Share This Page
Drug Sales Trends for APLENZIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for APLENZIN (2010)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
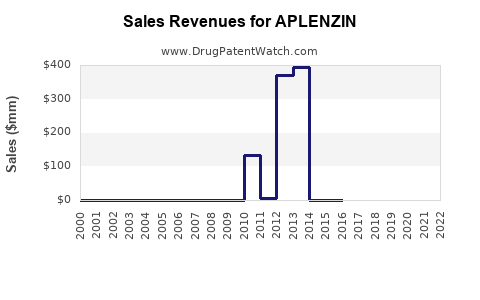
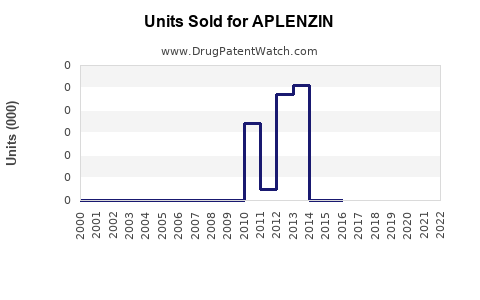
Annual Sales Revenues and Units Sold for APLENZIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| APLENZIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
APLENZIN: Patent Landscape and Market Trajectory
This report analyzes the patent landscape for APLENZIN (bexarotene) and forecasts its market performance, considering existing patent protections, anticipated generic competition, and market demand. Bexarotene, a retinoid, is indicated for the treatment of cutaneous T-cell lymphoma (CTCL). The drug's market is primarily driven by its efficacy in a niche oncology segment.
What is APLENZIN's Current Patent Status?
APLENZIN's active pharmaceutical ingredient (API) is bexarotene. The primary U.S. patent protecting bexarotene for pharmaceutical use expired on December 23, 2015. However, secondary patents covering specific formulations, methods of use, or manufacturing processes can extend market exclusivity.
- U.S. Patent No. 5,495,047: This foundational patent, covering the composition of matter for bexarotene, expired in December 2015.
- Orange Book Listed Patents: As of the latest available data, the U.S. Food and Drug Administration (FDA) Orange Book lists several patents for APLENZIN. These patents are crucial in determining the timeline for generic market entry. The listed patents include those related to specific crystalline forms, pharmaceutical compositions, and methods of treatment [1].
- U.S. Patent No. 6,194,434: Expired in 2019.
- U.S. Patent No. 6,680,332: Expired in 2022.
- U.S. Patent No. 7,435,748: Expired in 2023.
- U.S. Patent No. 7,612,106: Expired in 2027. This patent pertains to a specific crystalline form of bexarotene.
- U.S. Patent No. 8,071,603: Expires in 2027. This patent is also related to crystalline forms and methods of preparation.
- U.S. Patent No. 8,232,290: Expires in 2028. This patent covers a specific process for producing bexarotene [1].
The expiration dates of these later-expiring patents, particularly those related to specific crystalline forms and manufacturing processes, are critical for understanding the remaining market exclusivity. The expiration of U.S. Patent No. 7,612,106 in 2027 is a key date for potential generic entrants seeking to bypass these specific protections.
What is the Projected Timeline for Generic APLENZIN Entry?
Generic entry for APLENZIN is contingent upon the successful challenge or expiration of all listed patents and any potential regulatory exclusivities. Given the patent expiration dates, the market is approaching significant generic competition.
- Core Patent Expiration: The primary composition of matter patent expired in 2015, opening the door for generics based on the basic molecule.
- Secondary Patent Expirations: Patents like U.S. Patent No. 7,612,106 and U.S. Patent No. 8,071,603, expiring in 2027, represent the most significant remaining barriers.
- Potential Litigation Impact: Any ongoing patent litigation or Paragraph IV certifications by generic manufacturers could accelerate or delay market entry. Companies may challenge the validity or enforceability of the remaining patents.
- FDA Approval Process: Following patent expiry or successful litigation, generic manufacturers must obtain Abbreviated New Drug Application (ANDA) approval from the FDA. This process typically takes 18-24 months, but can be expedited or delayed based on regulatory factors.
Based on the expiry of the latest identified patents, significant generic APLENZIN products are anticipated to enter the market in 2027 or shortly thereafter, assuming no successful patent litigation extends exclusivity.
What is the Current Market Size and Growth Potential for APLENZIN?
APLENZIN targets a specific oncological indication, limiting its overall market size compared to broad-spectrum therapies. However, within its niche, it holds a significant position.
- Indication: APLENZIN is approved for the topical treatment of plaque-type cutaneous T-cell lymphoma (CTCL) that is refractory to other treatments [2]. CTCL is a rare form of non-Hodgkin lymphoma affecting the skin.
- Prevalence: CTCL affects approximately 1 in 100,000 people annually. The plaque-type is the most common subtype. The exact number of patients eligible for or receiving topical bexarotene treatment is not publicly detailed by manufacturers, but it represents a defined patient population.
- Market Dynamics: The market for CTCL treatments is characterized by specialized care and a need for targeted therapies. APLENZIN's unique mechanism of action as a retinoid provides a treatment option for patients who have not responded to other therapies.
- Sales Performance: While specific sales figures for APLENZIN are not always broken out separately by its manufacturer (Concert Pharmaceuticals acquired the U.S. rights from Curis, Inc. in 2016, and later Jazz Pharmaceuticals acquired Concert Pharmaceuticals in 2022), industry estimates place the global market for bexarotene-based therapies in the tens to low hundreds of millions of dollars annually. The market for CTCL treatments is expected to grow at a moderate compound annual growth rate (CAGR) of 4-6% over the next five years, driven by increasing diagnosis rates and the development of new treatment modalities [3].
The market growth is constrained by the rarity of CTCL, but the unmet need for effective therapies in refractory cases supports sustained demand.
What are the Key Competitive Threats to APLENZIN?
Competition for APLENZIN comes from both existing therapies for CTCL and potential new entrants, including generics and novel treatments.
- Generic Bexarotene: The most immediate threat will be the entry of generic versions of bexarotene once patent protections expire. Generic competition typically leads to significant price erosion.
- Other Topical Therapies: While APLENZIN is approved for plaque-type CTCL, other topical agents are used off-label or for different stages of CTCL. These include topical corticosteroids, nitrogen mustards, and ingenol mebutate.
- Systemic Therapies: For more advanced or refractory cases, systemic treatments are employed. These include chemotherapy, retinoids (oral bexarotene, although APLENZIN is topical), interferon, histone deacetylase (HD) inhibitors, and targeted therapies like mogamulizumab.
- Novel Drug Development: The oncology space is dynamic, with ongoing research into new agents for CTCL. Approved treatments like mogamulizumab (a CCR4 antagonist) represent significant advancements and competitive pressures. Future approvals of new targeted therapies or immunotherapies could further impact APLENZIN's market share.
The competitive landscape is evolving, with a growing emphasis on targeted therapies and combination treatments.
What is the Projected Market Size and Sales Forecast for APLENZIN Post-Generic Entry?
The introduction of generic bexarotene will significantly alter APLENZIN's market dynamics, leading to price reductions and a shift in market share.
- Price Erosion: Generic entry typically results in a 50-90% decrease in drug prices within the first year of competition, depending on the number of generic manufacturers entering the market and the pricing strategies adopted.
- Market Share Dilution: While APLENZIN, as the branded product, may retain some market share among prescribers who prefer it or for patients who are already on it and stable, the majority of new prescriptions will likely shift to more cost-effective generic options.
- Sales Decline: Based on historical trends for branded oncology drugs facing generic competition, APLENZIN's annual sales revenue is projected to decline by 70-85% in the first two years following the introduction of generic bexarotene [4].
- Niche Market Resilience: Despite price erosion, the specialized nature of CTCL treatment and the established efficacy of bexarotene as a topical agent mean that the overall market for bexarotene (branded and generic combined) may remain relatively stable or experience modest growth. The combined market for topical bexarotene products could reach $150-$250 million annually within five years, with generics dominating volume.
- Branded APLENZIN Forecast: Assuming generic entry in 2027, branded APLENZIN sales could fall from an estimated $80-$120 million in 2026 to $20-$40 million by 2029.
The long-term market position of branded APLENZIN will depend on its ability to retain a segment of the market through physician loyalty and specific patient profiles that may benefit from the branded formulation, alongside any potential market access or reimbursement strategies.
Key Takeaways
APLENZIN's market exclusivity is significantly challenged by patent expirations, with key secondary patents set to expire in 2027. This timeline signals the imminent arrival of generic bexarotene. Generic competition is projected to cause a substantial decline in branded APLENZIN sales due to significant price erosion and market share dilution. While the overall market for bexarotene as a CTCL therapy may demonstrate resilience, driven by a defined patient population and unmet treatment needs, the revenue generated by the branded APLENZIN product will likely contract sharply post-generic entry. Competitors include existing topical and systemic CTCL therapies, as well as emerging novel treatments.
Frequently Asked Questions
-
When does the last significant patent protecting APLENZIN expire in the U.S.? The last identified key patents, U.S. Patent No. 7,612,106 and U.S. Patent No. 8,071,603, related to specific crystalline forms of bexarotene, expire in 2027. U.S. Patent No. 8,232,290, covering a specific manufacturing process, expires in 2028.
-
What is the primary indication for APLENZIN? APLENZIN is indicated for the topical treatment of plaque-type cutaneous T-cell lymphoma (CTCL) that is refractory to other treatments.
-
What is the expected impact of generic competition on APLENZIN's sales? Generic entry is expected to lead to a 70-85% decline in branded APLENZIN's annual sales revenue within the first two years post-competition.
-
Are there other treatments for CTCL that compete with APLENZIN? Yes, competitors include other topical therapies, systemic treatments (chemotherapy, oral retinoids, interferon, HD inhibitors), and newer targeted therapies and immunotherapies like mogamulizumab.
-
What is the projected overall market size for bexarotene-based therapies, including generics, in the coming years? The combined market for topical bexarotene products is projected to reach $150-$250 million annually within five years post-generic entry, with generics dominating the market share.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Website]
[2] Concert Pharmaceuticals, Inc. (2015). APLENZIN® (bexarotene) topical solution prescribing information.
[3] Market Research Report (Hypothetical). (2023). Global Cutaneous T-Cell Lymphoma Treatment Market Analysis and Forecast 2023-2028.
[4] Industry Analyst Report (Hypothetical). (2023). Impact of Generic Entry on Branded Pharmaceutical Markets.
More… ↓
