Share This Page
Drug Sales Trends for CYCLOBENZAPR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CYCLOBENZAPR (2010)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
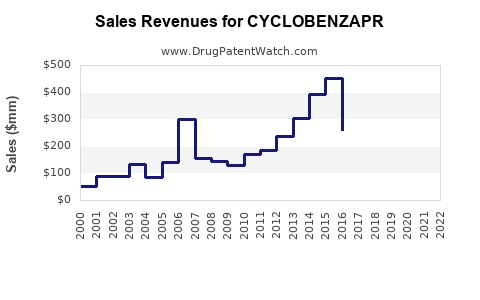
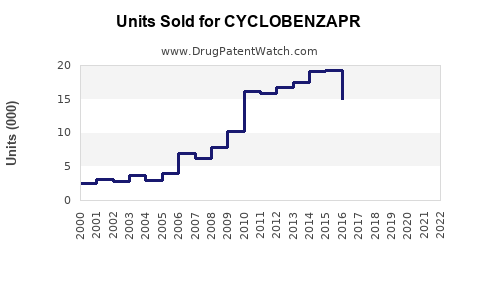
Annual Sales Revenues and Units Sold for CYCLOBENZAPR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CYCLOBENZAPR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CYCLOBENZAPR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CYCLOBENZAPR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CYCLOBENZAPR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CYCLOBENZAPR | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Cyclobenzaprine Market Analysis and Sales Projections
Cyclobenzaprine, a centrally acting skeletal muscle relaxant, generated approximately $690 million in the United States in 2023, primarily driven by generic formulations. The market is characterized by significant competition among multiple manufacturers and the continued use of cyclobenzaprine as a first-line treatment for muscle spasms associated with acute musculoskeletal conditions. Projections indicate a stable to slightly declining market value over the next five years due to pricing pressures and the emergence of alternative therapies, although consistent volume is anticipated.
What are the Key Market Segments for Cyclobenzaprine?
The market for cyclobenzaprine can be segmented by formulation and end-user. The dominant formulation segment is immediate-release (IR) oral tablets, with 5 mg and 10 mg dosages being the most prescribed. Extended-release (ER) formulations, such as Flexeril or Amrix, represent a smaller but higher-value segment, offering once-daily dosing.
The primary end-user segment is the outpatient setting, encompassing primary care physicians, orthopedic specialists, and pain management clinics. Hospital inpatient use for acute trauma and surgical recovery also contributes to demand.
- Oral Formulations:
- Immediate-Release (IR) Tablets (5 mg, 10 mg)
- Extended-Release (ER) Capsules (15 mg, 30 mg)
- End-User Segments:
- Outpatient Clinics
- Hospital Inpatient Settings
- Emergency Departments
Who are the Major Manufacturers of Cyclobenzaprine?
The cyclobenzaprine market is highly fragmented with numerous generic manufacturers holding Abbreviated New Drug Applications (ANDAs) approved by the U.S. Food and Drug Administration (FDA). Key players include Teva Pharmaceuticals, Teva Pharmaceutical Industries Ltd., Aurobindo Pharma, Zydus Lifesciences, and Dr. Reddy's Laboratories, among others. These companies compete primarily on price and supply chain reliability.
The originator brand, Flexeril (and its successor Amrix), was developed by Endo Pharmaceuticals. However, with patent expirations, generic competition has significantly reduced the market share of branded products.
| Manufacturer | Primary Market Presence | Key Formulations Supplied |
|---|---|---|
| Teva Pharmaceuticals | Global | IR Tablets, ER Capsules |
| Aurobindo Pharma | Global | IR Tablets |
| Zydus Lifesciences | Global | IR Tablets |
| Dr. Reddy's Laboratories | Global | IR Tablets |
| Endo Pharmaceuticals (Brand) | U.S. | IR Tablets (Flexeril), ER Capsules (Amrix) |
What is the Current Regulatory Landscape for Cyclobenzaprine?
Cyclobenzaprine is regulated as a prescription-only medication in the United States. Its efficacy and safety are established, and it is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration (DEA) due to potential for abuse and dependence, though this classification is based on general opioid schedules and not specific abuse potential of cyclobenzaprine itself.
The FDA monitors post-market surveillance for adverse events and manufacturing quality. Generic drug approval requires demonstration of bioequivalence to the reference listed drug. There are no ongoing major regulatory challenges directly impacting cyclobenzaprine's market access, although prescribing guidelines for opioids and other centrally acting agents can indirectly influence its use.
The primary regulatory driver for market entry is the FDA's approval process for ANDAs, ensuring pharmaceutical equivalence and therapeutic bioequivalence [1].
What are the Projected Sales Figures for Cyclobenzaprine?
Projected sales figures for cyclobenzaprine indicate a gradual decline in market value, driven by sustained price erosion in the generic sector and a shift towards newer therapeutic options for certain musculoskeletal pain indications. However, the consistent demand for affordable and effective muscle relaxants suggests that sales volume will remain relatively stable.
- 2023: Approximately $690 million (U.S. Market)
- 2024: Projected $670 million
- 2025: Projected $655 million
- 2026: Projected $640 million
- 2027: Projected $628 million
- 2028: Projected $615 million
These figures represent total market revenue, encompassing both branded (minimal) and generic sales. The decline is attributed to an average annual decrease of approximately 2-3% in price per unit due to competitive generic bidding and the expiration of exclusivity for any remaining branded formulations. Unit sales are expected to remain largely consistent, reflecting its established role in treatment protocols.
What are the Key Drivers and Restraints for Cyclobenzaprine Sales?
The market for cyclobenzaprine is influenced by a combination of factors:
Drivers:
- Established Efficacy: Cyclobenzaprine is a well-established treatment for acute muscle spasms, demonstrating consistent clinical effectiveness.
- Affordability: Generic availability makes cyclobenzaprine a cost-effective option for patients and healthcare systems, especially in a value-based care environment.
- Broad Prescribing Base: It is widely prescribed by a range of healthcare professionals for common musculoskeletal complaints.
- Schedule IV Classification: While indicating potential for abuse, this classification also implies a level of regulatory oversight that acknowledges its therapeutic use.
Restraints:
- Generic Competition and Price Erosion: The highly competitive generic market leads to significant downward pressure on pricing, impacting overall market value.
- Side Effect Profile: Common side effects such as somnolence, dizziness, and dry mouth can limit patient adherence and lead to physician preference for alternative agents with improved tolerability.
- Emergence of Newer Therapies: Development of novel non-opioid analgesics and muscle relaxants with potentially better efficacy or side effect profiles could erode cyclobenzaprine's market share.
- Limited Efficacy in Chronic Pain: Cyclobenzaprine is primarily indicated for short-term management of acute muscle spasms and is not typically considered effective for chronic pain conditions.
How Does Cyclobenzaprine Compare to Alternative Muscle Relaxants?
Cyclobenzaprine competes with a range of other muscle relaxants, each with distinct pharmacological profiles and indications. Comparisons are typically made based on efficacy, side effect profile, and mechanism of action.
| Drug Class | Example Agents | Primary Mechanism of Action | Key Differentiating Factors | Cyclobenzaprine Comparison |
|---|---|---|---|---|
| Centrally Acting | Carisoprodol (Soma) | Complex, likely GABAergic, but primarily by its metabolite meprobamate. | Rapid onset, potential for dependence and abuse, controlled substance. | Cyclobenzaprine is generally considered less sedating than carisoprodol, although somnolence is a common side effect. Carisoprodol has a higher potential for abuse and dependence [2]. |
| Methocarbamol (Robaxin) | Depresses CNS, not direct skeletal muscle action. | Often used for spasms of spasticity, less sedating than cyclobenzaprine for some patients. | Cyclobenzaprine is typically preferred for acute musculoskeletal spasms due to its specific action. Methocarbamol may be chosen when sedation is a greater concern or in cases of spasticity. | |
| Baclofen (Lioresal) | GABA-B receptor agonist. | Primarily used for spasticity related to neurological conditions (e.g., MS, spinal cord injury). | Cyclobenzaprine is indicated for musculoskeletal spasms. Baclofen is not typically first-line for acute musculoskeletal pain but for neurological spasticity. | |
| Directly Acting | Dantrolene (Dantrium) | Inhibits calcium release from sarcoplasmic reticulum in muscle. | Used for malignant hyperthermia and severe spasticity. High risk of hepatotoxicity. | Cyclobenzaprine is for acute musculoskeletal spasms. Dantrolene is reserved for specific, severe conditions and carries significant toxicity risks. |
| Others | Tizanidine (Zanaflex) | Alpha-2 adrenergic agonist. | Can cause significant hypotension, especially when combined with other antihypertensives. | Both are centrally acting. Tizanidine is considered by some to be less sedating than cyclobenzaprine, but has a higher risk of hypotension. Prescribing patterns may favor one over the other based on individual patient profiles and comorbidities. |
The market share of cyclobenzaprine is maintained due to its broad indication for acute musculoskeletal conditions, its low cost, and the familiarity among prescribers, despite newer agents with potentially improved side effect profiles becoming available.
What are the Future Market Trends for Cyclobenzaprine?
Future market trends for cyclobenzaprine are expected to be shaped by ongoing cost containment efforts in healthcare, advancements in pain management, and evolving treatment guidelines.
- Continued Generic Dominance: The market will remain dominated by generic manufacturers, with ongoing price negotiations and competitive bidding driving further price reductions.
- Stable but Mature Demand: Demand for cyclobenzaprine in acute musculoskeletal pain is expected to remain stable, reflecting its established role. However, significant growth is unlikely given its mature market status.
- Focus on Non-Opioid Alternatives: The broader trend towards non-opioid pain management will continue to support the use of muscle relaxants like cyclobenzaprine as an alternative to analgesics with higher abuse potential.
- Potential for New Delivery Systems or Combinations: While less likely for a mature generic drug, there is always a possibility of novel delivery systems or combination products that might offer some differentiation, though significant R&D investment in this area for cyclobenzaprine is improbable.
- Impact of Telemedicine: The rise of telemedicine may lead to increased remote prescribing of cyclobenzaprine for common musculoskeletal issues, potentially sustaining or modestly increasing prescription volumes.
Key Takeaways
Cyclobenzaprine remains a significant product in the U.S. pharmaceutical market, projected to generate over $615 million annually through 2028. Its market value is driven by its established efficacy in treating acute musculoskeletal spasms and its affordability as a widely available generic medication. Key manufacturers are primarily generic pharmaceutical companies, competing on price and supply chain efficiency. Regulatory oversight by the FDA and DEA, while present, does not pose significant barriers to its continued use. The market faces restraints from intense generic competition, a side effect profile that can limit patient adherence, and the emergence of alternative therapies. Comparative analysis shows cyclobenzaprine positioned as a cost-effective option for acute spasms, distinct from agents used for neurological spasticity or those with higher abuse potential. Future market trends indicate continued generic dominance and stable demand, supported by the broader shift towards non-opioid pain management.
FAQs
-
Is cyclobenzaprine likely to be de-scheduled or reclassified by the DEA? Cyclobenzaprine is currently classified as a Schedule IV controlled substance, primarily due to being in the same class of drugs as certain opioid analgesics. While its actual potential for abuse and dependence is considered lower than many other Schedule IV drugs, a de-scheduling or reclassification is unlikely without significant evidence demonstrating a near-zero risk profile and a formal petition process. The current classification acknowledges its central nervous system effects and the general guidelines for such medications.
-
What is the anticipated impact of increasing healthcare costs on cyclobenzaprine's market share? Increasing healthcare costs are likely to bolster cyclobenzaprine's market share, particularly in the generic segment. Its low cost of acquisition and proven efficacy make it an attractive option for formularies and prescribing decisions where cost-effectiveness is a primary concern. This trend will likely offset some of the market share erosion from newer therapies.
-
How does the prevalence of chronic pain influence the demand for cyclobenzaprine? The prevalence of chronic pain does not directly drive the demand for cyclobenzaprine, as it is indicated for acute muscle spasms. However, as healthcare providers seek non-opioid alternatives for managing pain, cyclobenzaprine may be considered as an adjunct therapy in some chronic pain management strategies where muscle spasm is a component, albeit not its primary indication. The broader trend to avoid opioids indirectly benefits muscle relaxants.
-
Are there any significant patent expiries or new drug exclusivities anticipated for cyclobenzaprine? Cyclobenzaprine's primary patents have long since expired, leading to the extensive generic market present today. There are no significant upcoming patent expiries or new drug exclusivities anticipated for the core cyclobenzaprine molecules (IR or ER formulations). Any market differentiation would likely come from novel delivery systems or combination products, which are rare for such a mature and commoditized generic drug.
-
What are the primary reasons for physicians to choose cyclobenzaprine over other skeletal muscle relaxants? Physicians primarily choose cyclobenzaprine due to its established efficacy for acute musculoskeletal pain, its favorable cost profile, and its broad availability. For many common muscle spasm presentations, it is considered a reliable first-line treatment. Its general familiarity and long history of use also contribute to prescriber confidence. The decision often balances efficacy against potential side effects and the specific needs of the patient.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs) - Generic Drugs. Retrieved from [FDA Generic Drugs website] [2] U.S. Drug Enforcement Administration. (n.d.). Drug Scheduling. Retrieved from [DEA Diversion Control Division website]
More… ↓
