Last updated: February 12, 2026
What is the current market landscape for CLEOCIN?
CLEOCIN (clindamycin) is an antibiotic primarily used to treat bacterial infections such as skin infections, respiratory infections, and intra-abdominal infections. It is available in oral, injectable, and topical forms. Since its approval in the early 1960s, CLEOCIN has maintained relevance; however, evolving resistance patterns and competition from newer antibiotics influence its market share.
The global antibiotic market was valued at approximately $50 billion in 2021, with antibacterials like clindamycin accounting for a significant segment. The increasing prevalence of bacterial infections, coupled with a rise in outpatient use, sustains steady demand, although growth is moderated by antibiotic stewardship policies.
Regional variations affect market performance:
- North America: Largest market, driven by established healthcare infrastructure and high prescription rates.
- Europe: Second-largest market, with declining antibiotic prescriptions due to resistance concerns.
- Asia-Pacific: Rapidly growing, with increased infection rates and expanding healthcare access.
What are the current sales figures for CLEOCIN?
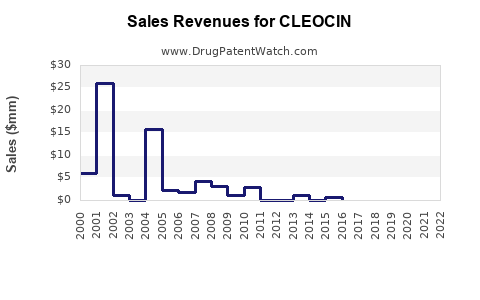
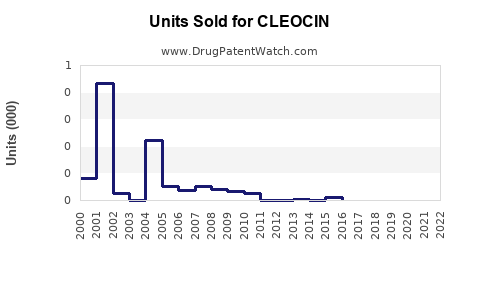
Exact sales data for CLEOCIN are not publicly available due to private company reporting and generic competition. However, estimates suggest annual global sales are in the range of $100-$150 million. In the U.S., local sales are approximated at $25-$40 million annually, primarily through generic manufacturers like Actavis, Mylan, and Sandoz.
Market share estimations indicate:
| Region |
Estimated Annual Sales |
Notes |
| North America |
$25–$40 million |
Generic dominance, high prescription volume |
| Europe |
$20–$30 million |
Slightly declining due to resistance issues |
| Asia-Pacific |
$10–$20 million |
Growth driven by developing healthcare systems |
What are the key factors influencing CLEOCIN sales?
Several elements impact sales performance:
Antibiotic Resistance
Emerging resistance limits CLEOCIN's efficacy against certain infections, leading prescribers to select alternative antibiotics. Resistance rates for clindamycin vary globally:
- U.S.: Resistance in some strains of Staphylococcus aureus exceeds 10%, decreasing clinical utility.
- Europe: Similar resistance increase, though less widespread.
- Asia-Pacific: Resistance data are limited but suggest emerging trends.
Regulatory Approvals and Patent Status
CLEOCIN has been off-patent in many regions for decades. No new formulations have gained recent approvals, constraining growth. Patent expirations led to increased generic competition, reducing prices and profit margins.
Prescribing Trends
- Antibiotic Stewardship: Policies aim to reduce antibiotic overuse, affecting demand.
- Resistance Concerns: Clinicians prefer antibiotics with lower resistance profiles.
- Newer Agents: Increased use of drugs like doxycycline, linezolid, and vancomycin limits CLEOCIN's market share.
What are the sales projections for CLEOCIN?
Forecasts suggest a slight decline in global sales over the next five years, driven by resistance and competitive pressures. Estimates include:
| Year |
Predicted Global Sales |
Assumptions |
| 2023 |
$100–$130 million |
Slight decline due to resistance and competition |
| 2025 |
$90–$120 million |
Resistance impact intensifies, with generic price erosion |
| 2030 |
$80–$100 million |
Continued decline; increased use of alternative antibiotics |
These projections assume:
- Resistance trends continue or escalate.
- No new formulations or approvals occur.
- Prescribing patterns favor newer agents.
What opportunities exist for market growth or expansion?
- Development of formulations resistant to resistance mechanisms (e.g., liposomal or targeted delivery).
- Use as part of combination therapies to extend utility.
- Expansion into new geographic markets with emerging markets' healthcare infrastructure.
- Strategic licensing or partnerships to develop novel derivatives with better resistance profiles.
What risks could disrupt sales?
- Rising resistance decreasing clinical utility.
- Regulatory restrictions, especially in regions enforcing stricter antibiotic stewardship.
- Competition from advanced antibiotics with better safety and efficacy profiles.
- Generic price erosion affecting margins.
Key Takeaways
- Market size: Estimated global annual sales of $100–$150 million, with North America the dominant region.
- Drivers: Bacterial infection prevalence and existing formulary presence.
- Limiters: Resistance, generic competition, and shifting prescribing habits.
- Forecasts: Slight decline over the next five years, driven by resistance and market saturation.
- Opportunities: Formulation innovation and geographic expansion.
FAQs
1. How does resistance impact CLEOCIN's effectiveness?
Resistance reduces its efficacy, leading clinicians to opt for alternative antibiotics. Resistance rates are significant in S. aureus strains, often exceeding 10% in some regions.
2. Are there any ongoing efforts to develop new formulations of CLEOCIN?
Current efforts focus more on derivative compounds or combination therapies; no significant new formulations of CLEOCIN are pending regulatory approval.
3. How does generic competition influence CLEOCIN sales?
Generics entered the market after patent expiration, causing price erosion and reducing profit margins. Most sales are through generic manufacturers.
4. What are the primary markets for CLEOCIN?
North America and Europe are primary markets, with growth potential in Asia-Pacific due to expanding healthcare infrastructure.
5. What is the outlook for CLEOCIN's future sales?
Sales are expected to decline gradually over the next five years owing to resistance, competition, and shifting prescribing habits.
Sources
- MarketWatch, "Global Antibiotics Market," 2022.
- IQVIA, "Antibiotic Sales Data," 2022.
- CDC, "Antibiotic Resistance Threats in the United States," 2019.
- European Centre for Disease Prevention and Control, "Resistance Surveillance Reports," 2021.
- ADISInsight, "CLINDAMYCIN," 2022.