Share This Page
Drug Sales Trends for DIVALPROEX
✉ Email this page to a colleague
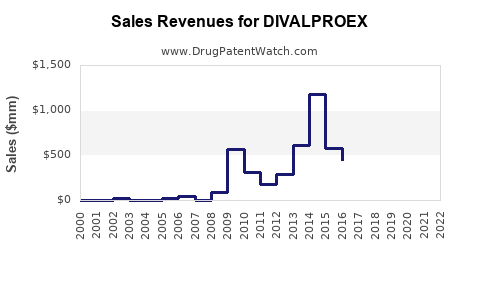
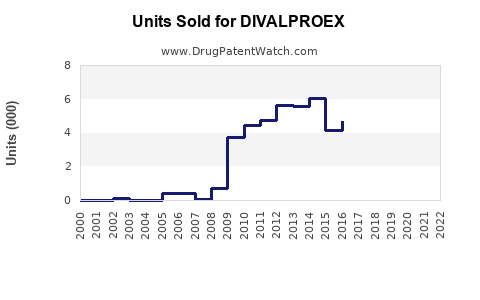
Annual Sales Revenues and Units Sold for DIVALPROEX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DIVALPROEX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DIVALPROEX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DIVALPROEX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DIVALPROEX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Divalproex Sodium Market Analysis and Sales Projections
Divalproex sodium, a combination of sodium valproate and valproic acid, is an antiepileptic and mood-stabilizing drug. Its primary indications include the treatment of epilepsy, bipolar disorder, and migraine prophylaxis. The market for divalproex sodium is mature, with established generic competition. However, ongoing clinical research and its established efficacy continue to support its market presence.
What is the Current Market Size for Divalproex Sodium?
The global market for divalproex sodium is estimated to be approximately USD 1.2 billion in 2023. This valuation is derived from sales data of both branded and generic formulations across major pharmaceutical markets, including North America, Europe, and Asia-Pacific.
Key Market Segments by Indication:
- Epilepsy: This segment represents the largest share, accounting for approximately 60% of divalproex sodium sales. It is used to treat various seizure types, including generalized tonic-clonic, partial, and complex partial seizures.
- Bipolar Disorder: This segment contributes about 35% of the market. Divalproex sodium is a first-line treatment for manic and mixed episodes associated with bipolar I disorder and for seizure control in individuals with epilepsy who also experience mood disorders.
- Migraine Prophylaxis: This segment accounts for the remaining 5% of sales. Its role in preventing migraines is well-documented, though other agents are also widely used.
Geographic Distribution of Sales (2023 Estimates):
- North America: 45%
- Europe: 30%
- Asia-Pacific: 15%
- Rest of the World: 10%
North America dominates due to high healthcare spending and established treatment protocols. Europe follows closely, with a significant patient population and widespread access to the drug. The Asia-Pacific region is showing growth driven by increasing diagnosis rates and improving healthcare infrastructure.
Who are the Key Manufacturers and Competitors?
The divalproex sodium market is characterized by a strong presence of generic manufacturers, alongside the originator product. Competition is primarily based on price and market penetration.
Major Manufacturers and Product Offerings:
- AbbVie Inc. (Depakote®): The originator brand, Depakote®, maintains a significant market share due to brand recognition and established patient trust. It is available in delayed-release and extended-release formulations.
- Generic Manufacturers: Numerous companies produce generic divalproex sodium, including but not limited to:
- Teva Pharmaceutical Industries Ltd.
- Apotex Inc.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Lupin Ltd.
These generic manufacturers compete aggressively on price, making cost-effectiveness a key differentiator. The market is fragmented, with no single generic manufacturer holding a dominant market share.
Competitive Landscape Dynamics:
- Price Erosion: The availability of multiple generic versions has led to significant price erosion since the patent expiry of Depakote®.
- Formulation Differences: While the active pharmaceutical ingredient is the same, variations in extended-release technologies and dissolution profiles can exist between different manufacturers, influencing prescribing patterns.
- Supply Chain Reliability: Consistent supply and robust distribution networks are critical for maintaining market share, particularly for generic providers.
What are the Key Drivers and Restraints for Market Growth?
Several factors influence the demand and supply dynamics of divalproex sodium.
Market Drivers:
- High Prevalence of Target Diseases: The persistent high incidence of epilepsy and bipolar disorder globally necessitates continuous treatment options, supporting sustained demand for divalproex sodium. The World Health Organization estimates that approximately 50 million people worldwide have epilepsy [1].
- Established Efficacy and Safety Profile: Divalproex sodium has a long history of clinical use, with a well-established efficacy and safety profile for its approved indications. This familiarity among healthcare providers reduces barriers to prescription.
- Cost-Effectiveness of Generic Options: The availability of affordable generic divalproex sodium makes it a viable treatment option, particularly in healthcare systems with budget constraints and for long-term patient management.
- Broad Spectrum of Activity: Its ability to treat multiple seizure types in epilepsy and its mood-stabilizing properties make it a versatile therapeutic agent.
Market Restraints:
- Emergence of Newer Therapies: The development of newer antiepileptic drugs (AEDs) and mood stabilizers with potentially improved side-effect profiles or novel mechanisms of action poses a competitive threat.
- Side Effect Profile: Divalproex sodium is associated with a range of potential side effects, including weight gain, hair loss, tremor, cognitive impairment, and hepatotoxicity. Teratogenicity concerns in pregnant women are also a significant limitation.
- Regulatory Scrutiny and Black Box Warnings: The drug carries several black box warnings from regulatory agencies like the U.S. Food and Drug Administration (FDA), particularly regarding potential fetal harm and the risk of pancreatitis and liver damage. These warnings can influence prescribing decisions.
- Patent Expiries and Generic Competition: The loss of patent exclusivity for branded divalproex sodium has intensified price competition from generic manufacturers, limiting revenue growth for originator products.
What are the Future Sales Projections for Divalproex Sodium?
The sales trajectory for divalproex sodium is projected to be stable with modest growth, largely driven by the consistent demand from its established indications and the cost-effectiveness of generic formulations.
Projected Market Growth:
- 2024-2028 CAGR: The market is forecast to grow at a Compound Annual Growth Rate (CAGR) of approximately 2.5% to 3.0% during the period 2024-2028.
- Projected Market Size (2028): The global market is expected to reach approximately USD 1.4 billion by 2028.
Factors Influencing Future Sales:
- Continued Use in Developing Markets: As healthcare infrastructure improves in emerging economies, the demand for established, affordable treatments like divalproex sodium is likely to increase.
- Management of Chronic Conditions: Epilepsy and bipolar disorder are chronic conditions requiring long-term management, ensuring a steady patient base for divalproex sodium.
- Off-Label Uses and Research: While the primary indications drive current sales, ongoing research into potential off-label uses or novel delivery mechanisms could introduce new growth avenues, though this is speculative.
- Impact of Biosimil Competition: The concept of biosimilars is not directly applicable to small-molecule drugs like divalproex sodium, but the competitive pressure from generic equivalents will remain a defining feature.
Regional Growth Projections:
- Asia-Pacific: Expected to exhibit the highest growth rate (estimated 4-5% CAGR) due to expanding healthcare access and increasing diagnosis rates.
- North America and Europe: Predicted to maintain stable demand, with growth rates aligning with the overall market CAGR.
What is the Patent Landscape for Divalproex Sodium?
The original patents for divalproex sodium (Depakote®) have long expired. This has paved the way for widespread generic manufacturing and intense market competition.
Key Patent Milestones:
- Original Composition of Matter Patents: Expired in the early 2000s.
- Formulation and Manufacturing Patents: Various secondary patents related to specific formulations (e.g., delayed-release, extended-release) and manufacturing processes have also expired or have been successfully challenged by generic companies.
- Patent Litigation: Divalproex sodium has been a subject of extensive patent litigation, particularly concerning its extended-release formulations. Generic manufacturers have successfully demonstrated non-infringement or the invalidity of certain patents, leading to earlier market entry.
Current Patent Status:
- The divalproex sodium market is characterized by a lack of strong, unexpired patents that could significantly impede generic competition or create new barriers to entry.
- Any new patent filings would likely pertain to novel delivery systems, new combination therapies, or specific therapeutic uses, rather than the core divalproex molecule itself.
What is the Regulatory Status and Clinical Pipeline?
Divalproex sodium is approved by major regulatory bodies worldwide. Its clinical pipeline is largely focused on post-market surveillance and understanding long-term effects.
Regulatory Approvals:
- U.S. Food and Drug Administration (FDA): Approved for epilepsy, acute manic and mixed episodes associated with bipolar I disorder, and migraine prophylaxis.
- European Medicines Agency (EMA): Approved for similar indications.
- Other National Regulatory Authorities: Approved in numerous countries globally.
Black Box Warnings and Precautions:
- Hepatotoxicity: Risk of severe liver damage, particularly in young children and patients with mitochondrial disorders.
- Teratogenicity: Divalproex is a known human teratogen, causing significant birth defects and developmental disorders if taken during pregnancy. This is a major contraindication.
- Pancreatitis: A risk of potentially life-threatening pancreatitis exists.
- Hepatitis and Pancreatitis Warnings: FDA requires specific warnings regarding these risks.
Clinical Pipeline and Research:
- The clinical pipeline for divalproex sodium is not characterized by new drug development efforts for novel indications. Instead, ongoing research typically focuses on:
- Pharmacovigilance: Monitoring long-term safety and efficacy in real-world settings.
- Comparative Effectiveness Studies: Evaluating divalproex sodium against newer treatment alternatives.
- Understanding Mechanisms of Action: Further research into how divalproex exerts its therapeutic effects.
- Pediatric and Adolescent Use: Studies to refine dosing and monitor outcomes in younger populations.
The focus is on optimizing the use of the existing drug rather than expanding its approved indications significantly.
Key Takeaways
Divalproex sodium remains a significant pharmaceutical product with an estimated global market value of USD 1.2 billion in 2023. The market is dominated by generic competition following patent expiries, leading to price-sensitive dynamics. Key drivers include the high prevalence of epilepsy and bipolar disorder, alongside the drug's established efficacy and cost-effectiveness. Market restraints stem from potential side effects, black box warnings, and the emergence of newer therapeutic alternatives. Future sales are projected to grow at a modest CAGR of 2.5-3.0%, reaching approximately USD 1.4 billion by 2028, with the Asia-Pacific region expected to show the highest growth. The patent landscape is characterized by expired core patents, ensuring continued generic access. Regulatory oversight emphasizes strict warnings, particularly regarding teratogenicity and hepatotoxicity. The clinical pipeline is focused on post-market surveillance and comparative research rather than novel indications.
Frequently Asked Questions
-
What is the primary therapeutic advantage of divalproex sodium over newer antiepileptic drugs? Divalproex sodium's primary therapeutic advantage lies in its established efficacy across a broad spectrum of seizure types and its dual action as both an antiepileptic and a mood stabilizer, offering a cost-effective, single-agent solution for patients with comorbid conditions.
-
How significant is the risk of teratogenicity associated with divalproex sodium, and what are the current recommendations for pregnant patients? Divalproex sodium is a known human teratogen with a significant risk of causing major congenital malformations and neurodevelopmental disorders, particularly if taken during the first trimester. Current recommendations strongly advise against its use in pregnant women unless absolutely necessary, and only when alternative treatments are ineffective or not tolerated. If use is unavoidable, the lowest effective dose should be prescribed, and folic acid supplementation is recommended [2].
-
What is the competitive impact of multiple generic manufacturers on the pricing of divalproex sodium? The presence of numerous generic manufacturers has resulted in substantial price erosion for divalproex sodium. Competition is intense, with generic prices typically being significantly lower than the originator brand, driving cost-effectiveness as a key market determinant.
-
Are there any ongoing clinical trials exploring novel indications or delivery methods for divalproex sodium? While extensive research continues on understanding divalproex sodium's pharmacology and long-term effects, the pipeline for novel indications or significantly improved delivery methods is limited. Most current clinical research focuses on pharmacovigilance, comparative effectiveness, and optimizing its use within its established therapeutic areas.
-
How do the black box warnings for hepatotoxicity and pancreatitis influence prescribing patterns for divalproex sodium? The black box warnings for hepatotoxicity and pancreatitis necessitate careful patient selection and ongoing monitoring. Healthcare providers are required to weigh the benefits against these significant risks, particularly in vulnerable populations like young children and individuals with pre-existing liver or pancreatic conditions. This scrutiny can lead to a preference for alternative treatments when suitable options exist.
Citations
[1] World Health Organization. (n.d.). Epilepsy. Retrieved from [WHO website] [2] U.S. Food and Drug Administration. (2013). FDA Drug Safety Communication: Valproate products and pregnancy. Retrieved from [FDA website]
More… ↓
