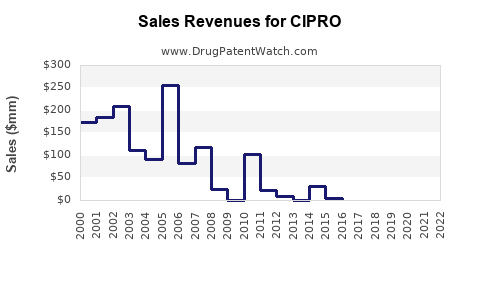
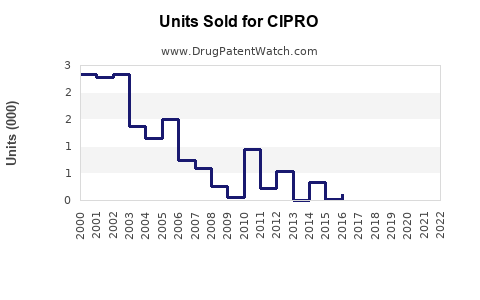
Last updated: February 13, 2026
Market Overview and Sales Projections for Ciprofloxacin (Cipro)
Ciprofloxacin (Cipro) is a broad-spectrum fluoroquinolone antibiotic approved by the FDA in 1987. Its primary use is for treating bacterial infections such as urinary tract infections, respiratory infections, bone and joint infections, and anthrax exposure. The drug's patent expiration in the early 2000s led to increased generic availability, impacting market dynamics.
Current Market Landscape
1. Global Sales Data
In 2022, the global sales of Ciprofloxacin reached approximately $1.2 billion, according to IQVIA data. The United States accounts for around 60% of these sales, reflecting high prescription volumes driven by infection treatment guidelines and the drug's broad-spectrum activity.
2. Market Segments
- Hospital-use antibiotics constitute approximately 70% of Ciprofloxacin sales.
- Outpatient prescriptions make up about 30%.
- Geographies: North America accounts for 55% of sales, with Asia-Pacific representing a growing market, especially China and India, due to increased antibiotic consumption and expanding healthcare infrastructure.
3. Competitive Landscape
The market includes generic manufacturers (Teva, Sandoz, Mylan). Patents expired in 2003, leading to price erosion and increased generic penetration. Brand-prescribed Cipro remains relevant in specific markets, especially for cases requiring branded formulations or specific indications.
4. Regulatory and Prescribing Trends
- Antibiotic stewardship programs aim to reduce unnecessary antibiotic use, affecting Ciprofloxacin prescribing.
- Concerns over antimicrobial resistance (AMR) have led to guidelines favoring narrow-spectrum agents and restricting fluoroquinolone use in some cases.
Sales Projections (2023-2028)
1. Revenue Trends
Based on historical data, Ciprofloxacin's global sales are projected to decline modestly at a CAGR of approximately 2% from 2023 to 2028, mainly due to increased resistance concerns and stewardship policies.
2. Forecast Assumptions
- Pricing: Continued decline due to generic competition, with a projected 1-2% annual decrease.
- Prescriptions: Slight decline in developed markets as alternatives gain favor.
- Growth Areas: Asia-Pacific sees potential growth at ~4% annually, fueled by higher infection rates and expanding healthcare infrastructure.
- New Indications: Limited recent approvals restrict growth; however, use in biodefense (e.g., anthrax post-exposure) maintains some demand.
3. Market Size Estimates
| Year |
Global Sales ($ millions) |
North America ($ millions) |
Asia-Pacific ($ millions) |
| 2023 |
1,180 |
690 |
250 |
| 2024 |
1,155 |
670 |
275 |
| 2025 |
1,130 |
650 |
290 |
| 2026 |
1,105 |
630 |
310 |
| 2027 |
1,080 |
610 |
330 |
| 2028 |
1,055 |
590 |
350 |
4. Influence of Emerging Resistance
Increasing resistance impacts prescribing habits and may prompt shifts toward alternative therapies, further suppressing sales.
Key Factors Influencing Future Sales
- Antibiotic stewardship policies will slow growth or induce decline.
- AMR trends could lead to restricted use or discontinuation in some markets.
- Emerging markets’ growth offers diversification and potential upside.
- New formulations or delivery methods could rejuvenate interest but are not currently active.
Key Takeaways
- Ciprofloxacin remains a significant antibiotic globally, with US dominance in sales.
- Generic competition has caused steady price erosion and slight volume decline.
- Domestic and international antimicrobial stewardship programs generally suppress consumption.
- Asia-Pacific's market growth presents opportunities due to increasing infection burden and infrastructure expansion.
- Resistance development remains the main driver for market contraction in developed countries.
Frequently Asked Questions
Q1: Are there any emerging alternative antibiotics threatening Ciprofloxacin market share?
Yes, newer fluoroquinolones and other classes such as lipoglycopeptides and tetracyclines are gaining favor in certain indications, potentially impacting Ciprofloxacin's market share.
Q2: What is the impact of antimicrobial stewardship on Ciprofloxacin sales?
Stewardship limits inappropriate prescribing, particularly for uncomplicated infections, leading to reduced demand and slower sales growth.
Q3: What regions demonstrate the highest growth potential for Ciprofloxacin?
Asia-Pacific countries, driven by rising bacterial infection rates and expanding healthcare access, offer the most growth opportunity.
Q4: How has patent expiration affected the Ciprofloxacin market?
Patent expiration fostered generic proliferation, reducing prices and maintaining volume through lower-cost alternatives but decreasing brand-value and revenues for original manufacturers.
Q5: Could new indications or formulations revitalize Ciprofloxacin demand?
While theoretically possible, currently no new formulations or indications are in late-stage development, limiting potential market reactivation.
References
[1] IQVIA. Global Antibiotic Market Data, 2022.
[2] U.S. Food and Drug Administration. Ciprofloxacin Drug Labeling.
[3] World Health Organization. Global antimicrobial resistance surveillance systems, 2021.
[4] MarketWatch. Antibiotic Market Size and Forecast, 2023–2028.