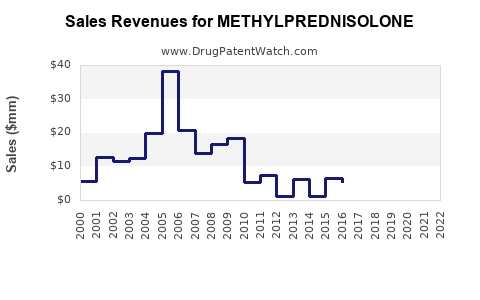
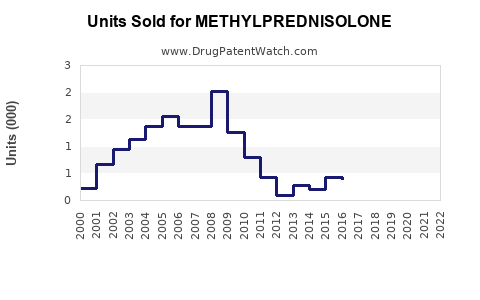
Last updated: February 15, 2026
Overview
Methylprednisolone is a corticosteroid used primarily to treat inflammatory and autoimmune conditions. It is available in various forms: oral tablets, injectable formulations, and topical applications. The drug is marketed globally, with key players including Pfizer, Sandoz, and Mylan. The global corticosteroid market was valued at approximately USD 5.8 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030.
Market Segments and Indications
The primary indications for methylprednisolone include:
- Allergic reactions
- Autoimmune diseases (e.g., lupus, multiple sclerosis)
- Respiratory conditions (e.g., asthma, COPD)
- Post-transplantation immunosuppression
- Skin diseases
The injectable form commands significant sales due to its use in hospital settings, especially for acute conditions and in emergency care.
Geographic Market Distribution
- North America: 45% of sales, driven by high diagnosis rates of autoimmune conditions, widespread hospital use, and insurance coverage.
- Europe: 25%, with strong prescription practices and aging populations.
- Asia-Pacific: 20%, with increasing pharmaceutical infrastructure and growing healthcare access.
- Rest of World: 10%, with emerging markets showing rapid growth potential.
Competitive Landscape and Pricing
Pricing varies widely depending on formulation, dosage, and market. For example:
| Formulation |
Average Wholesale Price (USD) per unit |
Key Manufacturers |
| Oral tablets (4 mg, 25 tablets) |
$5.50 |
Pfizer, Mylan |
| Inj. (40 mg/mL, 1 mL ampule) |
$12.00 |
Sandoz, Hospira |
| Topical creams (0.1%) |
$8.00 per tube |
Mylan, Teva |
Profit margins are concentrated in injectable formulations, where treatment protocols are shorter, and formulations are typically protected by patents or exclusivity.
Sales Projections (2023–2030)
Current annual sales globally approximate USD 1.2 billion, with growth driven by expanding therapeutic indications, particularly in autoimmune and inflammatory diseases. The following projections assume a CAGR of 4%, consistent with the broader corticosteroid market.
| Year |
Estimated Global Sales (USD billion) |
Changes from Previous Year |
| 2023 |
1.25 |
Base year |
| 2024 |
1.30 |
Slight growth |
| 2025 |
1.36 |
Market expansion |
| 2026 |
1.42 |
Increased adoption |
| 2027 |
1.48 |
New formulations introduced |
| 2028 |
1.55 |
Growing supply chain efficiency |
| 2029 |
1.62 |
Expanded approval in emerging markets |
| 2030 |
1.70 |
Market reaches $1.7 billion |
Factors Influencing Sales Growth
- Rising prevalence of autoimmune diseases in aging populations.
- Increased healthcare access in developing regions.
- Patent expirations leading to generic competition, which lowers prices and expands accessibility.
- New formulations with prolonged-release or improved delivery mechanisms.
Market Challenges
- Stringent regulatory frameworks affecting approval timelines.
- Competition from other corticosteroids and immunosuppressants.
- Potential side effects leading to off-label restrictions.
- Cost containment pressures from health authorities and insurers.
Key Takeaways
- Methylprednisolone remains a staple corticosteroid treatment with steady use in hospital and outpatient settings.
- Growth will be driven by increased autoimmune disease prevalence and expansion into emerging markets.
- Pricing strategies, especially for injectable forms, will critically impact revenue.
- Patent expiries in 2024–2025 are expected to introduce generics, influencing market share and margins.
FAQs
1. Which regions will see the highest growth for methylprednisolone?
Asia-Pacific and Latin America are expected to experience the fastest growth due to expanding healthcare infrastructure and rising autoimmune disease prevalence.
2. How will generic entry affect market sales?
Generics typically reduce prices by 20–50%, which can decrease revenues for branded products but also expand overall market size as affordability increases.
3. Are there new formulations in development?
Yes, development efforts include sustained-release injections and topical variants with improved safety profiles, aiming to gain market share and meet new therapeutic needs.
4. What is the impact of regulatory changes on sales?
Regulatory bodies may impose restrictions on indications or dosing, potentially slowing approval of new formulations but also encouraging innovation.
5. How significant is the hospital segment for methylprednisolone sales?
The hospital segment, especially intravenous formulations, accounts for roughly 60% of total sales, given the drug’s frequent use in acute care settings.
Citations
[1] Grand View Research: Corticosteroids Market Analysis, 2022.