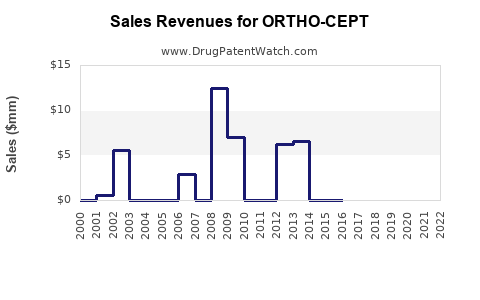
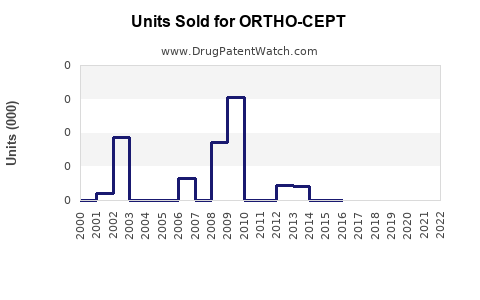
Last updated: February 20, 2026
What is ORTHO-CEPT?
ORTHO-CEPT is a combination oral contraceptive comprising ethynodiol diacetate and mestranol. It was approved by the FDA in 1972. It is prescribed primarily for birth control and hormonal regulation. Its generic versions are available in multiple markets at lower price points, influencing its potential market penetration.
Market Environment
Market Size and Revenue
The global oral contraceptive market was valued at approximately USD 6.3 billion in 2021. It is projected to reach USD 7.9 billion by 2026, with a compound annual growth rate (CAGR) of 4.7%. The segment including combination pills like ORTHO-CEPT is dominant, accounting for about 65% of the market.
Key Market Drivers
- Increasing awareness of family planning.
- Growing female workforce participation.
- Expanding access to reproductive health services in emerging economies.
- Rising sales of generic contraceptives due to price sensitivity.
Market Challenges
- Concerns over hormone-related side effects.
- Stringent regulatory environment.
- Competition from long-acting reversible contraceptives (LARCs), such as IUDs and implants.
- Cultural and religious opposition in certain regions.
Competitive Landscape
Major competitors include brands like Yaz, Loestrin, and Ortho Tri-Cyclen. Market share distribution favors oral combination pills, but LARCs are gaining ground due to efficacy and convenience.
Patent and Regulatory Status
ORTHO-CEPT has been off-patent since the early 1980s. No recent regulatory exclusivity extends its commercial potential. Its sales are primarily driven through generic manufacturers and branded generics.
Sales Projections
Assumptions
- The product maintains a stable market share within the oral contraceptive segment.
- Market growth proceeds at a CAGR of 4.7%.
- The drug’s market share remains steady at 2.5-3.0% of the global contraceptive market, considering generic competition.
- Potential market penetration in emerging markets increases gradually over five years.
Short-term Forecast (Next 2 Years)
| Year |
Estimated Global Market (USD) |
ORTHO-CEPT Market Share |
Projected Sales (USD) |
| 2023 |
6.8 billion |
2.5% |
170 million |
| 2024 |
7.1 billion |
2.7% |
192 million |
These estimates reflect incremental market share gains due to brand recognition and availability, particularly in markets where generics are in demand.
Mid-term Outlook (Next 5 Years)
| Year |
Estimated Global Market (USD) |
ORTHO-CEPT Market Share |
Projected Sales (USD) |
| 2025 |
7.4 billion |
3.0% |
222 million |
| 2026 |
7.9 billion |
3.2% |
253 million |
Growth driven by increased access in Asia, Africa, and Latin America, with a conservative acceleration of market share due to penetration in these regions.
Considerations
- Potential decline in sales due to the rising preference for LARCs.
- Impact of emerging contraceptive technologies or formulations.
- Regulatory changes affecting approval and marketing.
Key Factors Affecting Sales
- Generic availability reduces pricing and margins.
- Increasing competition from new oral and non-oral contraceptives.
- Shifting consumer preferences toward longer-acting methods.
- Societal and governmental policies on reproductive health.
Strategic Recommendations
- Focus on markets with low contraceptive adoption and high growth potential.
- Emphasize price competitiveness and distribution channels.
- Explore formulation updates to minimize side effects and counter LARC competition.
- Leverage educational campaigns to sustain and grow market share.
Key Takeaways
- ORTHO-CEPT operates in a mature, competitive segment with steady global growth.
- Sales are projected to increase modestly, reaching approximately USD 253 million by 2026.
- Market dynamics favor generic competition, necessitating strategic positioning.
- Regional expansion, especially in emerging markets, offers growth opportunities.
- Ongoing development and regulatory navigation are critical for maintaining and growing sales.
FAQs
1. How does ORTHO-CEPT compare to newer contraceptive methods?
It remains a cost-effective oral contraceptive but faces competition from LARCs, which have higher efficacy and longer durations.
2. What markets offer the highest growth potential for ORTHO-CEPT?
Emerging markets in Asia, Africa, and Latin America are key due to increasing contraceptive adoption and lower market saturation.
3. How does patent status influence sales projections?
Being off-patent allows generic versions to compete, generally reducing profit margins but expanding volume.
4. What regulatory challenges could impact sales?
Stringent approval processes, labeling changes, and safety concerns could delay or restrict product availability.
5. What strategies could extend the product’s market life?
Formulation improvements, targeted marketing, and expanding into new regional markets are effective.
References
[1] Global Market Insights. (2022). "Oral Contraceptives Market Size & Trends." Retrieved from marketinsights.com
[2] Research and Markets. (2021). "Global Contraceptive Market Forecast to 2026." Retrieved from researchandmarkets.com
[3] U.S. Food & Drug Administration. (1972). "Approval of ORTHO-CEPT." https://www.fda.gov