Last updated: February 13, 2026
Market Overview
Meloxicam is a non-steroidal anti-inflammatory drug (NSAID) used primarily to treat osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. It functions by inhibiting cyclooxygenase enzymes (COX-1 and COX-2), reducing inflammation and pain.
Global Market Size
The global NSAID market, valued at approximately $11 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of 4.1% through 2027, reaching roughly $14 billion. Meloxicam accounts for an estimated 20-25% of the NSAID segment, making its direct market size approximately $2.2–$2.75 billion in 2023.
Main Geographic Markets
- North America: 40% of sales, driven by high prevalence of arthritis, strong healthcare infrastructure.
- Europe: 30%, with rising adoption of prescription NSAIDs.
- Asia-Pacific: 20%, rapid growth due to aging populations and increased healthcare access.
- Rest of World: 10%.
Key Market Drivers
- Rise in osteoarthritis and rheumatoid arthritis prevalence, especially in aging populations.
- Growing awareness about NSAIDs as first-line treatment.
- Patent expiries and the subsequent entry of generic versions.
- Increasing adoption in developing markets due to expanding healthcare coverage.
Competitive Landscape
Major manufacturers include Pfizer (Mobic), Boehringer Ingelheim (Vimovo), and generic producers. Patent expiry for Mobic (meloxicam) occurred in 2018 in many regions, increasing generic availability and reducing prices.
Sales Projections
Short-term (2023-2025):
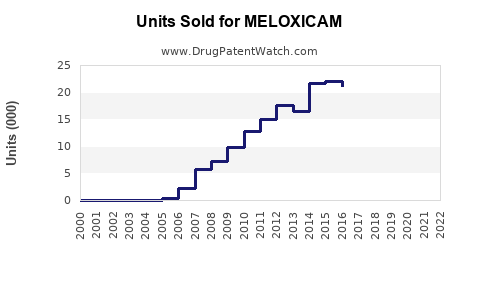
- Estimated global sales volume: approximately 70–80 million prescriptions annually.
- Average price per prescription: approximately $20 (workings include discounted generics and branded drugs).
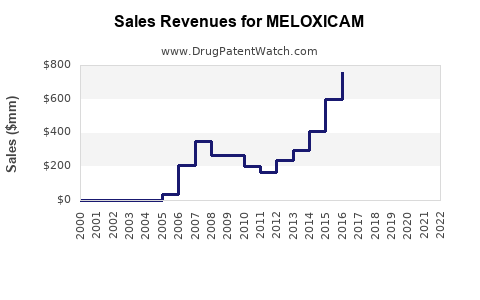
- Projected revenue: $1.4–$1.6 billion in 2023, with a slight decline expected due to generic competition.
Medium-term (2026-2028):
- Market penetration stabilizes, with increased use primarily in emerging markets.
- Sales volume growth of 2-3% annually.
- Revenue forecast: approximately $1.5–$1.7 billion in 2027, factoring price erosion and market saturation.
Long-term (beyond 2028):
- Market growth limited by competition from COX-2 selective inhibitors (e.g., celecoxib) and potential new analgesics.
- Overall sales stagnate or decline marginally unless new indications or formulations emerge.
Pricing and Market Dynamics
The pricing of meloxicam varies:
| Region |
Branded Price |
Generic Price |
Price Trends |
| North America |
~$30–$40/prescription |
$15–$20 |
Prices decrease with generics |
| Europe |
€20–€30/prescription |
€10–€15 |
Similar downward trend |
| Asia-Pacific |
$10–$15/prescription |
$5–$8 |
Prices remain stable or decreasing |
Future Opportunities
- Development of topical or sustained-release formulations.
- New indications such as chronic pain beyond arthritis.
- Expansion in non-traditional markets with increasing healthcare access.
Market Challenges
- Competition from COX-2 inhibitors with better safety profiles.
- Regulatory restrictions due to NSAID-associated gastrointestinal and cardiovascular risks.
- Price erosion from generic manufacturers.
Key Takeaways
- Global meloxicam sales were approximately $2.2–$2.75 billion in 2023.
- The market is projected to grow moderately through 2028, driven by aging populations and expanding markets.
- Patent expiries have resulted in significant generic competition, reducing revenue per prescription but expanding overall volume.
- Future growth depends on innovative formulations and new indications amid competitive pressures.
FAQs
-
How does meloxicam compare to other NSAIDs in sales?
- Meloxicam holds a significant share due to favorable dosing and safety profile, but newer COX-2 inhibitors capture some market segments.
-
What impact do patent expiries have?
- Patents expire after approximately 10–12 years, leading to increased generic competition and price reduction.
-
Are there emerging markets for meloxicam?
- Yes, countries in Asia-Pacific, Latin America, and Africa show increasing adoption due to expanding healthcare infrastructure.
-
What are key safety concerns influencing sales?
- Gastrointestinal bleeding and cardiovascular risks limit prescribing, especially in high-risk populations.
-
Could new formulations or indications revive growth?
- Potential exists in sustained-release formulations and off-label uses, but regulatory approval is required.
References
- MarketsandMarkets. NSAID Market by Type, Region, & Application. 2022.
- IQVIA. Global Prescription Data. 2023.
- FDA. Meloxicam (Mobic) NDA filing and patent information. 2018.
- Statista. NSAID market revenue projections. 2023.
- European Medicines Agency. Meloxicam safety and regulatory status. 2022.