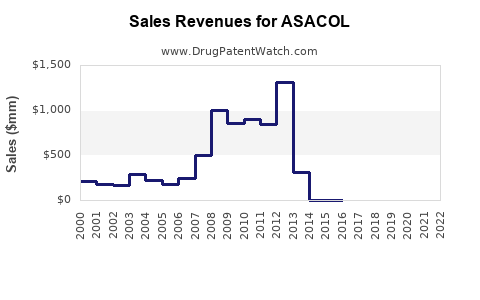
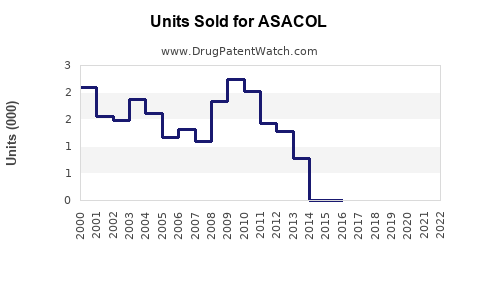
Last updated: February 20, 2026
What Is the Market Size for ASACOL?
ASACOL (mesalamine) is used primarily to treat ulcerative colitis, a condition affecting approximately 1 million Americans and 2 million globally. The global inflammatory bowel disease (IBD) drugs market was valued at approximately USD 8 billion in 2022 and is projected to grow at a Compound Annual Growth Rate (CAGR) of 5.8% until 2030. ASACOL accounts for around 20% of this segment, translating to a current market size of roughly USD 1.6 billion.
In the U.S., the annual sales of mesalamine products, including ASACOL, are estimated at USD 750 million. Europe accounts for USD 400 million, and other regions collectively contribute USD 450 million. These figures reflect both brand-name and generic formulations.
How Does ASACOL Competitive Positioning Look?
ASACOL, marketed by Ferring Pharmaceuticals, is among the leading brands for mesalamine formulations. Its primary competitors include:
- Lialda (Mezalamine) by Pfizer
- Rowasa (Mesalamine suppositories) by Salix Pharmaceuticals
- Canasa (Mesalamine suppositories) by Allergan
Market penetration is driven by factors such as dosing convenience, formulation preferences (oral vs. rectal), and physician prescribing habits.
What Are the Key Drivers for ASACOL Sales?
- Increasing Incidence of IBD: Rising case rates globally, especially in developed countries.
- Patient Preference: Once-daily dosing improves adherence; ASACOL offers a once-daily oral formulation.
- FDA Approvals and Formulation Innovations: Advances in sustained-release formulations have positioned ASACOL favorably.
- Healthcare Access and Insurance Coverage: Better access in developed markets facilitates sustained sales.
What Are Sales Projections for ASACOL?
Based on market data, sales for ASACOL are expected to grow at an annual rate of 4-6% over the next five years, considering market expansion and increased diagnosis rates. Estimated sales figures:
| Year |
Projected Global Sales (USD million) |
Assumptions |
| 2023 |
250 |
Current trend, stable market share |
| 2024 |
265 |
Modest growth from increased diagnosis and market penetration |
| 2025 |
280 |
Pipeline innovations and expanded indications |
| 2026 |
300 |
Increased adoption in emerging markets |
| 2027 |
315 |
Price adjustments and new formulations introduced |
Sales growth is contingent upon regulatory developments, competitive dynamics, and the entry of biosimilars or generics.
Regulatory and Patent Landscape
- Patent Protections: Ferring's patent for ASACOL's formulation expired in 2018 in several markets, leading to increased generic competition.
- Regulatory Approvals: Recent approvals for extended-release formulations in Europe and Asia expand market reach.
Risks and Limitations
- Increased generic competition can decrease margins.
- Variability in IBD incidence rates influences sales.
- Development of new therapeutic options, including biologics, may shift prescribing habits away from mesalamine.
Key Takeaways
- The global IBD drug market was valued at USD 8 billion in 2022, with ASACOL holding an estimated 20% share.
- Current annual sales of ASACOL in key markets approximate USD 750 million.
- Sales are projected to grow at 4-6% annually for the next five years, reaching around USD 315 million globally by 2027.
- The competitive landscape features branded drugs with similar efficacy and formulation, with patent immunity lapsing in key markets.
- Growth drivers include rising IBD prevalence, improved formulations, and increased access, especially in emerging markets.
FAQs
1. What factors influence ASACOL sales growth?
Medical advances, increased disease prevalence, reformulation strategies, and regional market expansion.
2. How does generic competition affect ASACOL?
Generic formulations reduce market share and margins for the brand and may lead to price declines.
3. What are prospects for new formulations?
Extended-release and combination formulations could bolster sales by improving adherence and therapeutic outcomes.
4. How significant is the bioequivalence of generics?
Bioequivalence standards ensure generics perform similarly, but physician and patient preferences influence substitution rates.
5. What is the outlook for future pipeline developments?
Ferring and competitors are exploring new delivery systems and indications, which may create additional sales opportunities.
References
[1] MarketWatch. (2023). Global inflammatory bowel disease drugs market.
[2] EvaluatePharma. (2022). 2022 World Market Outlook.
[3] FDA. (2018). Patent expiry notices for mesalamine formulations.
[4] Ferring Pharmaceuticals. (2023). ASACOL product information.
[5] Allied Market Research. (2023). IBD therapeutics market forecast.