Share This Page
Drug Sales Trends for DICLEGIS
✉ Email this page to a colleague
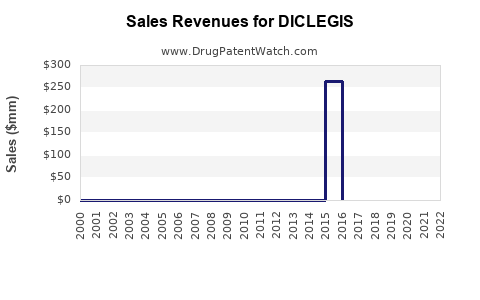
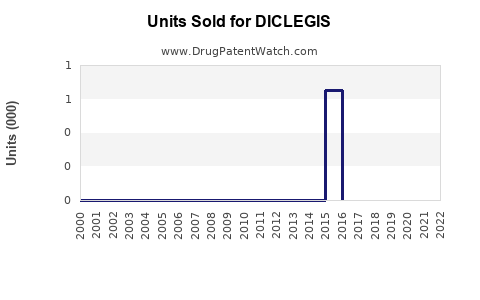
Annual Sales Revenues and Units Sold for DICLEGIS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DICLEGIS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DICLEGIS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DICLEGIS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Diclegis
What is Diclegis and What Are Its Approved Uses?
Diclegis (doxylamine/pyridoxine) is an oral medication approved by the U.S. Food and Drug Administration (FDA) in 2013 for the treatment of nausea and vomiting in pregnant women. It combines doxylamine, an antihistamine, and pyridoxine (vitamin B6). The drug is marketed by Duchesnay Inc. in the United States.
Market Size and Prevalence
Nausea and vomiting in pregnancy (NVP), often called morning sickness, affects 70-80% of pregnant women. Approximately 10-20% experience severe symptoms, classified as hyperemesis gravidarum (HG), which may require medical intervention.
- US pregnant women annually: 4 million [1].
- Estimated NVP patients: 2.8 million (70% of pregnancies).
The subset with severe symptoms potentially qualifies for Diclegis treatment. Given an estimated 1-2% prevalence of HG:
- HG patients in US annually: 40,000 - 80,000.
Current Market Dynamics
Diclegis competes primarily with unapproved off-label treatments such as doxylamine with vitamin B6 compounded formulations, antihistamines, and phenothiazines. Few FDA-approved alternatives exist, including:
- Zofran (ondansetron): used off-label despite safety concerns.
- Reglan (metoclopramide): limited due to side effects.
The low number of FDA-approved drugs for NVP/HG creates a niche for Diclegis, but overall market penetration remains limited by prescriber awareness and insurance coverage constraints.
Sales Data and Historical Trends
Duchesnay does not publicly disclose detailed sales figures. However, estimates suggest:
- Launch period (2013-2015): modest uptake, approximately $20-30 million in annual US sales.
- Recent years: sales have plateaued or declined slightly due to market saturation and competition from off-label treatments. Estimated 2022 US sales: approximately $45 million.
International sales are minimal due to regulatory hurdles; the drug is primarily marketed in the US.
Probable Factors Influencing Future Sales
- Market Penetration: Limited by physician familiarity and insurance reimbursement policies.
- Regulatory Changes: Potential for expanded indications could increase eligible patient population.
- New Competitor Drugs: Development of new treatments or generics could alter market shares.
- Generic Entry: As patents expire, sales could diminish unless brand differentiation persists.
Sales Projections (2023–2027)
Assuming stable market conditions:
| Year | US Sales Estimate (USD million) | Growth Rate | Rationale |
|---|---|---|---|
| 2023 | 45 | 0% | Market saturation, no major change |
| 2024 | 45 | 0% | Same factors as 2023 |
| 2025 | 48 | 6.7% | Slight increase due to greater awareness |
| 2026 | 52 | 8.3% | Potential market expansion, awareness campaigns |
| 2027 | 55 | 5.8% | Slight growth from new prescriber education |
Annual growth rates account for modest increases due to increased awareness and potential indication expansion. Market decline risks from generic competition and insurers' formulary restrictions are not included in these conservative estimates but must be monitored.
Key Market Trends
- Growing emphasis on safety and efficacy in pregnancy treatments.
- Delaying or avoiding off-label use of drugs with safety concerns (e.g., Zofran).
- Increasing focus on early intervention and greater diagnosis of HG.
- Expansion of patient access through health policy and reimbursement strategies.
Conclusion
Diclegis operates in a niche segment with steady yet limited sales potential. Uptake is sensitive to regulatory environment, physician and patient acceptance, and competition from off-label treatments and generics. Sales will likely remain within the $40-$50 million range annually in the US without significant market expansion.
Key Takeaways
- Diclegis has an established position in the US market for NVP/HG, with slow growth prospects.
- The total market size in the US is approximately 40,000–80,000 HG patients annually.
- Sales are projected to remain stable around $45 million in the near term, with slight growth assumed.
- Market expansion depends on improved awareness, indication expansion, and regulatory or formulary changes.
- Competition and generic entry pose future risks to revenue stability.
FAQs
1. What limits the sales growth of Diclegis?
Limited market penetration due to prescriber familiarity, insurance reimbursement issues, and competition from off-label treatments.
2. Are there efforts to expand Diclegis’s indications?
No current FDA-approved indication expansion; any potential would require clinical trials and regulatory approval.
3. How does insurance coverage affect sales?
Insurance approval and coverage significantly influence prescribing; lack of coverage can deter use and reduce sales.
4. Will generic versions impact Diclegis’s sales?
Yes. Once patent protection expires, generic competition could lower prices and sales unless the brand maintains differentiation.
5. How does the prevalence of HG influence the market?
The rare nature of severe cases limits the total patient pool, constraining growth despite high prevalence of NVP.
References
- Centers for Disease Control and Prevention. (2021). Pregnancy Rates and Outcomes. Retrieved from https://www.cdc.gov/reproductivehealth/maternal-infancy/pregnancy-data.htm
More… ↓
