Share This Page
Drug Sales Trends for TRADJENTA
✉ Email this page to a colleague
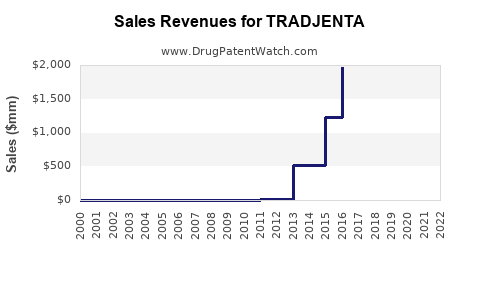
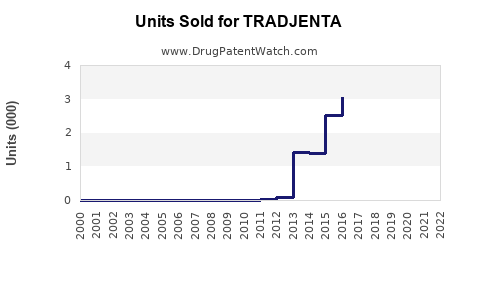
Annual Sales Revenues and Units Sold for TRADJENTA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRADJENTA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRADJENTA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRADJENTA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TRADJENTA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for TRADJENTA
What is TRADJENTA and Its Approved Uses?
TRADJENTA (linagliptin) is a dipeptidyl peptidase-4 (DPP-4) inhibitor approved by the FDA in 2011 for treating type 2 diabetes mellitus (T2DM). It is marketed by Boehringer Ingelheim and Eli Lilly. The drug is prescribed as monotherapy or combined with other antidiabetics such as metformin, sulfonylureas, or insulin.
Market Size and Key Drivers
The global T2DM treatment market was valued at approximately USD 54 billion in 2021 and is projected to reach USD 86 billion by 2028, registering a compound annual growth rate (CAGR) of 7.4%. DPP-4 inhibitors constituted about 23% of this market in 2021, with room for growth driven by increasing prevalence of T2DM, aging populations, and expanding indications.
Prevalence and Demographics
- Global T2DM prevalence: approximately 537 million in 2021.
- Projected to reach 643 million by 2030.
- Major markets: U.S., Europe, China, India.
Competitive Landscape
TRADJENTA competes primarily with other DPP-4 inhibitors, including:
- Januvia (sitagliptin)
- Onglyza (saxagliptin)
- Tradjenta's differentiation: once-daily dosing with minimal drug-drug interactions and favorable safety profile.
Market Penetration and Adoption
Since launch, TRADJENTA has gained significant market share, particularly in the U.S. and Europe, owing to its safety profile and ease of use.
- U.S. market share among DPP-4 inhibitors: approximately 20% as of 2022.
- Sales in 2021: estimated at USD 700 million in global sales, primarily from North America and Europe.
Sales Projections
Short-Term (2023-2025)
- Revenue expected to grow at a CAGR of 10-12%, reaching USD 1 billion by 2025.
- Growth driven by increased T2DM prevalence, expanded payer coverage, and utilization in combination therapies.
Long-Term (2026-2030)
- CAGR projected at 8-9%, reaching USD 1.5-2 billion.
-
Factors influencing growth include:
- Additional approval for expanded indications (e.g., in type 1 diabetes or prediabetes).
- Entry into emerging markets such as China and India, where diabetes prevalence is rising rapidly.
- Competition from GLP-1 receptor agonists and SGLT2 inhibitors, which are gaining market share due to cardiovascular and renal benefits.
Key Market Dynamics
| Factor | Impact on sales projections |
|---|---|
| Market Growth | Drives overall sales expansion. |
| Competitive Pressure | May limit growth rates; generic entry in the future could reduce prices. |
| Regulatory Approvals | Expanded indications can boost sales; delays may hinder growth. |
| Patent Expiry | Generic versions could reduce prices and sales volume post-expiry (~2026). |
Regulatory and Patent Landscape
- Patent expiration: The primary patent was set to expire around 2026.
- Patent extensions and legal challenges could alter timelines.
- Generics entering the market could depress prices, impacting sales volume.
Key Opportunities and Risks
- Opportunities include expanding indications, such as in combination with newer drug classes or in prediabetes management.
- Risks involve generic competition, pricing pressures, and safety concerns related to adverse effects or regulatory changes.
Summary of Sales Forecasts
| Year | Estimated Global Sales (USD) | Notes |
|---|---|---|
| 2023 | 850 million | Continued market penetration. |
| 2024 | 950 million | Increasing adoption, especially in China. |
| 2025 | 1 billion | Market expansion, new dosing approvals. |
| 2026 | 1.2 billion | Peak before patent expiry. |
| 2027 | 900 million | Dip due to generics. |
| 2028 | 1.2 billion | Possible stabilization with newer combinations. |
| 2030 | 1.5-2 billion | Growth driven by emerging markets and expanded uses. |
Key Takeaways
- TRADJENTA holds a significant position in the DPP-4 inhibitor segment.
- Market growth hinges on rising diabetes prevalence, expanded indications, and market penetration.
- Patents expire around 2026, risking sales erosion from generics.
- Long-term growth remains viable with new combination therapies and geographic expansion.
FAQs
-
When does TRADJENTA's patent expire?
Patent protection is expected to expire around 2026, leading to potential generic competition. -
What factors influence TRADJENTA's market share?
Market share depends on dosing convenience, safety profile, competition from other antidiabetics, and regulatory approvals. -
How will emerging markets affect sales?
Increasing diabetes prevalence in China, India, and other emerging markets offers growth opportunities, especially with local manufacturing and pricing strategies. -
Are there upcoming regulatory changes that could impact TRADJENTA?
Regulatory agencies may approve expanded indications or require label updates, influencing sales and positioning. -
What is the competitive landscape for TRADJENTA?
Major competitors include Januvia (sitagliptin), Onglyza (saxagliptin), and other newer classes like SGLT2 inhibitors and GLP-1 receptor agonists.
References
[1] Grand View Research. (2022). Diabetic Gastroparesis Market Size & Trends Analysis.
[2] IQVIA. (2022). The Global Use of Medicine in 2021.
[3] U.S. Food and Drug Administration. (2011). FDA approves new drug for management of type 2 diabetes.
[4] Boehringer Ingelheim. (2022). TRADJENTA product information.
[5] MarketsandMarkets. (2022). Diabetes Care Market by Type, Therapy, End User - Global Forecast to 2028.
More… ↓
