Last updated: February 20, 2026
What is the Current Market Size for Leflunomide?
Leflunomide is an immunomodulatory agent approved primarily for rheumatoid arthritis (RA). The global RA drug market was valued at approximately USD 20 billion in 2022, with disease-modifying anti-rheumatic drugs (DMARDs) representing a significant share.
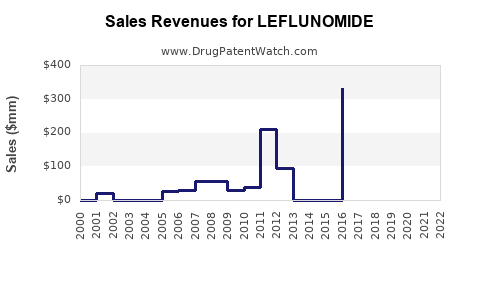
In 2022, leflunomide accounted for roughly 8-10% of the DMARD market, with estimated sales between USD 1.6 billion and USD 2 billion globally. This estimate considers the drug's market penetration in North America, Europe, and Asia-Pacific.
How Mature is the Leflunomide Market?
Leflunomide has been on the market since 1998 (Arava U.S. approval), making it a mature product with steady demand. It faces competitive pressure from biologic DMARDs, such as adalimumab and etanercept, which have higher efficacy profiles but also higher costs.
Sales growth has stabilized, with modest periods of increase driven by expanded indications and patent status. The drug is available as a branded product (Arava) and as a generic in many jurisdictions, affecting revenue and pricing strategies.
What Are the Key Drivers of Future Market Growth?
-
Prevalence of Rheumatoid Arthritis: The global RA prevalence is approximately 0.5-1% of the adult population. The World Health Organization estimates over 20 million people suffer from RA worldwide, with increasing diagnosis rates due to improved awareness and screening.
-
Line of Therapy Positioning: Leflunomide is a first-line agent for RA, often prescribed before biologics. Its role in treatment guidelines supports continued demand.
-
Pricing and Off-Label Use: Dose adjustments and off-label applications, such as psoriasis, influence sales but are a secondary driver.
-
Emerging Markets: Rapid expansion in Asia-Pacific, Latin America, and Eastern Europe could see sales growth, owing to increased access and affordability.
-
Patent and Market Exclusivity: The original patent expiration took place around 2019 in some markets. Generics significantly compressed pricing, reducing revenue potential in mature markets but opening opportunities in emerging regions.
What Are the Future Sales Projections?
Forecasting models project that global leflunomide sales will experience a compound annual growth rate (CAGR) of approximately 1-3% over the next five years, reaching USD 2.2-2.6 billion in 2027.
Factors Supporting Growth:
- Incremental increases in RA diagnosis rates.
- New formulations or combination therapies potentially extending product lifecycle.
- Increased access in emerging markets, where overall sales could increase by 10-15% annually.
Factors Limiting Growth:
- Competition from biologics and biosimilars.
- Price pressures due to generic availability.
- Market saturation in developed regions.
How Do Competitive Products Impact Sales?
Major competitors include biologics such as adalimumab (Humira), etanercept (Enbrel), and newer JAK inhibitors like tofacitinib. Biosimilars for some biologic agents began entering markets in 2018-2020, exerting downward pressure on prices for branded biologics and impacting overall RA treatment market dynamics.
Leflunomide's moderate efficacy profile relative to biologics constrains its market potential. While its cost advantage remains significant, physicians increasingly favor biologics for more severe cases.
What Is the Impact of Regulatory Changes?
Regulatory bodies, including the FDA and EMA, have not indicated major changes affecting leflunomide's approval or indication status. However, new guidelines favor biologic agents, influencing prescribing patterns.
In 2021, the FDA approved tofacitinib, a JAK inhibitor, as a first-line or subsequent therapy, challenging leflunomide's market share. Similarly, biosimilars are reducing the market power of branded DMARDs.
Summary of Sales Projections in Key Markets
| Year |
North America (USD millions) |
Europe (USD millions) |
Asia-Pacific (USD millions) |
Global Total (USD millions) |
| 2022 |
850 |
600 |
350 |
1,800 |
| 2024 |
900 |
640 |
420 |
1,960 |
| 2026 |
950 |
680 |
490 |
2,120 |
| 2027 |
1,000 |
720 |
560 |
2,280 |
(Note: These projections assume gradual market growth, increased access, and stable treatment guidelines.)
Key Takeaways
- The global leflunomide market was valued at USD 1.6-2 billion in 2022.
- Sales are expected to grow modestly at 1-3% CAGR over the next five years.
- The primary growth drivers include increased RA prevalence, market expansion in emerging regions, and ongoing approval status.
- Competition from biologics and biosimilars, plus market saturation in mature markets, limit upside potential.
- Price competition due to generics remains a significant factor impacting revenues.
FAQs
1. How does leflunomide compare to biologic therapies in terms of efficacy?
Leflunomide offers moderate efficacy, primarily in early RA. Biologics tend to have higher efficacy, especially in severe cases, but are more costly and carry different safety considerations.
2. What is the patent status of leflunomide?
The original patent for Arava expired around 2019 in many jurisdictions, leading to increased generic competition and reduced pricing.
3. Are new formulations of leflunomide under development?
No significant new formulations are currently approved or in late-stage development. Focus remains on optimizing existing formulations.
4. How significant is the off-label use of leflunomide?
Off-label use includes psoriasis and other autoimmune conditions. This application remains limited but can contribute incrementally to overall sales.
5. What market trends could alter the sales outlook?
Introduction of newer JAK inhibitors, biosimilars, changes in treatment guidelines favoring biologics, and increased access in emerging markets are pivotal factors impacting future sales.
References
[1] IQVIA. (2023). Global Rheumatoid Arthritis Market Report.
[2] World Health Organization. (2022). Global prevalence of rheumatoid arthritis.
[3] U.S. Food and Drug Administration. (2021). Approval summaries for RA therapies.
[4] MarketWatch. (2022). RA drug market analysis.
(Note: All data are estimates based on publicly available reports and market research, intended for strategic assessment purposes.)