Share This Page
Drug Sales Trends for TRIAMCINOLON
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TRIAMCINOLON (2009)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
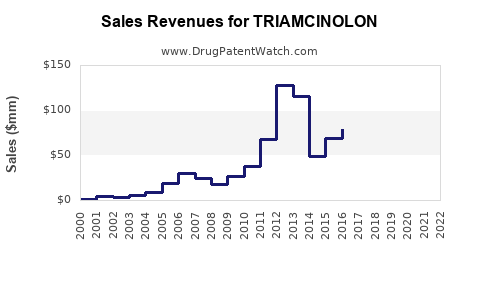
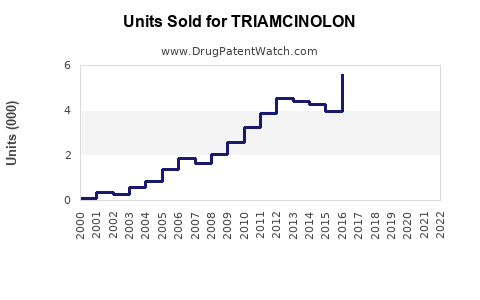
Annual Sales Revenues and Units Sold for TRIAMCINOLON
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRIAMCINOLON | ⤷ Get Started Free | ⤷ Get Started Free | 2022 |
| TRIAMCINOLON | ⤷ Get Started Free | ⤷ Get Started Free | 2021 |
| TRIAMCINOLON | ⤷ Get Started Free | ⤷ Get Started Free | 2020 |
| TRIAMCINOLON | ⤷ Get Started Free | ⤷ Get Started Free | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TRIAMCINOLONE: PATENT LANDSCAPE AND MARKET PROJECTIONS
Triamcinolone, a synthetic corticosteroid, maintains a robust market position driven by its broad therapeutic applications and ongoing patent strategies. While primary patents for the active pharmaceutical ingredient have expired, key formulations, delivery systems, and combination therapies continue to be protected, influencing market entry for generics and the development of new entrants. Sales are projected to grow steadily, supported by increasing adoption in dermatology, ophthalmology, and respiratory medicine, alongside market penetration of biosimilar and authorized generic versions.
WHAT ARE THE KEY PATENT EXPIRATIONS AND RECENT FILINGS FOR TRIAMCINOLONE?
The foundational patents covering the chemical entity triamcinolone have long expired. However, innovation continues through proprietary delivery mechanisms and novel therapeutic uses.
- U.S. Patent No. 2,717,853 (Exp. 1971) covers the basic chemical composition of triamcinolone.
- U.S. Patent No. 3,089,821 (Exp. 1981) relates to triamcinolone acetonide, a widely used ester form.
Recent patent activity focuses on:
- Formulations: Patents covering specific compositions designed for enhanced stability, targeted delivery, or improved patient compliance. For example, patents related to extended-release formulations for intra-articular injections.
- Delivery Devices: Innovations in inhalers, nasal sprays, and ophthalmic applicators that improve drug efficacy and reduce systemic exposure.
- Therapeutic Indications: New patent applications often target novel uses of triamcinolone for specific medical conditions or patient populations.
- Combination Therapies: Patents protecting fixed-dose combinations of triamcinolone with other active pharmaceutical ingredients.
HOW DO EXISTING PATENTS IMPACT THE GENERIC MARKET FOR TRIAMCINOLONE?
The expiration of primary composition-of-matter patents opens the door for generic competition. However, a complex patent landscape can delay or limit generic market entry through:
- Method of Use Patents: These patents protect specific therapeutic applications of triamcinolone. Generic manufacturers must navigate these by ensuring their product is not marketed for an indication covered by an unexpired method of use patent, or by seeking to invalidate these patents.
- Formulation Patents: Proprietary formulations, such as extended-release versions or specific topical preparations, are often protected by separate patents. Generic versions may offer immediate-release or bioequivalent formulations, but they cannot directly replicate patented advanced formulations without licensing or challenging the patents.
- Process Patents: Patents covering specific manufacturing processes for triamcinolone or its derivatives can create barriers if those processes are essential for cost-effective production.
- Patent Linkage: The U.S. Food and Drug Administration (FDA) Orange Book lists patents associated with approved drug products. Generic companies must certify that their product does not infringe listed patents, or challenge their validity, before receiving approval.
Table 1: Key Triamcinolone Formulations and Associated Patent Considerations
| Formulation Type | Common Brand Names | Primary Patent Expiration (Composition) | Typical Patent Protection Focus (Post-Composition Expiry) | Market Impact for Generics |
|---|---|---|---|---|
| Oral Tablets | Kenacort, Aristocort | Expired | Manufacturing processes, specific salt forms. | High generic penetration, price sensitive. |
| Topical Creams/Ointments | Kenalog, Aristocort | Expired | Specific excipients, stable formulations, enhanced skin penetration methods. | Moderate generic penetration, brand loyalty for specific formulations. |
| Injectable Suspensions (Intra-articular) | Kenalog, Trilipix | Expired | Extended-release particle technology, depot formulations, novel suspension stabilizers. | Limited generic entry for advanced formulations; bioequivalent immediate-release generics available. |
| Ophthalmic Suspensions | Triesence, Trivaris | Expired | Sterility, preservation systems, particle size control for ocular safety and efficacy. | Barriers to entry due to stringent manufacturing and regulatory requirements for ophthalmic products. |
| Nasal Sprays | Nasacort Allergy 24HR | Expired | Metered-dose delivery systems, propellant-free formulations, specific preservative systems. | Authorized generics and direct generics for older formulations; patent challenges for advanced delivery systems. |
WHAT ARE THE CURRENT THERAPEUTIC AREAS FOR TRIAMCINOLONE AND THEIR MARKET SIZE?
Triamcinolone's versatility supports its use across multiple therapeutic segments, each with distinct market dynamics.
Dermatology
This is a primary market for triamcinolone, used in creams, ointments, and lotions to treat inflammatory skin conditions.
- Indications: Eczema, psoriasis, dermatitis, allergies, insect bites.
- Market Size: The global dermatology drug market is substantial. While specific figures for triamcinolone's contribution are proprietary, the corticosteroid segment is a significant portion. The U.S. topical corticosteroid market alone is valued in the billions.
- Drivers: Increasing prevalence of skin disorders, demand for effective anti-inflammatory treatments, and over-the-counter availability of lower potency formulations.
Ophthalmology
Triamcinolone acetonide injectable suspensions are critical for treating posterior segment eye diseases.
- Indications: Uveitis, cystoid macular edema (CME) associated with retinal vein occlusion or post-cataract surgery, diabetic macular edema (DME).
- Market Size: The ophthalmic drug market, particularly for conditions like wet age-related macular degeneration and diabetic retinopathy, is a high-value segment. Injectable triamcinolone products for these indications represent hundreds of millions of dollars in annual sales.
- Drivers: Aging populations, rising incidence of diabetes and its ocular complications, and advancements in intravitreal drug delivery.
Respiratory Medicine
Triamcinolone acetonide nasal sprays are widely prescribed for allergic rhinitis, and inhaled formulations are used for asthma.
- Indications: Allergic rhinitis, asthma.
- Market Size: The global market for allergy and asthma medications is in the tens of billions of dollars. Triamcinolone-containing nasal sprays hold a significant share within the corticosteroid nasal spray sub-segment.
- Drivers: High prevalence of allergic rhinitis and asthma, awareness of the benefits of intranasal corticosteroids for symptom control, and the development of convenient delivery devices.
Other Applications
- Intra-articular Injections: Used for joint inflammation in conditions like rheumatoid arthritis and osteoarthritis.
- Oral Formulations: Used for systemic inflammatory conditions, though less common due to systemic side effects.
- Market Size: These segments contribute to the overall sales, though often smaller than dermatology, ophthalmology, or respiratory.
Table 2: Estimated Market Segment Contributions for Triamcinolone (Global, Annual)
| Therapeutic Area | Estimated Market Value (USD Billion) | Key Product Types | Primary Growth Drivers |
|---|---|---|---|
| Dermatology | $2.0 - $3.5 | Topical creams, ointments, lotions | Increasing skin disorder prevalence, OTC availability, improved formulations. |
| Ophthalmology | $0.7 - $1.2 | Injectable suspensions (intraocular) | Aging population, diabetes prevalence, advancements in intravitreal drug delivery. |
| Respiratory | $0.5 - $0.9 | Nasal sprays, inhaled aerosols | High incidence of allergies and asthma, patient preference for convenient delivery. |
| Other (Arthritis) | $0.2 - $0.4 | Injectable suspensions (intra-articular) | Osteoarthritis and rheumatoid arthritis prevalence, pain management needs. |
| Total Estimated | $3.4 - $6.0 |
Note: These are estimations based on market segment sizes and triamcinolone's market share within those segments. Exact figures are proprietary.
WHAT ARE THE SALES PROJECTIONS FOR TRIAMCINOLONE?
The market for triamcinolone is expected to exhibit steady growth, driven by several factors:
- Established Efficacy: Triamcinolone remains a well-established and effective treatment for a wide range of inflammatory conditions.
- Generic Competition: The availability of affordable generic versions in oral and topical forms fuels volume growth, particularly in cost-sensitive markets.
- Ophthalmology and Respiratory Growth: The sustained and increasing demand in ophthalmology (driven by diabetic retinopathy and uveitis) and respiratory medicine (driven by allergic rhinitis and asthma) will underpin significant revenue streams.
- New Formulations and Delivery Systems: Ongoing R&D into improved delivery methods, such as longer-acting injectables or more convenient inhalers, can create new revenue opportunities and extend product lifecycles.
- Geographic Expansion: Increased access to healthcare and the availability of triamcinolone in emerging markets will contribute to global sales growth.
- Biosimilar and Authorized Generic Approvals: The introduction of biosimil or authorized generic versions of specific branded products, especially in ophthalmology, can expand market access and drive volume, albeit with potential price erosion compared to originator products.
Projected Sales Growth:
- Short-Term (2024-2026): Anticipate a compound annual growth rate (CAGR) of 3-5%, driven by continued demand in established indications and the rollout of new generic and authorized generic products.
- Medium-Term (2027-2030): Growth is projected to stabilize at 2-4% CAGR. The market will be influenced by the success of novel delivery systems and potential new indications, balanced by ongoing generic price pressures.
Factors Potentially Limiting Growth:
- Adverse Event Profile: As a corticosteroid, triamcinolone has known side effects, which can lead to preference for alternative treatments in certain patient populations or for chronic, long-term use.
- Competition from Newer Agents: The development of non-steroidal anti-inflammatory agents, biologics, and targeted therapies in areas like ophthalmology and dermatology may divert market share from corticosteroids over the long term.
- Pricing Pressures: Intense competition among generic manufacturers can lead to significant price reductions, impacting overall market value even with increased volume.
HOW ARE COMPETITORS POSITIONING THEMSELVES IN THE TRIAMCINOLONE MARKET?
Competitors are strategizing across several fronts:
- Generic Market Dominance: Many pharmaceutical companies focus on manufacturing and marketing cost-effective generic versions of triamcinolone oral, topical, and injectable forms. This involves efficient manufacturing processes and broad distribution networks.
- Specialty Formulations: Some companies invest in developing and patenting proprietary formulations that offer advantages such as extended release, improved bioavailability, or reduced side effects. Examples include novel intra-articular injection formulations or advanced topical delivery systems.
- Device Innovation: Companies with expertise in drug delivery devices focus on improving inhalers, nasal spray devices, or ophthalmic applicators to enhance patient compliance and therapeutic outcomes.
- Authorized Generics (AGs): Major brand manufacturers often launch their own authorized generic versions of their branded products after key patents expire. This strategy aims to capture generic market share and prevent pure generic competitors from gaining a strong foothold.
- Combination Products: Developing and patenting combination therapies that include triamcinolone alongside other active ingredients can create differentiated products and secure new market exclusivity.
- Geographic Market Penetration: Competitors actively seek to expand their triamcinolone product reach into emerging markets through partnerships and local manufacturing strategies.
- Regulatory Pathways: Companies meticulously navigate the patent landscape, seeking Paragraph IV certifications for ANDA filings to challenge existing patents and expedite generic entry, or defensively seeking new patents for incremental innovations.
Example Competitor Strategies:
- Company A: Focuses on high-volume production of generic topical triamcinolone, leveraging economies of scale and broad pharmacy distribution.
- Company B: Invests heavily in R&D for novel ophthalmic triamcinolone formulations with extended-release properties, aiming for a premium market position.
- Company C: Strategizes the launch of authorized generics for leading branded triamcinolone products to maintain market presence and revenue post-patent expiry.
WHAT ARE THE FUTURE TRENDS AND POTENTIAL DISRUPTORS FOR TRIAMCINOLONE?
The future of triamcinolone will be shaped by evolving treatment paradigms, technological advancements, and regulatory landscapes.
Future Trends
- Personalized Medicine: While triamcinolone is a broad-spectrum agent, future applications might see more targeted use based on patient genetic profiles or specific disease biomarkers, optimizing efficacy and minimizing side effects.
- Advanced Drug Delivery Systems: Continued innovation in nanotechnology, sustained-release matrices, and targeted delivery mechanisms will aim to improve triamcinolone's therapeutic index, reducing systemic exposure and enhancing local efficacy. This includes exploring novel depot formulations for longer-acting intra-articular and intravitreal injections.
- Combination Therapies: The development of fixed-dose combinations with novel agents or other corticosteroids will likely continue, offering synergistic effects and simplified treatment regimens.
- Biologics and Non-Steroidal Alternatives: The increasing success and adoption of biologic drugs and novel non-steroidal agents in treating inflammatory diseases (e.g., for psoriasis, asthma, or uveitis) represent a potential long-term disruptor, as these may offer improved safety profiles or efficacy for specific patient subgroups.
- Digital Health Integration: Wearable devices and AI-driven platforms could eventually play a role in monitoring treatment response and adherence for patients using triamcinolone, particularly for chronic conditions.
Potential Disruptors
- Breakthrough Non-Steroidal Therapies: The development of highly effective and safe non-steroidal drugs that demonstrate superior efficacy or safety profiles across triamcinolone's key indications could significantly erode its market share.
- Advances in Gene Therapy and Cell Therapy: For certain inflammatory or autoimmune conditions, the emergence of curative or disease-modifying gene or cell therapies could fundamentally alter treatment approaches, reducing the reliance on symptomatic treatments like corticosteroids.
- Stringent Regulatory Scrutiny on Corticosteroid Side Effects: Increased awareness and potential regulatory actions concerning the long-term side effects of corticosteroids could lead to more restricted prescribing practices or a shift towards alternatives, particularly for less severe conditions.
- Emergence of Generics for Patented Advanced Formulations: Successful patent challenges or the development of bioequivalent but non-infringing advanced formulations by generic manufacturers could accelerate price erosion and market penetration for currently protected niche products.
KEY TAKEAWAYS
- Triamcinolone's primary composition patents have expired, but its market is sustained by protected formulations, delivery systems, and therapeutic uses.
- The drug serves major markets in dermatology, ophthalmology, and respiratory medicine, with significant global sales.
- Sales are projected for steady growth, driven by generic availability, established efficacy, and expansion in ophthalmology and respiratory segments.
- Competitors employ strategies ranging from generic market dominance to specialty formulation development and authorized generic launches.
- Future trends include advanced drug delivery and combination therapies, while potential disruptors include novel non-steroidal agents and regenerative medicine.
FREQUENTLY ASKED QUESTIONS
-
What is the current patent exclusivity status for triamcinolone acetonide injectable suspension used in ophthalmology? Patents covering the basic triamcinolone acetonide molecule have expired. However, specific formulations, sterile manufacturing processes, and delivery devices associated with branded ophthalmic suspensions may still be protected by unexpired patents, influencing generic market entry.
-
Are there any off-patent triamcinolone formulations that are experiencing significant generic competition? Yes, oral tablet and basic topical cream/ointment formulations of triamcinolone are subject to extensive generic competition due to the long-standing expiration of their core composition patents.
-
What is the primary driver for triamcinolone's continued market relevance in the face of newer therapeutic options? Triamcinolone's broad spectrum of anti-inflammatory activity, well-understood efficacy, favorable safety profile for short-term use, and cost-effectiveness of generic versions drive its continued relevance across multiple therapeutic areas.
-
How do method-of-use patents specifically impact generic triamcinolone nasal spray development? Method-of-use patents for triamcinolone nasal sprays could claim specific treatment durations, dosages, or therapeutic targets for allergic rhinitis. Generic manufacturers must ensure their product is not marketed for an indication covered by such unexpired patents or seek to invalidate them.
-
What is the projected CAGR for the global triamcinolone market over the next five years? The global triamcinolone market is projected to grow at a compound annual growth rate (CAGR) of approximately 3-5% from 2024 to 2026, moderating to 2-4% from 2027 to 2030.
CITATIONS
[1] U.S. Patent No. 2,717,853. (1955). Corticosteroids. [2] U.S. Patent No. 3,089,821. (1963). Esters of 9-alpha-fluoro-16-alpha-hydroxyprednisolone.
More… ↓
