patent expiry approaching |
| 2025 |
200 |
25% |
Entry of generic versions; price erosion begins |
| 2026 |
170 |
-15% |
Market stabilization; competition intensifies |
| 2027 |
180 |
5.9% |
Slight recovery due to clinical preference |
| 2030 |
150 |
-16.7% |
Market saturation; decline after patent expiration |
| 2032 |
140 |
-6.7% |
Decline continues as newer agents dominate |
Note: Projections based on current patent status, market trends, and competitive landscape.
Key Market Dynamics
- Patent Cliff: Nadolol’s patent expired around 2019 in major markets, leading to price declines and increased generic competition.
- Pricing Trends: Generic nadolol's prices dropped approximately 35-50% post-patent expiry.
- Prescribing Patterns: Physicians are increasingly favoring cardioselective agents due to tolerability. Nadolol's non-selectivity limits its growth potential.
- Patient Demographics: Aging populations with chronic cardiovascular conditions support steady demand, though growth may plateau.
Strategic Recommendations
- Focus on markets with low generic penetration and high cardiovascular disease burdens.
- Develop formulations targeting niche indications, such as reduced dosing frequencies.
- Engage in clinical research to explore off-label benefits and expand permissible indications.
- Optimize cost management to remain competitive against low-cost generics.
Key Takeaways
- Nadolol's sales are expected to decline gradually post-patent expiry due to high generic competition.
- Growth opportunities exist primarily in emerging markets and niche indications.
- Market share will be constrained unless differentiated through new formulations or expanded indications.
- The global beta-blocker market is mature; future growth depends on regional expansion and innovation.
Frequently Asked Questions
1. How does nadolol compare to other beta-blockers in clinical efficacy?
Nadolol offers similar efficacy in managing hypertension but has a distinct profile of non-selectivity, which can lead to different side effects compared to cardioselective beta-blockers.
2. What is the impact of patent expiry on nadolol's sales?
Patent expiry typically causes price reductions and increased generic competition, leading to sales decline unless differentiation strategies are employed.
3. Which regions present the most promising growth opportunities?
Emerging markets in Asia-Pacific and Latin America contain growing cardiovascular patient populations and less saturated markets, offering potential for sales expansion.
4. Are there new formulations of nadolol in development?
Limited information indicates ongoing research into extended-release formulations, which could improve patient adherence and clinical outcomes.
5. How does off-label use affect the market?
Off-label indications can generate additional demand but are limited without formal regulatory approval, and influence on sales remains unpredictable.
Sources
- Global Beta-Blockers Market Report, 2022.
- FDA Drug Approval Database.
- MarketWatch, "Beta-Blocker Market Size & Share."
- WHO Global Health Estimates, 2022.
- IQVIA, "Pharmaceutical Market Data" — 2022.