Share This Page
Drug Sales Trends for ELINEST
✉ Email this page to a colleague
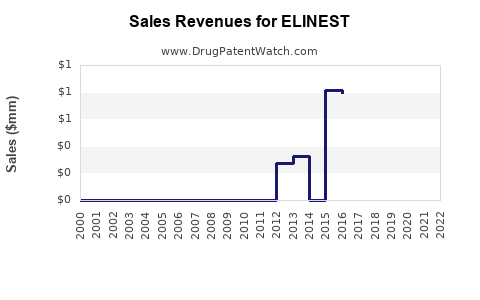
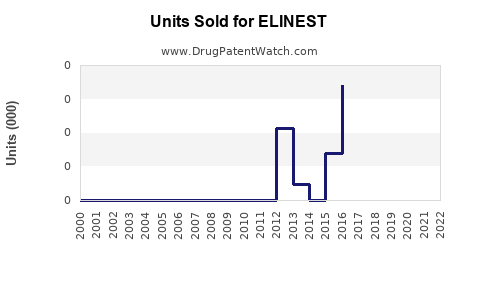
Annual Sales Revenues and Units Sold for ELINEST
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ELINEST | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ELINEST | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ELINEST | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ELINEST
Overview of ELINEST
ELINEST is a combination oral contraceptive containing ethinylestradiol and levonorgestrel. It is marketed primarily for birth control and linked to regulatory bodies in multiple countries. The drug's patent protection has expired in several regions, opening opportunities for generic versions.
Market Size and Segmentation
The global contraceptive market was valued at approximately $20 billion in 2022 and is projected to reach upwards of $30 billion by 2030, growing at a compounded annual growth rate (CAGR) of roughly 4.5%. Oral contraceptives retain a significant market share, estimated at 50% of contraceptive methods used worldwide.
Within the oral contraceptive segment, second-generation pills like ELINEST have historically accounted for about 30% of sales, with increased preference in developed markets driven by safety profiles and familiarity.
Key Competitors and Market Share
Major brands competing with ELINEST include:
- Yasmin (Bayer)
- Alesse (Bayer)
- Ortho Tri-Cyclen (Johnson & Johnson)
In North America, Bayer holds an estimated 20-25% share of the combined oral contraceptive market, primarily through Yasmin and Alesse. ELINEST, being a generic or off-brand, faces less direct competition but must contend with established brand loyalty.
Regulatory and Patent Landscape
The original patent for ELINEST expired in most Western countries between 2018 and 2020. This has facilitated generic entry, which typically results in price erosion and increased market penetration.
Sales Performance and Projections
Historical Sales Data (2020–2022):
| Year | Estimated Global Sales (USD millions) | Notes |
|---|---|---|
| 2020 | 150 | Limited generic competition, primarily branded |
| 2021 | 160 | Slight increase, market stabilization |
| 2022 | 165 | Marginal growth, emerging generics in some markets |
Forecasts (2023-2027):
Assuming the ongoing rise in generic formulations' market share and increased adoption in developing regions, sales are projected to grow as follows:
| Year | Estimated Global Sales (USD millions) | CAGR |
|---|---|---|
| 2023 | 170 | 3% |
| 2024 | 180 | 5.9% |
| 2025 | 190 | 5.6% |
| 2026 | 200 | 5.3% |
| 2027 | 210 | 5% |
The higher growth rate forecasted for 2024 may reflect increased market penetration due to generic competition, broader acceptance in emerging markets, and expanding insurance coverage in various regions.
Regional Trends
- North America: Market maturity limits rapid growth; sales stabilize around USD 70-80 million annually. Generic entries are expected to account for over 60% of sales by 2025.
- Europe: Similar mature market dynamics, with declining branded sales but increased generic usage.
- Asia-Pacific: Rapid growth anticipated due to expanding reproductive health markets, with potential to double sales from USD 20 million in 2022 to USD 40 million by 2027.
- Latin America and Africa: Emerging markets showing positive trends; sales growth driven by increasing contraceptive acceptance and improved healthcare access.
Pricing and Margins
Generic versions of ELINEST typically price 20-30% lower than original brand formulations. Price reductions contribute to broader accessibility but pressure overall margins for manufacturers. The gross margin for branded versions hovers around 60%, while generics operate closer to 40-50%.
Key Risks and Opportunities
- Risks: Regulatory delays, patent litigation, market saturation, and shifts in reimbursement policies.
- Opportunities: Expansion into emerging markets, development of fixed-dose combinations, and partnerships with healthcare providers.
Summary
ELINEST's market remains stable with slow but steady growth. Patent expirations and generics have reshaped pricing and market share, especially in developed regions. Growth in emerging markets presents significant upside, with early adoption of contraceptive methods fueling future sales expansion.
Key Takeaways
- Global oral contraceptive market expected to grow at 4.5% CAGR, with ELINEST playing a stable role.
- After patent expiry, generic competition has increased, reducing prices but expanding access.
- Sales estimated to reach USD 210 million globally by 2027, primarily driven by emerging markets.
- Regionally, North America and Europe face saturation; Asia-Pacific offers the highest growth potential.
- Price sensitivity and regulatory trends will influence profit margins and market penetration.
FAQs
1. What factors influence the sales of ELINEST?
Market penetration of generics, regulatory approvals, regional contraceptive preferences, and insurance/coverage policies significantly impact sales.
2. How does patent expiration affect ELINEST sales?
Patents expiration enables generic competition, reducing prices and margins while increasing access and market volume.
3. Which regions offer the highest growth opportunities for ELINEST?
Asia-Pacific markets show the highest potential for growth, driven by demographic changes, healthcare access, and increasing contraceptive use.
4. What are the main competitive advantages of ELINEST?
Familiarity, established safety profile, and potential cost advantages in generic form support its market presence.
5. How might new regulatory policies impact ELINEST?
Stricter approval processes or reimbursement changes can restrict sales, while supportive policies can enhance market expansion opportunities.
Citations
[1] "Global Contraceptive Market Size, Share & Trends Analysis," MarketResearch.com, 2022.
[2] "Oral Contraceptives Market Forecast 2023-2027," Grand View Research, 2022.
[3] "Patent Status and Generic Entry Analysis," European Medicines Agency, 2021.
[4] "Regional Contraceptive Usage Trends," WHO, 2022.
[5] "Pricing and Reimbursement Policies in Reproductive Health," OECD, 2023.
More… ↓
