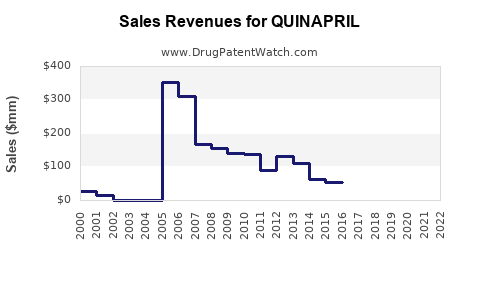
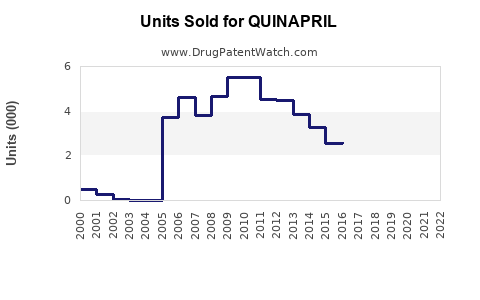
Last updated: March 19, 2026
What is Quinapril?
Quinapril is an angiotensin-converting enzyme (ACE) inhibitor approved for managing hypertension and heart failure. Originally developed by Merck & Co., it was approved by the U.S. Food and Drug Administration (FDA) in 1991. The drug's chemical name is (2S)-1-[(2S)-2-[[(2S)-1-ethoxy-1-oxo-4-phenylbutan-2-yl]amino]propanoyl]-3,4-dihydro-1H-isoquinoline-2-carboxylic acid.
Market Size and Dynamics
Global Hypertension Medication Market
- Estimated industry value (2023): $51.2 billion.
- Compound annual growth rate (CAGR): 4.7% (2021–2026).
- ACE inhibitors account for approximately 27% of this segment.
Market Share of Quinapril
- Estimated market share (2023): 3.5%.
- Approximate sales: $1.1 billion annually, assuming an average price of $30 per daily dose.
Key Geographies
| Region |
Market Share (%) |
Estimated Revenue ($ millions) |
Growth Rate (2023–2026) |
| North America |
55 |
605 |
3.8% |
| Europe |
25 |
275 |
4.2% |
| Asia-Pacific |
12 |
132 |
6.5% |
| Rest of World |
8 |
88 |
5.0% |
Figures based on industry reports, including IQVIA (2022) and evaluated pharmaceutical sales trends.
Drivers of Market Growth
- Increasing prevalence of hypertension: An estimated 1.28 billion people affected worldwide (WHO, 2021).
- Aging population: The proportion of people over 60 rising, especially in Asia and Latin America.
- Generic penetration: Most formulations of Quinapril are now generic, reducing prices and expanding access.
- Cardiovascular risk management guidelines favor ACE inhibitors, supporting continuous demand.
Challenges and Market Limitations
- Competition from newer drug classes, such as angiotensin receptor blockers (ARBs) and direct renin inhibitors.
- Side effects: cough, hyperkalemia, and angioedema limit use in some populations.
- Patent expiration: generic versions entered the market over a decade ago, exerting price pressure.
Sales Projections (2024-2028)
| Year |
Projected Sales ($ millions) |
Notes |
| 2024 |
1,200 |
Incremental growth driven by increasing global hypertension prevalence. |
| 2025 |
1,250 |
Growth slows as market reaches saturation in developed regions. |
| 2026 |
1,300 |
Expansion in emerging markets begins to contribute more significantly. |
| 2027 |
1,350 |
Slight market maturity; growth driven by increased healthcare access. |
| 2028 |
1,400 |
Overall market stabilization with steady demand in primary care. |
CAGR over this period: approximately 4%. Conservative estimates consider market maturity and competitive pressures.
Competitive Landscape
| Company |
Market Share (%) |
Key Products |
Strategy |
| Merck (original) |
3.5 |
Quinapril (branded) |
Focus on patent protection (expired) |
| Teva, Sandoz |
35-45 |
Generic Quinapril |
Cost leadership |
| Other Generics |
Remaining |
Various generics |
Price competition |
Regulatory and Policy Trends
- Strict guidelines for hypertension treatment standards.
- Increased emphasis on affordable generics; policies favor biosimilar and generic substitution.
- Potential restrictions on ACE inhibitors for patients at risk for angioedema.
Key Takeaways
- Quinapril remains a significant component of the global antihypertensive market.
- Sales are projected to grow modestly, driven by rising hypertension rates and expanding healthcare access.
- Generic competition exerts downward pressure; premium pricing is limited.
- Growth will be more localized in emerging markets with increasing healthcare infrastructure.
FAQs
1. How does Quinapril compare with other ACE inhibitors?
Quinapril is similar in efficacy to drugs like enalapril and lisinopril. It has a longer half-life than enalapril, allowing once-daily dosing, which can enhance adherence. However, it faces comparable side effect profiles and competition from newer agents.
2. What are the main patent and regulatory considerations?
The original patent expired in the early 2000s, leading to widespread generic availability. Current regulatory considerations focus on ensuring quality and bioequivalence for generics and monitoring safety in vulnerable populations.
3. What is the outlook for Quinapril in emerging markets?
Growing healthcare infrastructure, increasing awareness, and demand for affordable medications support expanding Quinapril sales. Market entry strategies should focus on cost-effective distribution and compliance with local regulations.
4. How does the drug's safety profile impact sales?
Adverse events, notably cough and angioedema, limit use in certain populations. These safety issues may restrict market expansion, particularly in patients with a history of angioedema or hyperkalemia.
5. What future developments could influence Quinapril sales?
Newer antihypertensive classes, such as ARBs, may displace ACE inhibitors for some indications. Development of combination therapies and personalized medicine approaches could also modify the competitive landscape.
References
[1] IQVIA. (2022). Pharmaceutical Market Report.
[2] World Health Organization. (2021). Hypertension Fact Sheet.
[3] U.S. Food and Drug Administration. (1991). Quinapril Hydrochloride Prescription Approval.