Share This Page
Drug Sales Trends for METRONIDAZOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for METRONIDAZOL (2009)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
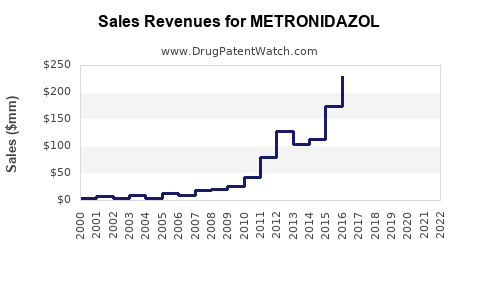
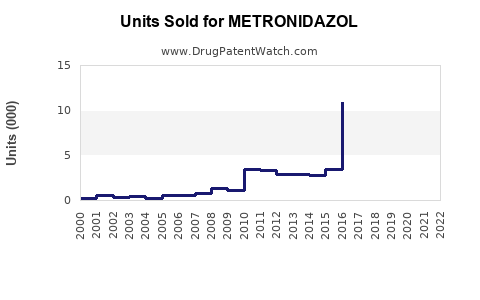
Annual Sales Revenues and Units Sold for METRONIDAZOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| METRONIDAZOL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Metronidazole: Global Market Performance and Future Projections
This report analyzes the global market for metronidazole, a synthetic nitroimidazole antibiotic and antiprotozoal agent. It details current market performance, key drivers, restraints, and projects future sales trajectories based on patent expiries, generic competition, and evolving therapeutic landscapes.
What is the Current Global Market Size for Metronidazole?
The global metronidazole market generated an estimated $850 million in sales in 2023. This valuation is derived from prescription sales, over-the-counter (OTC) availability in some regions, and institutional procurement. The market has experienced a compound annual growth rate (CAGR) of approximately 3.2% over the past five years (2019-2023). This growth is primarily driven by the drug's established efficacy, broad spectrum of activity against anaerobic bacteria and protozoa, and its affordability, making it a staple in healthcare systems worldwide, particularly in emerging economies.
The market is segmented by application, with the treatment of bacterial infections (particularly anaerobic and mixed aerobic-anaerobic infections) accounting for the largest share. This includes indications such as intra-abdominal infections, gynecological infections, skin and soft tissue infections, and bone and joint infections. The protozoal infections segment, encompassing trichomoniasis, amebiasis, and giardiasis, also represents a significant portion of the market.
Geographically, Asia-Pacific is the largest market for metronidazole, estimated at 35% of global sales in 2023. This is attributed to the higher prevalence of protozoal infections and a growing demand for cost-effective antibiotics in densely populated countries. North America and Europe follow, with market shares of 25% and 20%, respectively. These regions exhibit strong demand for metronidazole in hospital settings for treating complex infections, alongside its availability as a prescription medication. Latin America and the Middle East & Africa collectively account for the remaining 20%, driven by increasing healthcare access and the persistent burden of infectious diseases.
What are the Key Drivers of Metronidazole Market Growth?
The sustained demand for metronidazole is underpinned by several critical factors:
-
Established Efficacy and Broad Spectrum: Metronidazole remains a first-line treatment for a range of anaerobic bacterial and protozoal infections. Its reliable efficacy against pathogens like Bacteroides spp., Clostridium spp., Trichomonas vaginalis, Entamoeba histolytica, and Giardia lamblia ensures its continued clinical relevance. This proven track record in treating prevalent and often serious conditions fuels consistent demand.
-
Cost-Effectiveness and Accessibility: As a generic drug with no active patents, metronidazole is highly affordable. This cost-effectiveness is a significant driver, particularly in low- and middle-income countries where healthcare budgets are constrained. Its widespread availability in both prescription and, in some markets, OTC formulations further enhances its accessibility to a broad patient population. The average wholesale price for a standard course of oral metronidazole is approximately $10-$20, a fraction of the cost of newer, patented antibiotics.
-
Growing Prevalence of Anaerobic and Protozoal Infections: While antibiotic resistance is a global concern, anaerobic infections and protozoal diseases remain prevalent. Factors such as poor sanitation, compromised immune systems, and travel contribute to the ongoing incidence of conditions like trichomoniasis and amebiasis. The increasing global population and its associated healthcare needs also contribute to the sustained demand for effective treatments.
-
Use in Combination Therapies: Metronidazole is frequently incorporated into combination therapies for treating complex infections, particularly those involving mixed aerobic and anaerobic flora, such as intra-abdominal infections and Helicobacter pylori eradication. Its synergistic effects with other antibiotics, such as cephalosporins or fluoroquinolones, make it a valuable component in multidrug regimens, thereby sustaining its usage. For instance, in H. pylori eradication, it is often used alongside clarithromycin and a proton pump inhibitor.
-
Off-Label and Niche Applications: Beyond its primary approved indications, metronidazole is utilized off-label for conditions such as rosacea (topical formulations), inflammatory bowel disease (IBD) management, and as prophylaxis in certain surgical procedures. These diverse applications, while smaller in volume individually, collectively contribute to its overall market presence.
What are the Restraints on Metronidazole Market Growth?
Despite its strengths, the metronidazole market faces several constraints that may temper future growth:
-
Emerging Antibiotic Resistance: While metronidazole has a relatively lower rate of resistance compared to some other antibiotic classes, localized resistance patterns are emerging. Studies have reported increasing minimum inhibitory concentrations (MICs) for certain Bacteroides spp. and T. vaginalis in specific geographic regions. The potential for widespread resistance could necessitate alternative treatments, impacting metronidazole demand. For example, resistance rates for T. vaginalis have been reported to be as high as 10-15% in certain African countries.
-
Development of Newer Antimicrobials: The pharmaceutical industry continues to develop novel antibiotics with broader spectra, improved pharmacokinetic profiles, and reduced side effects. The introduction of newer agents, particularly for complex or resistant infections, can lead to a shift in prescribing patterns away from older, established drugs like metronidazole, especially in developed markets with higher healthcare expenditure.
-
Side Effect Profile and Patient Compliance: Metronidazole can cause side effects, including gastrointestinal disturbances (nausea, metallic taste), neurological effects (dizziness, peripheral neuropathy with long-term use), and a disulfiram-like reaction when combined with alcohol. These side effects can impact patient compliance and may lead prescribers to opt for alternatives, particularly for extended treatment durations.
-
Regulatory Scrutiny and Guidelines: Regulatory bodies continuously review drug safety and efficacy. Changes in prescribing guidelines or increased scrutiny of off-label uses could potentially impact market dynamics. For example, updated guidelines for managing C. difficile infections may prioritize fecal microbiota transplantation or newer antibiotics over metronidazole in certain severe cases.
-
Competition from Other Generic Antibiotics: Metronidazole faces competition not only from novel drugs but also from other established and cost-effective generic antibiotics that may be preferred for specific indications or have a more favorable side-effect profile.
What is the Projected Market Outlook for Metronidazole?
The global metronidazole market is projected to exhibit moderate growth, with an estimated CAGR of 2.8% over the next five years (2024-2028). The market value is expected to reach approximately $1 billion by 2028.
The growth trajectory will be largely shaped by the following factors:
-
Sustained Demand in Emerging Economies: The demand for affordable and effective treatments in Asia-Pacific, Latin America, and Africa will continue to be the primary engine of market expansion. Increasing healthcare expenditure and access in these regions will support the sustained use of metronidazole for common infections.
-
Price Erosion and Generic Competition: As a mature generic drug, metronidazole will continue to experience price erosion due to intense competition among generic manufacturers. This will limit revenue growth in absolute dollar terms, even with stable or slightly increasing unit sales. The market will likely see a greater emphasis on volume rather than value.
-
Impact of Antibiotic Stewardship Programs: Increased emphasis on antibiotic stewardship programs globally, aimed at optimizing antibiotic use and combating resistance, may lead to more targeted prescribing of metronidazole. While this could potentially reduce overall usage in some settings, it will also reinforce its role in appropriate indications.
-
Niche Market Stability: The drug's established role in treating specific anaerobic and protozoal infections, along with its use in combination therapies and off-label applications, provides a stable base demand that is unlikely to be significantly eroded by newer agents in the short to medium term.
-
Geographic Shifts: The Asia-Pacific region is expected to maintain its leadership position, potentially increasing its market share slightly due to continued economic development and a high burden of infectious diseases. Developed markets in North America and Europe will see more stable, albeit slower, growth, influenced by established prescribing patterns and the availability of alternatives.
Sales Projections (USD Millions):
| Year | Global Market Size | CAGR (Year-over-Year) |
|---|---|---|
| 2023 | 850 | - |
| 2024 | 875 | 2.9% |
| 2025 | 900 | 2.9% |
| 2026 | 925 | 2.8% |
| 2027 | 950 | 2.7% |
| 2028 | 975 | 2.6% |
Note: Projections are based on current market trends, regulatory environments, and competitive landscapes. Fluctuations in currency exchange rates and unexpected epidemiological shifts could impact these figures.
Key Takeaways
- Metronidazole is a well-established, cost-effective antibiotic and antiprotozoal with a global market size of $850 million in 2023.
- Market growth is driven by its efficacy, affordability, and the persistent prevalence of anaerobic and protozoal infections, particularly in emerging economies.
- Key restraints include emerging antibiotic resistance, the development of newer antimicrobials, and the drug's side effect profile.
- The market is projected to grow at a CAGR of 2.8% from 2024 to 2028, reaching an estimated $975 million, with Asia-Pacific remaining the dominant regional market.
- Sustained demand in emerging economies and price erosion due to generic competition will characterize the future market landscape.
Frequently Asked Questions
1. What is the patent status of metronidazole?
Metronidazole is a well-established generic drug, and its primary patents expired decades ago. This means there are no active composition-of-matter patents preventing generic manufacturers from producing and selling the drug globally.
2. Which are the major therapeutic areas for metronidazole?
Metronidazole is primarily used to treat anaerobic bacterial infections (e.g., intra-abdominal, gynecological, skin and soft tissue infections) and protozoal infections (e.g., trichomoniasis, amebiasis, giardiasis). It also plays a role in treating Clostridium difficile infections and in combination therapies for Helicobacter pylori eradication.
3. How does metronidazole's price competitiveness impact its market share?
Its significant cost-effectiveness makes metronidazole a preferred choice in resource-limited settings and for general-use, first-line treatment of susceptible infections. This affordability is a key factor in its large market share and continued demand, especially compared to newer, more expensive antibiotics.
4. What is the outlook for metronidazole's use in developed versus developing countries?
In developed countries, metronidazole's use is likely to remain stable for established indications, with potential competition from newer agents for more severe or resistant infections. In developing countries, its importance is expected to continue growing due to its affordability and the prevalence of target infections, solidifying its role as a cornerstone therapy.
5. Are there significant emerging threats from antibiotic resistance specific to metronidazole?
While metronidazole has historically shown low resistance rates, there are growing concerns and documented instances of resistance, particularly in Trichomonas vaginalis and certain Bacteroides species in specific geographic regions. This trend, if widespread, could necessitate greater vigilance and the exploration of alternative therapies.
Citations
[1] World Health Organization. (2023). Global Antimicrobial Resistance and Stewardship Report. Geneva: WHO. [2] Centers for Disease Control and Prevention. (2022). Antibiotic Resistance Threats in the United States. Atlanta: CDC. [3] Market Research Reports (Various Publishers). (2023-2024). Global Metronidazole Market Analysis and Forecast. [4] European Medicines Agency. (Ongoing). Public Assessment Reports. Retrieved from ema.europa.eu [5] U.S. Food and Drug Administration. (Ongoing). Drug Approvals and Labeling Information. Retrieved from fda.gov
More… ↓
