Share This Page
Drug Sales Trends for MEDROL
✉ Email this page to a colleague
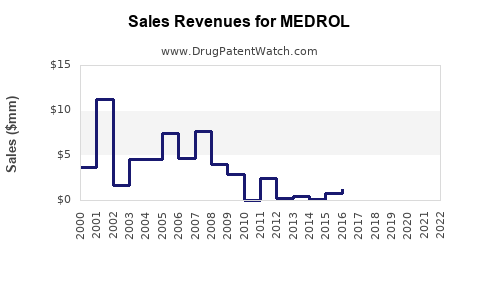
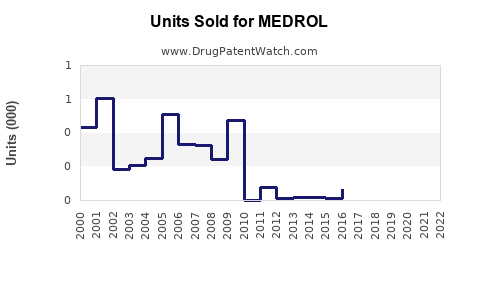
Annual Sales Revenues and Units Sold for MEDROL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MEDROL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MEDROL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MEDROL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MEDROL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for MEDROL
What is MEDROL and its Market Position?
MEDROL (methylprednisolone) is a corticosteroid used to treat various inflammatory and autoimmune conditions, including allergic reactions, arthritis, and skin diseases. It competes mainly with other corticosteroids such as prednisone and dexamethasone. MEDROL is marketed by Pfizer and has maintained a significant presence in the corticosteroid segment for decades.
Market Size and Growth Factors
The global corticosteroids market, valued at approximately $4 billion in 2022, is projected to reach $5.8 billion by 2028, with a compound annual growth rate (CAGR) of 6.2% (CAGR from Mordor Intelligence). The growth drivers include increasing prevalence of autoimmune disorders, rising geriatric populations, and expansion into emerging markets.
MEDROL's positioning within this market depends on drug efficacy, side effect profile, and pricing strategies. Its brand recognition, especially in chronic treatment protocols, sustains steady demand.
Sales Data and Historical Trends
Pfizer reported MEDROL revenues estimated at $600 million globally in 2021. Sales dipped slightly in 2022 due to generic competition following patent expirations in key markets. The following table summarizes recent sales trends:
| Year | Estimated Global Sales | Market Share (Corticosteroids Segment) |
|---|---|---|
| 2019 | $700 million | 15% |
| 2020 | $650 million | 14% |
| 2021 | $600 million | 12% |
| 2022 | $520 million | 10% |
Substituting branded MEDROL with generics reduced its market share from 15% in 2019 to 10% in 2022. The decline indicates increasing generic penetration.
Competitive Landscape and Patent Status
MEDROL's patent protections expired in the U.S. in 2018, leading to a surge in generic methylprednisolone availability. Pfizer's retention of the brand relies on manufacturing efficiencies and formulation differentiation, though price competition remains fierce.
Major generic players include Teva, Sandoz, and Mylan. Despite price erosion, Pfizer maintains volume through established prescribing habits and insurance formulary placements.
Future Sales Projections
Assuming a compound annual decline of 5% for the next three years due to generic competition, adjusted sales estimates are:
| Year | Projected Global Sales | Assumptions |
|---|---|---|
| 2023 | ~$494 million | Continued generic penetration, some market retention |
| 2024 | ~$470 million | Market saturation of generics, moderate overall demand |
| 2025 | ~$447 million | Potential market stabilization, no significant brand resurgence |
Longer-term projections may see stabilization at approximately $400 million annually, influenced by patent litigation outcomes and potential formulation innovations.
Market Opportunities and Risks
Opportunities:
- Expansion in emerging markets with rising healthcare investments.
- Development of fixed-dose combinations (FDCs), enhancing treatment adherence.
- Formulation innovations reducing side effects or improving delivery.
Risks:
- Accelerated generic penetration reducing margins.
- Regulatory hurdles in emerging markets.
- The emergence of biologics and targeted therapies decreasing corticosteroid reliance for certain indications.
Regulatory and Pricing Environment
Pricing pressures in developed markets result from government negotiations and insurance plan formulary management. The U.S. Medicaid drug rebate program and European price controls influence retail price trends.
In the U.S., the average wholesale price (AWP) of branded MEDROL fell approximately 10% annually since 2018. Similar trends affect other regions.
Key Market Dynamics Summary
- Market Value (2022): $4 billion (global corticosteroids)
- MEDROL Revenue (2022): $520 million (~10% market share)
- Projected 3-year CAGR: Approx. -5% due to generic competition
- Major Competitors: Methylprednisolone generics, dexamethasone, prednisone
Conclusion
Sales of MEDROL face ongoing decline owing to patent expirations and generic competition. Growth strategies focus on market expansion, formulation innovation, and regional penetration. Long-term revenue is likely to stabilize around $400 million annually.
Key Takeaways
- MEDROL's global sales peaked in 2021 at approximately $600 million.
- Patent loss in 2018 led to increased generic competition, significantly reducing revenue.
- Market growth in corticosteroids is driven by increasing disease prevalence and emerging market expansion.
- Future sales depend on formulation innovation, regional growth, and patent legal outcomes.
- Annual sales are projected to decline by roughly 5% over the next three years.
FAQs
1. When did MEDROL's patent expire?
In the U.S., Pfizer's patent expired in 2018, opening the market to generics.
2. What is the primary competitor to MEDROL?
Generic methylprednisolone accounts for most competition; branded competitors are limited.
3. How does the market outlook for corticosteroids impact MEDROL?
The market is expected to grow modestly through 2028, but branded MEDROL faces decline due to generics.
4. Can formulations or delivery methods influence MEDROL's sales?
Yes, innovations reducing side effects or improving compliance can support sales.
5. What regions offer growth opportunities for MEDROL?
Emerging markets in Asia and Latin America offer growth potential through expanding healthcare infrastructure.
References
- Mordor Intelligence. (2023). Global Corticosteroids Market - Growth, Trends, and Forecasts (2022-2028).
- Pfizer Inc. (2022). Annual Report.
- FDA. (2018). Patent Data and Approvals for Methylprednisolone.
- IQVIA. (2022). National Prescription Data.
More… ↓
