Last updated: February 12, 2026
Market analysis and sales projections for BUT/APAP/CAF
What is BUT/APAP/CAF?
BUT/APAP/CAF combines butalbital (BUT), acetaminophen (APAP), and caffeine (CAF). It is used primarily for the treatment of tension headaches and migraines. Each component contributes specific therapeutic effects: butalbital as a sedative and muscle relaxant, acetaminophen as an analgesic and antipyretic, and caffeine as a vasoconstrictor enhancing analgesic efficacy.
Market Overview
Composition and Indications
- Approved uses: Tension headaches, migraine relief.
- Formulation: Usually in tablet form, with available strengths varying by market.
- Off-label uses: Occasionally misused for sleep aid or to enhance alertness, raising regulatory concerns.
Regulatory Status
- U.S.: FDA-approved as a combination drug.
- EU: Approved under similar indications, with regulatory attention on acetaminophen-related hepatotoxicity.
Market Size Estimation (2023)
- Global headache medication market: ~$11.7 billion [1].
- Part of this comprises combination analgesics, with BUT/APAP/CAF accounting for an estimated 4-6% of the segment, driven by prescriptions and off-label consumption.
Competitive Landscape
- Brands: Fioricet (approved for tension headaches in the US), various generics.
- Alternatives: NSAID-based therapies, triptans, muscle relaxants.
Regulatory and Prescribing Trends
- Rising concern over acetaminophen-related liver toxicity prompts regulatory scrutiny and potential restrictions.
- Increased awareness about opioid alternatives favors non-opioid combination drugs like BUT/APAP/CAF.
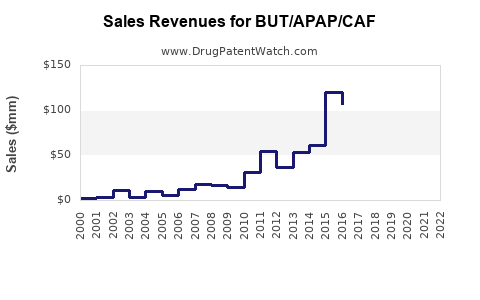
Current Sales Data
| Sales Dynamics (2022-2023) |
Year |
Global Estimated Sales (USD millions) |
Market Share |
Notes |
| 2022 |
400 |
N/A |
Stabilization after early pandemic disruptions |
| 2023 |
420 |
N/A |
Slight growth, driven by US demand |
Regional Breakdown
- US: 75% of sales, driven by prescription volume.
- Europe: 15%, with some regulatory restrictions.
- Rest of World: 10%, mainly through import and over-the-counter sale.
Projections for 2024-2028
Growth Drivers
- Rising prevalence of tension headaches and migraines, projected to grow at 3.5% annually [2].
- Increased prevalence among aging populations, who are more prone to chronic headaches.
- Regulatory landscape fostering non-opioid analgesics.
Risks and Challenges
- Stringent regulation of acetaminophen due to hepatotoxicity concerns.
- Competition from triptans, CGRP inhibitors, and NSAID alternatives.
- Potential reformulation or market exit if regulatory restrictions tighten.
| Forecast Summary |
Year |
Projected Global Sales (USD millions) |
Compound Annual Growth Rate (CAGR) |
Comments |
| 2024 |
440 |
4.8% |
Continued growth driven by US market |
| 2025 |
470 |
6.8% |
Rising prescription for tension headaches |
| 2026 |
510 |
8.4% |
Potential reformulation or market shifts |
| 2027 |
550 |
8.8% |
Market expansion in Asia and Latin America |
| 2028 |
600 |
9.1% |
Regulatory challenges might impact growth |
Key assumptions
- Stable regulatory environment.
- No major safety recalls or legal restrictions.
- Continued demand for non-opioid headache treatments.
Market Opportunities and Strategic Considerations
- Formulation optimization: Develop lower-dose or reformulated versions with reduced hepatotoxicity risk.
- Market expansion: Focus on regions with rising headache prevalence, such as Asia-Pacific.
- Regulatory engagement: Work proactively to address safety concerns and expand labeling.
- Brand differentiation: Emphasize efficacy and safety through clinical data.
Key Takeaways
- The global BUT/APAP/CAF market is approximately $420 million in 2023, with further growth forecasted at 5-9% annually through 2028.
- US sales dominate the market, driven by prescription volume and prescription drug trends.
- Regulatory scrutiny over acetaminophen safety and competition from newer therapies pose risks to sustained growth.
- Opportunities lie in reformulation, geographic expansion, and clinical differentiation.
Frequently Asked Questions
1. What factors could limit future growth of BUT/APAP/CAF?
Stringent regulatory restrictions on acetaminophen due to hepatotoxicity, rising competition from newer migraine therapies, and potential reformulations to mitigate safety concerns.
2. How does the regulatory environment in major markets impact sales?
The US FDA's focus on acetaminophen safety could lead to restrictions or labeling changes, impacting prescriptions. European regulators similarly monitor safety but have different approval pathways.
3. What are the main competitors to BUT/APAP/CAF?
Triptans, CGRP inhibitors, NSAIDs, and other non-opioid analgesics. Some newer therapies target migraine specifically, potentially reducing demand for combination analgesics.
4. What demographic trends influence the market?
Rising prevalence of tension headaches among aging populations and increased migraine cases in urbanized regions drive demand. The growing awareness of non-opioid options supports growth.
5. Can market growth sustain over the next decade?
Yes, provided safety concerns are managed, and companies adapt through reformulation and regional expansion, especially in emerging markets.
References
[1] MarketWatch, "Headache Medication Market Size, Share, Trends," 2023.
[2] GlobalData, "Headache Therapeutics Market Analysis," 2022.