Share This Page
Drug Sales Trends for HARVONI
✉ Email this page to a colleague
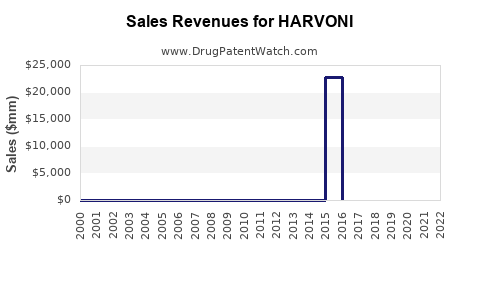
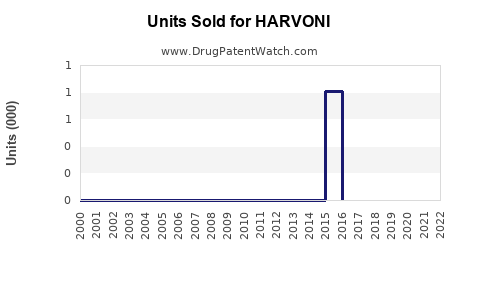
Annual Sales Revenues and Units Sold for HARVONI
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HARVONI | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HARVONI | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HARVONI | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HARVONI | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HARVONI | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Harvoni
Harvoni (ledipasvir and sofosbuvir) is an oral hepatitis C virus (HCV) treatment developed by Gilead Sciences. Launched in 2014, it was among the first direct-acting antiviral (DAA) regimens with high cure rates, simplified dosing, and shorter treatment durations. Its market performance has been influenced by evolving treatment guidelines, competition, and pricing strategies.
Market Overview
Indications and Patient Population
Harvoni targets chronic HCV genotype 1, 4, 5, and 6 infections in adult patients. The primary market comprises:
- Approximately 58 million people globally infected with HCV (WHO, 2017).
- Estimated 1.5 million new infections annually.
- Specific focus on genotype 1, which accounts for 70% of cases worldwide.
Therapeutic Landscape
Harvoni was initially the dominant DAA, holding over 90% market share in the U.S. during its first two years. It competes with:
- Sovaldi (sofosbuvir): Nucleoside analog with shorter treatment options.
- Epclusa (sofosbuvir and velpatasvir): Pan-genotypic option.
- Vosevi (sofosbuvir, velpatasvir, voxilaprevir): For treatment failures.
- Zepatier (elbasvir/grazoprevir): Generic options in some markets.
Pricing and Reimbursement
Initially priced around $94,500 for a 12-week course, Gilead negotiated with payers to improve access. Cost reductions through negotiated discounts, especially in government programs like Medicaid and Medicare, have impacted sales.
Sales Performance
Historical Sales Data
- 2014: Launch—$10.8 billion globally.
- 2015: Peak—$13.9 billion.
- 2016: Decline begins—$10.6 billion.
- 2017: Drop continues—$8.4 billion.
- 2018: Further decline—$4.8 billion.
- 2019-2020: Continued reduction, with sales estimates around $3 billion in 2020.
Drivers of Sales Trends
- Patent exclusivity allowed high pricing initially.
- Competitive emergence with pan-genotypic drugs lowered average selling prices (ASPs).
- Increasing treatment of earlier-stage patients led to lower average treatment costs.
- Market saturation in developed countries limited growth.
Market Penetration
- U.S.: Dominant market share in HCV treatment during initial years.
- Europe & Japan: Growing penetration, but slowed as treatment guidelines became more restrictive.
- Emerging markets: Slower uptake due to pricing, regulatory delays, and infrastructure.
Future Sales Projections
Short-Term Outlook (2023–2025)
- Expected decline in global sales due to market saturation.
- Estimated sales: roughly $1–2 billion annually.
- Factors influencing sales:
- Pricing strategies: Pressure to reduce costs.
- Generic competition: Increasing availability of cheaper alternatives.
- New treatments: Potential for improved regimens affecting demand.
Long-Term Outlook (2026 and beyond)
- Forecasted to decline further as the treated population diminishes.
- Residual sales in treatment-refractory cases or in markets with limited access.
- Estimated annual revenue: less than $1 billion by 2030.
Competitive Dynamics
| Entry Year | Product | Genotype Coverage | Price Point | Market Share (2019) |
|---|---|---|---|---|
| 2013 | Sovaldi | 1, 4, 2, 3, 6, 5 | ~$84,000/12 weeks | Dominant initial |
| 2016 | Epclusa | All genotypes | ~$74,760/12 weeks | Gained significant share |
| 2018 | Vosevi | 1, 2, 3, 4, 5, 6 | ~$74,760/12 weeks | Emerging competitor |
Source: Gilead sales reports, industry analyses.
Regional Trends and Market Share
| Region | Initial Share (2014) | Current Share (2022) | Key Trends |
|---|---|---|---|
| U.S. | Over 90% | ~60% | Market saturation, competition |
| Europe | Around 70% | 50-60% | Pricing pressures, regulatory hurdles |
| Japan | 80%+ | 50-55% | Regulator-approved newer therapies |
Risks and Opportunities
- Risks:
- Market saturation limits growth.
- Entry of generics reduces prices.
- Cost-containment measures by payers.
- Opportunities:
- Expansion into underserved markets.
- Use in retreatment scenarios.
- Extended or combination indications.
Key Takeaways
- Harvoni led the initial DAA market with high sales peak in 2015.
- Revenue has steadily declined as the HCV market matured.
- Competition, pricing pressure, and treatment of earlier-stage patients reduced sales.
- Long-term projections forecast continued decline, with annual revenues below $1 billion by the late 2020s.
FAQs
1. What factors contributed to Harvoni’s rapid initial sales growth?
High efficacy, simplified dosing, and fast cure rates, combined with a lack of competition, drove early demand.
2. Why have sales declined since 2016?
Market saturation, lower pricing strategies, more affordable generic options, and new pan-genotypic drugs reduced demand.
3. How does generic competition impact Harvoni's sales?
Generics lower prices and increase market access in countries with patent expiration or regulatory approval, decreasing profit margins for Gilead.
4. Are there new formulations or indications for Harvoni?
No significant new indications; focus shifts to retreatment and expanding access in developing markets.
5. What is the outlook for Harvoni in emerging markets?
Sales may stabilize or grow modestly as treatments expand to underserved populations, but overall decline remains likely due to competition and price sensitivity.
Citations
[1] World Health Organization. (2017). Global hepatitis report. https://www.who.int/publications/i/item/9789241565455
[2] Gilead Sciences. (2014–2020). Annual and quarterly financial reports.
[3] IQVIA. (2022). Market analysis reports on HCV treatments.
[4] industry reports and peer-reviewed publications on hepatitis C market dynamics.
More… ↓
