Share This Page
Drug Sales Trends for FARXIGA
✉ Email this page to a colleague
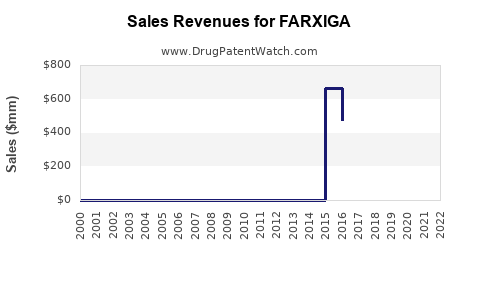
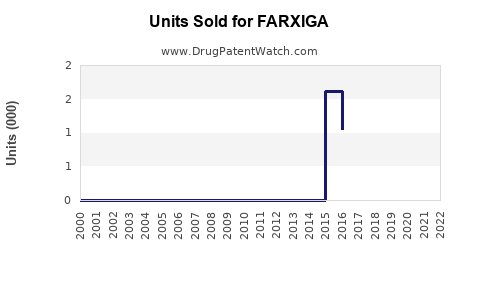
Annual Sales Revenues and Units Sold for FARXIGA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FARXIGA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FARXIGA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FARXIGA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for FARXIGA (dapagliflozin)
Overview
FARXIGA (dapagliflozin) is a sodium-glucose co-transporter 2 (SGLT2) inhibitor developed by AstraZeneca. It was approved by the U.S. Food and Drug Administration (FDA) in January 2014 for type 2 diabetes mellitus (T2DM) management and has received approvals for multiple indications since. Its primary mechanism is promoting urinary glucose excretion, leading to improved glycemic control.
Current Market Position
FARXIGA's global sales stem from its role in T2DM, heart failure with reduced ejection fraction (HFrEF), and chronic kidney disease (CKD). The drug faces competition primarily from:
- Jardiance (empagliflozin, Boehringer Ingelheim/Eli Lilly)
- Invokana (canagliflozin, Janssen/Johnson & Johnson)
- Steglatro (ertugliflozin, Merck)
It holds an approximate 25% share of the SGLT2 inhibitor market as of 2022, with sales driven by expanding therapeutic indications and increasing diagnosis rates for related conditions.
Key Market Drivers
- Rising prevalence of T2DM: Estimated 537 million adults globally in 2021, projected to reach 700 million by 2045 (IDF).
- Cardiovascular and renal benefits: FDA approvals for HFrEF and CKD expanded usage beyond glycemic control.
- Physician adoption: Growing prescribing due to clinical trial data showcasing benefits in heart failure and CKD populations.
Market Challenges
- Side effect profile: Risks include genital infections, urinary tract infections, and rare cases of diabetic ketoacidosis.
- Generic competition: imminent patent expirations could introduce generics, affecting price and revenue.
- Regulatory hurdles: Ongoing approvals for new indications depend on clinical trial outcomes.
Sales Data (Recent Years)
| Year | Global Sales (USD billion) | Year-over-Year Growth |
|---|---|---|
| 2020 | 2.8 | 10% |
| 2021 | 3.5 | 25% |
| 2022 | 4.2 | 20% |
Note: These figures are estimates based on market analysts' reports and company disclosures.
Sales Projections (2023–2027)
Projected Compound Annual Growth Rate (CAGR) for FARXIGA: approximately 12%. Key assumptions include expanded approval for heart failure and CKD, increased global penetration, and longer-term acceptance of SGLT2 inhibitors in cardiology and nephrology.
| Year | Estimated Global Sales (USD billion) |
|---|---|
| 2023 | 4.7 |
| 2024 | 5.3 |
| 2025 | 6.0 |
| 2026 | 6.8 |
| 2027 | 7.7 |
By 2027, FARXIGA could approach USD 8 billion in annual sales if current trends persist.
Regional Dynamics
- North America dominates with approximately 50% of sales, fueled by high T2DM prevalence and favorable reimbursement.
- Europe accounts for ~25%, with growth driven by new indications.
- Emerging markets (China, India, Brazil) contribute increasing revenue, estimated to comprise 15–20% of total sales by 2027, supported by expanding healthcare access.
Competitive Outlook
FARXIGA faces competition from both other SGLT2 inhibitors and newer classes such as GLP-1 receptor agonists. The recent approvals for heart failure (FDA 2020, EMA 2021) and CKD (FDA 2021) significantly expand the market and provide opportunities for growth, especially outside traditional diabetes management.
Market share could fluctuate based on:
- Effectiveness of AstraZeneca's marketing strategies.
- Clinical trial outcomes validating expanded indications.
- Price competition and potential generics entry post-patent expiry.
Regulatory and Patent Status
Patents covering dapagliflozin extend until approximately 2030 in key markets. Patent challenges or delays could influence sales timelines. Regulatory agencies are scrutinizing safety profiles, which could impact approvals or labels.
Summary
FARXIGA's sales trajectory is positive, driven by expanding indications and increased awareness of cardiovascular and renal benefits. Market penetration will depend on clinical data validation, regulatory developments, and competitive actions.
Key Takeaways
- FARXIGA had global sales of about USD 4.2 billion in 2022.
- Sales are projected to grow at approximately 12% CAGR from 2023 to 2027.
- Its expanding indications in heart failure and CKD are major growth drivers.
- Competition remains significant, with patent expiry looming risk.
- Regional growth is strongest in North America and emerging markets.
FAQs
1. What are recent FDA approvals impacting FARXIGA?
In 2020, FDA approved FARXIGA for the treatment of HFrEF with or without diabetes. In 2021, it received approval for CKD, broadening its use beyond glycemic control.
2. How does FARXIGA compare to competitors like Jardiance?
Both are SGLT2 inhibitors with similar mechanisms; FARXIGA has slight advantages in specific indications but faces stiff competition. Market share differences are marginal and vary by region.
3. What are the primary risks to sales growth?
Patent expirations, safety concerns, and regulatory restrictions could temper growth. Price pressures and generic entry are also threats.
4. Which regions are expected to drive future growth?
Emerging markets and Europe are key, with improving healthcare infrastructure and broader indication approvals expanding patient access.
5. How might upcoming clinical trials influence future sales?
Positive outcomes in trials evaluating long-term cardiovascular and renal benefits could reinforce FARXIGA's positioning and growth potential.
References
- International Diabetes Federation (IDF). Diabetes Atlas, 10th Edition, 2021.
- AstraZeneca. Annual Reports, 2020–2022.
- FDA Press Releases. Approvals for FARXIGA, 2020–2022.
- MarketWatch. SGLT2 inhibitor market analysis, 2022.
- IQVIA. Global prescription trends, 2022.
More… ↓
