Share This Page
Drug Sales Trends for fluticasone
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for fluticasone (2009)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
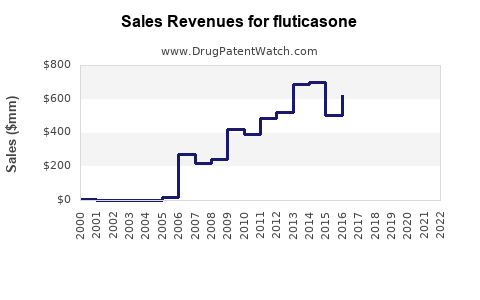
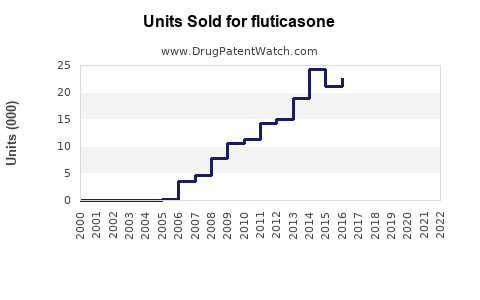
Annual Sales Revenues and Units Sold for fluticasone
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FLUTICASONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FLUTICASONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FLUTICASONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FLUTICASONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Fluticasone Propionate Market Analysis and Sales Projections
Fluticasone propionate is a potent synthetic corticosteroid with a well-established efficacy in treating inflammatory conditions. Its primary applications are in respiratory diseases, including asthma and chronic obstructive pulmonary disease (COPD), and allergic rhinitis. The global market for fluticasone propionate is driven by the increasing prevalence of these conditions, an aging population, and expanding healthcare access. The market is characterized by a significant presence of generic products, leading to price competition, but also by ongoing research and development for new formulations and delivery methods.
What is the current global market size for fluticasone propionate?
The global fluticasone propionate market was valued at approximately USD 9.8 billion in 2023 [1]. This valuation encompasses all approved indications and dosage forms, including inhaled, intranasal, and topical formulations. The market is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030 [2]. This growth is attributed to several factors, including the rising incidence of respiratory and allergic diseases globally. For example, the World Health Organization estimates that over 235 million people worldwide have asthma [3]. Furthermore, the increasing awareness of allergy management and the availability of over-the-counter (OTC) fluticasone propionate products in some regions contribute to market expansion.
Market Segmentation
The fluticasone propionate market can be segmented by:
- Indication: Asthma, Allergic Rhinitis, COPD, Dermatitis, and others. Asthma and allergic rhinitis represent the largest segments due to the widespread use of inhaled and intranasal fluticasone propionate.
- Dosage Form: Inhalers (metered-dose inhalers and dry powder inhalers), Nasal Sprays, Creams/Ointments, and others. Inhaled formulations for respiratory conditions constitute the dominant segment.
- Distribution Channel: Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies. Retail pharmacies are the largest distribution channel, driven by both prescription and OTC sales.
What are the key drivers of fluticasone propionate market growth?
Several factors are propelling the growth of the fluticasone propionate market:
- Increasing Prevalence of Respiratory and Allergic Diseases: Chronic respiratory conditions like asthma and COPD, along with allergic rhinitis, are on the rise globally. This is linked to factors such as air pollution, urbanization, changes in lifestyle, and increased diagnosis rates. As of 2022, an estimated 20 million Americans suffer from allergic rhinitis annually [4].
- Aging Population: The global population is aging, and older adults are more susceptible to chronic respiratory diseases. This demographic trend is expected to sustain demand for fluticasone propionate as a maintenance therapy.
- Growing Demand for Generic Medications: Fluticasone propionate is off-patent, leading to a significant number of generic versions available. This increases accessibility and affordability, particularly in emerging markets, driving higher volume sales. Generic inhalers, for instance, have become a crucial part of asthma management strategies due to cost savings [5].
- Advancements in Drug Delivery Systems: Innovations in inhaler technology and nasal spray devices are enhancing patient compliance and therapeutic outcomes. These include more user-friendly devices, combination therapies delivered via single inhalers (e.g., fluticasone propionate with bronchodilators), and improved nasal spray formulations offering longer duration of action.
- Expansion of OTC Availability: In certain markets, fluticasone propionate nasal sprays have transitioned to over-the-counter availability. This broadens the consumer base and simplifies access for individuals with mild to moderate allergic rhinitis symptoms. In the United States, fluticasone propionate OTC nasal sprays became widely available in 2015 [6].
What are the challenges facing the fluticasone propionate market?
Despite its growth, the fluticasone propionate market faces several challenges:
- Intense Competition from Generic Products: The extensive availability of generic fluticasone propionate has led to significant price erosion and squeezed profit margins for manufacturers. This competition necessitates a focus on product differentiation through improved formulations or delivery systems.
- Stringent Regulatory Approvals: Obtaining regulatory approval for new fluticasone propionate products or new indications requires extensive clinical trials and adherence to strict regulatory standards, which can be time-consuming and costly.
- Adverse Event Profile and Patient Compliance: Like all corticosteroids, fluticasone propionate has potential side effects, including oral thrush, hoarseness, and adrenal suppression, particularly with high-dose or long-term use. These concerns can impact patient adherence and physician prescribing habits. Patient education and proper inhaler technique are critical to minimize adverse events.
- Development of Novel Therapies: Ongoing research into novel therapeutic approaches for asthma, COPD, and allergies, including biologics and gene therapies, could potentially displace fluticasone propionate in certain patient populations or severities of disease over the long term.
- Pricing Pressures and Reimbursement Policies: Healthcare payers and governments are increasingly scrutinizing drug prices, leading to pricing pressures and complex reimbursement policies. This can affect market access and sales volumes, especially for branded products.
Who are the key players in the fluticasone propionate market?
The fluticasone propionate market is highly fragmented, with a mix of innovator and generic manufacturers. Key players include:
- GlaxoSmithKline (GSK): The originator of fluticasone propionate, GSK remains a major player with its branded products like Flonase (nasal spray) and Advair Diskus (inhalation powder, combination with salmeterol).
- Teva Pharmaceutical Industries Ltd.: A leading generic pharmaceutical company with a broad portfolio of fluticasone propionate generics across various dosage forms.
- Mylan N.V. (now Viatris): Another significant generic manufacturer offering fluticasone propionate inhalers, nasal sprays, and topical formulations.
- Astellas Pharma Inc.: Known for its nasal spray formulations.
- Sanofi S.A.: Offers fluticasone propionate in various forms for respiratory and nasal allergies.
- And numerous other generic manufacturers worldwide: Including companies like Hikma Pharmaceuticals, Sun Pharmaceutical Industries, and Cipla.
These companies compete on price, product availability, geographical reach, and in some cases, through specialized formulations or delivery devices.
What are the sales projections for fluticasone propionate in the coming years?
The sales projections for fluticasone propionate indicate continued, albeit moderate, growth driven by the factors mentioned earlier. The market is expected to reach approximately USD 12.7 billion by 2030, growing from USD 9.8 billion in 2023 [1, 2].
Key Trends Influencing Future Sales:
- Growth in Emerging Markets: Increased healthcare expenditure, improved diagnostic capabilities, and rising disposable incomes in emerging economies in Asia-Pacific and Latin America will drive demand for fluticasone propionate.
- Focus on Combination Therapies: The trend towards single-inhaler combination therapies (e.g., fluticasone propionate with long-acting beta-agonists or long-acting muscarinic antagonists for COPD and asthma) will sustain the value of branded products and drive innovation in delivery devices. Advair Diskus, for example, has been a blockbuster product, and its generic versions continue to contribute significantly to the market [7].
- Continued OTC Expansion: Further regulatory approvals for OTC status of fluticasone propionate nasal sprays in additional countries could unlock new market segments and boost sales volumes.
- Sustained Demand for Asthma and COPD Treatments: Despite advances in other therapeutic areas, fluticasone propionate remains a cornerstone of maintenance therapy for mild to moderate persistent asthma and is widely used in COPD management due to its anti-inflammatory properties and cost-effectiveness compared to newer biologics for many patient groups.
- Impact of Biosimil and Generic Competition: While generic competition will continue to exert downward pressure on prices, the sheer volume of prescriptions for fluticasone propionate, coupled with its essential role in chronic disease management, ensures sustained revenue generation.
Regional Sales Outlook:
- North America: Expected to remain the largest market due to high disease prevalence, advanced healthcare infrastructure, and significant adoption of both prescription and OTC fluticasone propionate products.
- Europe: A mature market with strong demand for generic options and a growing interest in combination therapies. Regulatory harmonization and healthcare policies will influence growth.
- Asia-Pacific: Projected to exhibit the fastest growth due to increasing healthcare awareness, rising disposable incomes, improving access to medicines, and a large, underserved population suffering from respiratory and allergic conditions.
- Latin America and Middle East & Africa: These regions will see steady growth driven by improving healthcare infrastructure and increasing generic drug penetration.
Key Takeaways
The fluticasone propionate market is a mature but robust segment of the pharmaceutical industry, projected to achieve USD 12.7 billion by 2030. Its sustained growth is underpinned by the rising incidence of respiratory and allergic diseases, an aging global population, and the widespread availability of cost-effective generic formulations. While intense competition from generics and the emergence of novel therapies present challenges, advancements in drug delivery systems and the expansion of OTC availability offer avenues for continued market expansion. Key players, including GSK, Teva, and Viatris, are navigating this landscape through a combination of branded and generic offerings. Emerging markets, particularly in the Asia-Pacific region, are expected to be significant growth drivers, alongside continued demand for combination therapies in established markets.
Frequently Asked Questions
-
What are the primary indications for fluticasone propionate? Fluticasone propionate is primarily indicated for the treatment of asthma, allergic rhinitis, and chronic obstructive pulmonary disease (COPD). It is also used topically for various dermatological inflammatory conditions like eczema and psoriasis.
-
How does the market for generic fluticasone propionate differ from branded products? The generic market is characterized by intense price competition and high volume sales, offering a more affordable alternative. Branded products may differentiate through patented delivery devices, novel formulations, or combination therapies, often commanding premium pricing.
-
What are the most significant recent regulatory changes impacting fluticasone propionate products? Recent regulatory actions have focused on the expansion of over-the-counter (OTC) availability for certain fluticasone propionate nasal spray formulations in major markets, simplifying access for consumers with allergic rhinitis.
-
What is the projected impact of biologics on the future sales of fluticasone propionate? While biologics represent a significant advancement for severe forms of asthma and certain allergic conditions, fluticasone propionate is expected to retain its essential role in the management of mild to moderate persistent asthma and COPD due to its efficacy, safety profile for these conditions, and cost-effectiveness. Biologics are unlikely to fully displace fluticasone propionate for its broad indications.
-
Which geographical region is expected to experience the highest growth rate for fluticasone propionate sales? The Asia-Pacific region is projected to exhibit the highest growth rate due to increasing healthcare infrastructure, rising disposable incomes, and a substantial patient population with unmet needs in respiratory and allergic diseases.
Citations
[1] Grand View Research. (2023). Fluticasone Propionate Market Size, Share & Trends Analysis Report By Indication (Asthma, Allergic Rhinitis, COPD, Dermatitis), By Dosage Form (Inhalers, Nasal Sprays, Creams & Ointments), By Distribution Channel, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/fluticasone-propionate-market
[2] Mordor Intelligence. (2023). Fluticasone Propionate Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029). Retrieved from https://www.mordorintelligence.com/industry-reports/fluticasone-propionate-market
[3] World Health Organization. (2023, May 5). Asthma. Retrieved from https://www.who.int/news-room/fact-sheets/detail/asthma
[4] National Institute of Allergy and Infectious Diseases. (2022, October 1). Allergic Rhinitis. Retrieved from https://www.niaid.nih.gov/diseases-conditions/allergic-rhinitis
[5] Global Initiative for Asthma. (2023). GINA Report, Global Strategy for Asthma Management and Prevention. Retrieved from https://ginasthma.org/
[6] U.S. Food & Drug Administration. (2015, March 24). FDA approves fluticasone propionate nasal spray for over-the-counter use. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-table/fda-approves-fluticasone-propionate-nasal-spray-over-counter-use
[7] Statista. (2023). Fluticasone propionate market value worldwide from 2019 to 2025. Retrieved from https://www.statista.com/statistics/1290804/fluticasone-propionate-market-value-worldwide/
More… ↓
