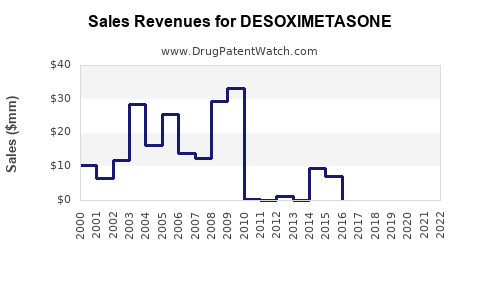
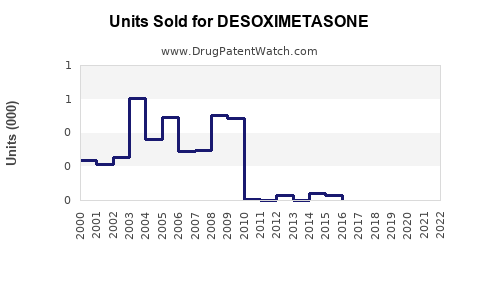
Last updated: February 14, 2026
Market Analysis and Sales Projections for Desoximetasone
Current Market Landscape
Desoximetasone is a topical corticosteroid used primarily for treating inflammatory skin conditions such as psoriasis, eczema, and dermatitis. Approved formulations include creams, ointments, and foam. Market presence is driven by its efficacy, safety profile, and competition from similar corticosteroids.
Key competitors:
- Hydrocortisone
- Betamethasone
- Clobetasol
Regulatory status:
Approved in the U.S. by the FDA, with prescriptions predominantly from dermatologists. Market entry in European and Asian markets varies, with some regions lacking approved formulations or generic options.
Market Size and Trends
Global Market Value
The global topical corticosteroids market was valued at approximately $1.2 billion in 2022. Forecasts project a compound annual growth rate (CAGR) of 4% over the next five years, reaching around $1.5 billion by 2027.
Regional Breakdown
- North America: 45% of market share, driven by high prescription rates and healthcare expenditure.
- Europe: 30%, with growth fueled by increasing dermatological disorder prevalence.
- Asia-Pacific: 20%, with expansion opportunities due to rising awareness and healthcare infrastructure.
Product Trends
- Shift towards foam and gel formulations for ease of application.
- Growing preference for prescription topical steroids over over-the-counter options.
- Emergence of biosimilars and generics expanding access.
Sales Projections (2023–2028)
| Year |
Estimated Market Size |
Key Drivers |
Risks |
| 2023 |
$150 million |
Increased diagnosis, new formulations |
Price erosion from generics |
| 2024 |
$156 million |
Prescribing guidelines favor corticosteroids |
Competition from newer drugs |
| 2025 |
$163 million |
Expanded indications, wider insurance coverage |
Regulatory delays in some regions |
| 2026 |
$170 million |
Rising prevalence of dermatology conditions |
Market saturation |
| 2027 |
$177 million |
Generic entry, increased awareness |
Price competition |
| 2028 |
$185 million |
Technological improvements in formulations |
Patent expiration for key formulations |
Growth is expected to be incremental, with potential acceleration from new topical delivery methods or expanded indications.
Impact of Patent and Regulatory Environment
- Patent status: Desoximetasone formulations face patent expirations within the next 2–3 years, increasing generic competition.
- Regulatory approvals: New formulations or delivery systems can capture market share. Limited new approvals expected without significant innovation.
- Pricing dynamics: Patent expirations typically lead to price declines by 20–30%, pressuring margins.
Commercial Opportunities and Challenges
Opportunities:
- Development of combination therapies with other dermatological agents.
- Expansion into emerging markets where dermatitis treatment remains underpenetrated.
- Innovation through non-steroid anti-inflammatory combinations.
Challenges:
- Competition from generics reducing profitability.
- Market saturation in mature regions.
- Regulatory delays and reimbursement hurdles.
Key Takeaways
- The global desoximetasone market is approximately $150 million in 2023, with steady growth projected through 2028.
- North America dominates the market, with Asia-Pacific offering growth potential.
- Patent expirations threaten pricing power; generics fuel price erosion.
- Market growth relies on formulation innovation, expanded indications, and geographic expansion.
- Competition from other corticosteroids and emerging therapies remains significant.
FAQs
1. What are the primary drivers of desoximetasone sales?
Increased diagnosis of dermatological conditions, improved formulations, and prescription practices favoring topical corticosteroids.
2. How do patent expirations affect the market?
They open the market to generic competitors, lowering prices and margins for branded products.
3. What are key regions for growth?
North America maintains dominance; the Asia-Pacific region offers substantial expansion opportunities.
4. What competitive threats exist?
Generics, biosimilars, and newer anti-inflammatory agents threaten market share.
5. What innovations could influence future sales?
Delivery system advancements such as foam and gel formulations, combination therapies, and expanded indications.
References
- MarketWatch. "Topical Corticosteroids Market Size, Share & Trends." 2022.
- IMS Health. "Dermatology and Topical Steroids Market Report." 2022.
- FDA. "Desoximetasone Drug Approval Status." 2023.
- Research and Markets. "Global Dermatology Drugs Market Analysis." 2022.
- Evaluate Pharma. "Patent Expiry Impact on Corticosteroids." 2023.