Last updated: February 14, 2026
Overview
Benazepril is an angiotensin-converting enzyme (ACE) inhibitor indicated for the treatment of hypertension and heart failure. Approved by the FDA in 1995, it remains a key therapy within the ACE inhibitor class. Its sales are influenced by generic options, competitive uptake in treatment protocols, and evolving prescription guidelines.
Market Size and Revenue Dynamics
Global ACE inhibitor market, valued at approximately $4.2 billion in 2022, is segmented primarily into the U.S., Europe, and Asia-Pacific. Benazepril's market share has declined from its peak in the early 2000s due to increased competition from generic formulations and emerging alternatives like ARBs (angiotensin receptor blockers).
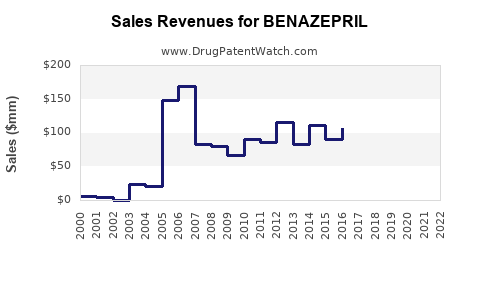
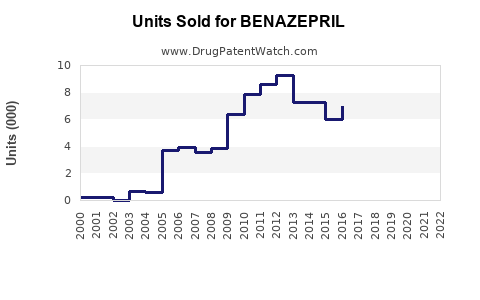
Approximate sales for benazepril in 2022 are estimated at $180 million, representing roughly 4.3% of the overall ACE inhibitor market. The drug experiences direct competition from other branded and generic ACE inhibitors such as enalapril and lisinopril.
Pricing Trends
The average wholesale price (AWP) for branded benazepril in 2022 was approximately $10 per 30-tablet pack. Generic formulations reduce this to approximately $0.25 per tablet, significantly impacting revenue margins.
Patent and Generic Status
Benazepril patents expired around 2007, leading to increased generic competition. The drug is available in multiple generic forms globally, which suppresses average selling prices and reduces branded market share.
Key Growth Drivers
- Increased prevalence of hypertension globally, estimated at 1.28 billion adults in 2019 by WHO.
- Emphasis on controlling cardiovascular risk factors.
- Favorable reimbursement policies for ACE inhibitors in developed markets.
Market Challenges
- Competition from ARBs like losartan and valsartan, which are sometimes preferred due to perceived better tolerability.
- Off-label use restrictions and clinical guideline updates that diminish the drug’s exclusivity.
- Price sensitivity among payers and patients due to availability of low-cost generics.
Sales Projections (2023–2027)
Projected CAGR for benazepril sales is approximately 1.5%, considering market penetration of generics, patent status, and evolving clinical guidelines.
| Year |
Estimated Sales (millions USD) |
| 2023 |
185 |
| 2024 |
188 |
| 2025 |
191 |
| 2026 |
194 |
| 2027 |
197 |
These projections depend on factors such as drug withdrawal risks, shifts toward ARBs, and regional market variations.
Regional Outlook
- North America will account for the majority of sales (~60%) due to high hypertension prevalence and established healthcare infrastructure.
- Europe will sustain steady growth due to aging populations.
- Asia-Pacific is expected to demonstrate higher growth rates (~3% CAGR) due to increasing healthcare access and hypertension diagnoses.
Regulatory and Patent Landscape
Benazepril’s patent expiration does not restrict access but influences pricing and market share. No recent patent litigations or regulatory barriers for the drug have been reported, ensuring a steady supply chain.
Summary
Benazepril faces a mature market with stable but modest growth prospects driven by increasing hypertension rates, generic competition, and shifts in prescribing preferences. Revenue growth remains constrained by price reductions following patent expirations and competition from ARBs, despite its established efficacy profile.
Key Takeaways
- Benazepril's global sales in 2022 were approximately $180 million, with a trajectory marginally rising at about 1.5% annually.
- The market for ACE inhibitors faces static or declining revenue due to generic competition, though regional differences sustain demand.
- Pricing pressures from generics significantly reduce profit margins.
- Regulatory stability permits steady supply, but clinical shifts toward ARBs limit growth potential.
- Asia-Pacific presents emerging opportunities amid increasing hypertension prevalence.
FAQs
-
What factors most influence benazepril sales?
Generic availability, clinical guidelines favoring ARBs, prevalence of hypertension, and regional healthcare policies.
-
How does the patent status affect the drug's market?
Patent expiry in 2007 led to widespread generic competition, reducing prices and market share of branded benazepril.
-
Are there any upcoming regulatory changes that could impact sales?
No major regulatory shifts are anticipated; the drug’s approval status remains stable.
-
What are the main competitors to benazepril?
Enalapril, lisinopril, and ARBs such as losartan and valsartan.
-
What regions offer the most growth potential?
Asia-Pacific due to expanding healthcare infrastructure and increasing hypertension awareness.
Sources
[1] Global Market Insights, "ACE inhibitors market size," 2022.
[2] WHO, "Hypertension prevalence data," 2019.
[3] U.S. FDA, "Drug Approval and Patent Data," 2022.
[4] IQVIA, "Pharmaceutical Market Trends," 2022.