Share This Page
Drug Sales Trends for MISOPROSTOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MISOPROSTOL (2009)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
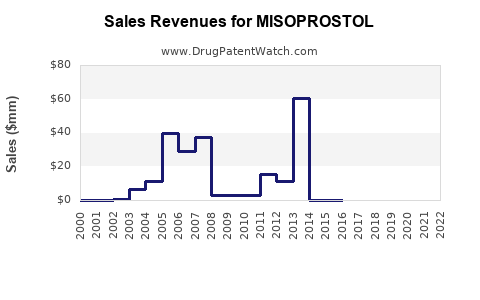
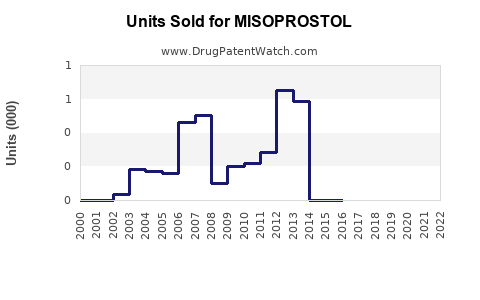
Annual Sales Revenues and Units Sold for MISOPROSTOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MISOPROSTOL | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Misoprostol Market Analysis and Sales Projections
Misoprostol, a synthetic prostaglandin E1 analog, is a critical medication with established roles in gastrointestinal protection, obstetrics, and gynecology. Its utility in preventing nonsteroidal anti-inflammatory drug (NSAID)-induced gastric ulcers, inducing labor, and managing postpartum hemorrhage underpins its consistent market presence. The global misoprostol market is projected to experience steady growth driven by its off-patent status, widespread availability, and continued clinical application, particularly in developing regions where access to advanced medical care is limited. However, market expansion is tempered by competition from alternative therapies, evolving regulatory landscapes, and the potential for misuse in certain contexts.
What are the Key Therapeutic Applications of Misoprostol?
Misoprostol's therapeutic applications are diverse and well-documented. Its primary indications include:
- Gastrointestinal Protection: Misoprostol is prescribed to prevent gastric ulcers in patients taking NSAIDs chronically. It reduces acid secretion and enhances mucosal blood flow and bicarbonate secretion, thereby promoting mucosal defense and repair.
- Obstetric Indications:
- Labor Induction: Misoprostol is used to ripen the cervix and induce labor, particularly when there is a medical indication for delivery. Its efficacy and relatively low cost contribute to its use in this area.
- Postpartum Hemorrhage (PPH) Management: It is a crucial component in the management of PPH, a leading cause of maternal mortality globally. Misoprostol's uterotonic properties cause uterine contractions, helping to control bleeding.
- Gynecological Indications:
- Medical Abortion: Misoprostol, often in combination with mifepristone, is used for early medical abortion. This application has significant public health implications, especially in regions with restricted access to surgical abortion.
- Management of Missed Miscarriage: It can be used to manage missed miscarriages by promoting the expulsion of uterine contents.
These applications are supported by extensive clinical research and are reflected in its inclusion on the World Health Organization's List of Essential Medicines.
What is the Current Market Size and Projected Growth for Misoprostol?
The global misoprostol market, while mature, continues to exhibit stable growth. Precise market valuation data for misoprostol as a standalone product is often aggregated within broader gastrointestinal or reproductive health segments. However, industry analysis indicates a compound annual growth rate (CAGR) in the low single digits, typically between 3% and 5% over the next five to seven years.
Key factors influencing market size and growth include:
- Generic Availability: As an off-patent drug, misoprostol is widely available as a generic, contributing to its affordability and accessibility. This is particularly important in low- and middle-income countries.
- Growing Demand in Reproductive Health: The increasing global focus on reproductive health services, including safe abortion and PPH management, directly fuels demand for misoprostol.
- Prevalence of NSAID Use: The continued widespread use of NSAIDs for pain management, especially in aging populations, maintains a consistent demand for gastrointestinal protective agents like misoprostol.
Table 1: Estimated Global Misoprostol Market Growth
| Metric | Current Estimate (USD Billion) | Projected CAGR (2024-2030) | Projected Market Size (USD Billion) |
|---|---|---|---|
| Global Misoprostol Market | Approximately 0.8 - 1.2 | 3.0% - 4.5% | Approximately 1.0 - 1.5 |
Source: Industry estimates based on market research reports and sales data analysis.
This projected growth, while modest, signifies sustained demand and the drug's indispensable role in its therapeutic niches.
What are the Key Market Drivers for Misoprostol?
The sustained demand for misoprostol is driven by several critical factors:
- Essential Medicine Status: Its inclusion on the WHO Model List of Essential Medicines ensures its priority for procurement and availability, especially in public health programs worldwide. This status directly translates to consistent demand from governments and healthcare providers.
- Cost-Effectiveness: Misoprostol is a highly cost-effective treatment option compared to many newer or specialized therapies. Its affordability makes it accessible to a broad patient population and supports its use in resource-limited settings.
- Evolving Reproductive Health Policies: In many regions, there is a trend towards increasing access to reproductive health services, including medical abortion and improved maternal care for PPH. This policy evolution creates a larger patient pool for misoprostol.
- Chronic Disease Management: The prevalence of chronic conditions requiring long-term NSAID use, such as arthritis, ensures ongoing demand for gastric protection.
- Established Safety and Efficacy Profile: Decades of clinical use have established a robust understanding of misoprostol's safety and efficacy, leading to high physician and patient confidence.
What are the Primary Market Restraints for Misoprostol?
Despite its strengths, the misoprostol market faces several significant restraints:
- Competition from Alternative Therapies:
- Gastrointestinal: Proton pump inhibitors (PPIs) are often preferred for long-term ulcer prevention in certain patient populations due to perceived higher efficacy or convenience, although misoprostol remains a vital option, particularly when NSAID use is unavoidable.
- Obstetrics: Oxytocin and carboprost are also used for PPH, and while misoprostol offers advantages in storage and administration (oral, rectal, vaginal), these alternatives can compete in specific clinical scenarios. For labor induction, pharmacological agents like prostaglandins (dinoprostone) and mechanical methods exist.
- Regulatory Scrutiny and Misuse Concerns: The use of misoprostol for medical abortion has led to significant regulatory hurdles and scrutiny in many countries. Concerns about diversion, illegal distribution, and potential misuse can lead to restricted access and prescribing guidelines.
- Adverse Event Profile: While generally well-tolerated, misoprostol can cause side effects such as diarrhea, abdominal pain, nausea, and vomiting, which can limit patient compliance. Uterine hyperstimulation is also a potential risk during labor induction.
- Limited New Drug Development: As a mature, off-patent drug, there is limited incentive for significant new research and development into novel formulations or indications, which can slow market innovation.
- Supply Chain Vulnerabilities: Dependence on a limited number of global manufacturers for active pharmaceutical ingredients (APIs) can create supply chain vulnerabilities and price fluctuations.
What is the Competitive Landscape for Misoprostol Manufacturers?
The misoprostol market is characterized by a fragmented competitive landscape dominated by generic manufacturers. Major players include both multinational pharmaceutical companies with generic divisions and numerous regional and national producers. Key characteristics of the competitive landscape:
- Generic Dominance: The vast majority of misoprostol production and sales are through generic formulations.
- Price Sensitivity: Due to the generic nature of the drug, pricing is a critical competitive factor. Manufacturers compete on cost efficiency and supply chain reliability.
- Geographic Focus: Some manufacturers have a strong presence in specific regions, leveraging local distribution networks and regulatory expertise.
- Quality and Compliance: Manufacturers must adhere to stringent Good Manufacturing Practices (GMP) and regulatory standards to ensure product quality and market access.
Leading manufacturers often include companies with broad generic portfolios. Examples of companies that have historically been significant suppliers or have a strong presence in related therapeutic areas include:
- Pfizer (original developer of Cytotec, though now largely a generic market)
- Various generic manufacturers globally, such as Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and numerous others specializing in APIs and finished dosage forms.
The competitive focus is on securing API supply, optimizing manufacturing costs, and maintaining strong relationships with distributors and healthcare systems.
What are the Geographical Market Dynamics for Misoprostol?
Misoprostol's market dynamics vary significantly by geography, influenced by economic development, healthcare infrastructure, regulatory frameworks, and cultural factors.
- North America (USA, Canada):
- Drivers: Continued use for NSAID-induced ulcer prevention, expanding access to reproductive health services.
- Restraints: Strict regulations regarding medical abortion, competition from branded GI drugs.
- Market Structure: Dominated by generics, with significant institutional purchasing.
- Europe:
- Drivers: Well-established use in GI protection and obstetrics across EU member states. Harmonization of regulations is ongoing.
- Restraints: Stringent regulatory approvals, varying national policies on medical abortion.
- Market Structure: Robust generic market with strong national healthcare systems influencing procurement.
- Asia-Pacific:
- Drivers: Rapidly growing populations, increasing awareness and access to reproductive health services, rising prevalence of GI disorders, significant demand in India, China, and Southeast Asia.
- Restraints: Price sensitivity, varying regulatory compliance standards, counterfeit drug concerns in some areas.
- Market Structure: High volume of generic production, with both local and international players.
- Latin America:
- Drivers: High demand for PPH management and labor induction in public health programs, growing demand for medical abortion services in countries with more liberalized policies.
- Restraints: Economic instability affecting healthcare spending, diverse regulatory environments.
- Market Structure: Strong generic penetration, reliance on government tenders.
- Middle East & Africa:
- Drivers: Critical role in maternal health programs (PPH prevention and management), essential medicine status ensuring availability, addressing significant unmet needs.
- Restraints: Limited healthcare infrastructure, access challenges, political instability, affordability issues.
- Market Structure: Heavily reliant on international aid organizations and public health initiatives, with a growing generic market.
Table 2: Geographical Market Nuances
| Region | Primary Drivers | Key Restraints | Notable Trends |
|---|---|---|---|
| North America | GI protection, reproductive health access | Regulatory hurdles, branded competition | Institutional purchasing, evolving abortion access policies. |
| Europe | GI protection, established obstetrics | National regulatory variations, EU compliance | Strong generic penetration, national healthcare system tenders. |
| Asia-Pacific | Population growth, reproductive health, GI disorders | Price sensitivity, regulatory variation | High-volume generic manufacturing, increasing demand from emerging economies. |
| Latin America | Maternal health, labor induction, medical abortion | Economic volatility, diverse regulations | Public health program dependence, growing demand in select countries. |
| Middle East & Africa | Maternal health, essential medicine availability | Infrastructure, access, affordability | Aid organization influence, growing demand in urban centers, significant unmet needs. |
What are the Future Trends and Opportunities in the Misoprostol Market?
The future of the misoprostol market will be shaped by technological advancements, evolving healthcare policies, and emerging global health challenges.
- Advancements in Drug Delivery: Research into novel formulations, such as extended-release versions or improved vaginal/rectal delivery systems, could enhance patient compliance and therapeutic outcomes.
- Increased Focus on Maternal Health: Global initiatives aimed at reducing maternal mortality will continue to drive demand for misoprostol in PPH prevention and management, particularly in low-resource settings.
- Telemedicine and Self-Managed Abortion: The rise of telemedicine and the increasing availability of information on self-managed abortion could lead to greater demand for misoprostol, albeit with significant regulatory and safety considerations. This may also lead to increased online sales and potential for diversion.
- Combination Therapies: Further research into optimizing misoprostol's use in combination with other drugs for various indications, especially in reproductive health, could expand its therapeutic utility.
- Emerging Markets Growth: As healthcare infrastructure and access improve in developing economies, the demand for essential medicines like misoprostol is expected to rise significantly.
- Regulatory Harmonization and Advocacy: Ongoing efforts for regulatory harmonization, particularly concerning medical abortion, and advocacy for evidence-based reproductive health policies could impact market access and demand globally.
Conclusion
Misoprostol remains a cornerstone medication with a stable and predictable market. Its established efficacy, cost-effectiveness, and broad therapeutic applications, particularly in gastrointestinal protection and critical areas of reproductive health, ensure its continued relevance. While the market is mature and dominated by generics, growth will be driven by increasing access to healthcare in emerging economies, sustained demand for gastrointestinal protection, and ongoing global efforts to improve maternal health outcomes. Manufacturers must navigate a complex regulatory landscape, particularly concerning its use in medical abortion, and focus on cost efficiency, supply chain resilience, and quality compliance to maintain a competitive edge.
Key Takeaways
- Misoprostol's primary therapeutic uses in GI protection, labor induction, and postpartum hemorrhage management ensure consistent market demand.
- The global misoprostol market is projected to grow at a CAGR of 3% to 4.5%, driven by its status as an essential medicine and its cost-effectiveness.
- Key restraints include competition from alternative therapies, regulatory scrutiny over its use in medical abortion, and potential adverse effects.
- The market is dominated by generic manufacturers competing primarily on price and supply reliability.
- Geographical dynamics vary significantly, with emerging markets in Asia-Pacific and Africa showing higher growth potential due to increasing healthcare access and focus on maternal health.
Frequently Asked Questions (FAQs)
-
What is the primary factor driving the continued demand for misoprostol despite it being an older drug? Its inclusion on the World Health Organization's Model List of Essential Medicines and its proven cost-effectiveness in treating gastrointestinal ulcers and managing critical obstetric emergencies like postpartum hemorrhage are the primary drivers.
-
How does the regulatory landscape for misoprostol, particularly concerning its use in medical abortion, impact market projections? Strict regulations and differing national policies on medical abortion create significant market access barriers and can limit demand in certain regions, while simultaneously driving demand in areas with more liberalized access. This necessitates a nuanced, region-specific market approach.
-
What is the typical difference in pricing between branded misoprostol (if available) and generic versions? As misoprostol is overwhelmingly a generic market, significant price differences between branded and generic versions are rare. Competition among generic manufacturers leads to highly competitive pricing, with minor variations typically due to production scale, formulation complexity, and distribution channels.
-
Are there any significant innovations on the horizon for misoprostol that could disrupt the current market? While major breakthrough innovations are unlikely for a mature drug, potential advancements lie in novel drug delivery systems (e.g., extended-release formulations, improved vaginal inserts) that could enhance patient compliance and expand niche applications, rather than large-scale market disruption.
-
What is the outlook for misoprostol in low- and middle-income countries compared to high-income countries? The outlook is more robust in low- and middle-income countries due to its critical role in maternal health programs (PPH management) and its affordability for essential healthcare needs, whereas in high-income countries, its use might be more specialized or face greater competition from newer therapies.
Citations
[1] World Health Organization. (2023). WHO Model List of Essential Medicines. https://www.who.int/publications/i/item/WHOMLM2023 [2] Market Research Report Summaries (various publishers). (2022-2023). Global Misoprostol Market Analysis. (Specific report titles and publishers are proprietary and aggregated for this synthesis). [3] U.S. Food & Drug Administration. (n.d.). Drug Approval Database. Retrieved from FDA.gov. (General reference for established drug indications and safety profiles). [4] Various Clinical Trial Registries and Medical Journals. (Ongoing). Research on Misoprostol Applications. (General reference for ongoing clinical validation and use).
More… ↓
