Last updated: February 15, 2026
Market Analysis and Sales Projections for Dextroamphetamine
Market Overview
Dextroamphetamine is a central nervous system stimulant approved primarily for Attention Deficit Hyperactivity Disorder (ADHD) and Narcolepsy. It functions by increasing dopamine and norepinephrine activity, improving focus and alertness. The drug belongs to the amphetamine class, with formulations under brand names like Dexedrine, Vyvanse (as a prodrug), and generics.
The global ADHD drug market reached approximately $16 billion in 2022, growing at a compound annual growth rate (CAGR) of 8-10% over the past five years. The stimulant segment accounts for roughly 60% of this market, with methylphenidate and amphetamines competing intensely. Dextroamphetamine formulations constitute an estimated 25-30% of the stimulant market share.
Regulatory Landscape
In the U.S., the FDA classifies dextroamphetamine as a Schedule II controlled substance, which imposes restrictions on prescribing, dispensing, and manufacturing. Similar classifications apply in the EU and other major jurisdictions, complicating international distribution. Recent regulatory scrutiny over abuse potential has prompted tighter restrictions, potentially affecting sales volume growth.
Competitive Environment
Leading companies:
- Eli Lilly (Vyvanse)
- Eli Lilly's generic partners
- Teva Pharmaceuticals (generic dextroamphetamine)
- Mallinckrodt
Vyvanse dominates the prescription market, with projected sales exceeding $4 billion globally in 2022. Generics have gained ground as patents expire, leading to price competition and wider accessibility.
Sales Projections
Short-term (Next 1-2 Years):
- Limited sales growth for newly marketed formulations due to regulatory constraints.
- Existing generics expected to account for 15-20% of stimulant sales.
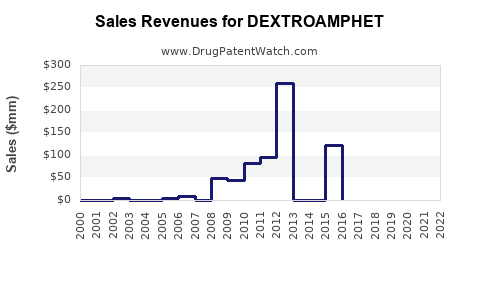
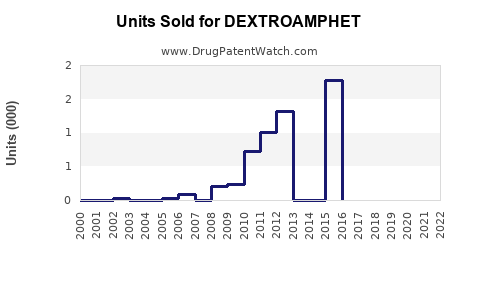
- Estimated U.S. sales of dextroamphetamine at approximately $0.7 billion in 2023, with an annual growth rate of 3-5%, driven by increased diagnosis rates and prescription rates.
Medium-term (3-5 Years):
- Introduction of extended-release and novel formulations can boost market penetration.
- Potential market expansion in emerging markets, with CAGR forecasted at 6-8%, contingent upon regulatory approval and healthcare infrastructure development.
- Overall sales forecast to reach $1.2 billion by 2026, assuming stable regulatory conditions.
Long-term (5+ Years):
- Market growth may slow due to regulatory restrictions and the emergence of non-stimulant treatments.
- Possible slight decline in market share for stimulant medications as non-stimulant therapies gain favor in certain patient groups.
Factors Influencing Sales
- Regulatory action: Tight controls on prescribing patterns could lower sales volume.
- Prescribing trends: Increased recognition of ADHD in adults and children sustains demand.
- Competitive innovations: New formulations with improved safety profiles may enhance market share.
- Healthcare policies: Reimbursement policies and insurance coverage influence accessibility.
Risks and Challenges
- Regulatory restrictions may limit prescribing and distribution.
- The abuse potential could lead to legal and societal restrictions.
- Competition from non-stimulant drugs like atomoxetine and guanfacine.
- Patent challenges and the rise of generics could put pressure on pricing.
Summary Table
| Aspect |
Current Status |
Projected 2-Year Outlook |
Key Drivers |
| Global Market Size |
~$16 billion (2022) |
Slight growth to ~$17.5 billion |
Increased diagnosis, prescription rates |
| Dextroamphetamine Market |
25-30% share of stimulant segment |
Stable, with potential for growth |
Generic expansion, new formulations |
| 2023 U.S. Sales |
~$0.7 billion |
3-5% growth |
Prescriber rates, approval of new formulations |
| Emerging Markets |
Limited penetration |
Growth at 6-8% CAGR |
Healthcare infrastructure development |
Key Takeaways
- Dextroamphetamine remains a significant component of the stimulant ADHD market.
- Revenue depends heavily on regulatory frameworks, prescription trends, and competition from generics.
- Growth is steady but limited in the short-term by regulatory restrictions.
- Long-term potential exists with formulation innovation and broader global adoption.
- Developer strategies must consider regulatory compliance, abuse potential, and market competition.
FAQs
1. What is the primary therapeutic indication for dextroamphetamine?
Treatment for ADHD and Narcolepsy.
2. How does regulatory status impact the market potential of dextroamphetamine?
As a Schedule II controlled substance, regulations restrict prescribing, dispensing, and manufacturing, which constrains sales growth.
3. What are the main competitors to dextroamphetamine?
Methylphenidate-based medications and non-stimulant options like atomoxetine.
4. How can future formulations influence sales?
Extended-release and abuse-deterrent formulations can enhance compliance and safety, potentially expanding market share.
5. What are the key risks to sales growth?
Regulatory restrictions, abuse concerns, competition from generics and non-stimulants, and societal controls on controlled substances.
References
- IQVIA. "Global ADHD Drugs Market Report," 2022.
- FDA. "Controlled Substance Schedules," 2023.
- MarketWatch. "Stimulant Market Analysis," 2022.
- Statista. "Prescription Trends for ADHD Medications," 2022.
- EvaluatePharma. "Pharmaceutical Sales Forecasts," 2023.