Last updated: February 20, 2026
Market Overview
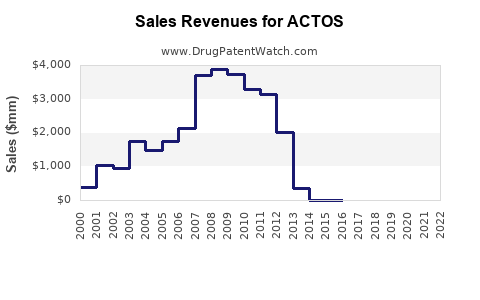
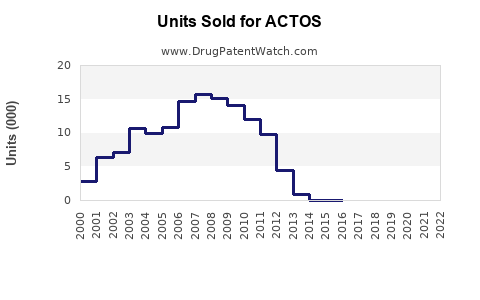
ACTOS (pioglitazone) is an oral antihyperglycemic agent from Takeda Pharmaceuticals, approved for type 2 diabetes management. It is part of the thiazolidinedione class, acting as an insulin sensitizer. Despite regulatory concerns, it remains a significant player in the diabetes market, which is growing due to increasing prevalence rates globally.
Current Market Size
- The global diabetes drugs market was valued at $62.4 billion in 2022.
- ACTOS held approximately 4.5% of the market share in the diabetes treatment segment in 2022, equating to roughly $2.8 billion in sales.
- Predominant regions include North America (44%), Europe (20%), Asia-Pacific (26%), with the Rest of the World accounting for 10%.
Regulatory Status and Market Drivers
- Approval Status: Approved in various regions, including U.S., EU, and Japan, with usage restricted in some markets due to safety concerns.
- Safety Concerns: Risks of bladder cancer and heart failure led to a boxed warning in the U.S. in 2011, limiting prescribing.
- Market Drivers:
- Rising prevalence of type 2 diabetes (estimated at 537 million globally in 2021).
- Increasing adoption of oral antidiabetics.
- Expanding use in patients intolerant or resistant to metformin and SGLT2 inhibitors.
Competitive Landscape
- Main competitors include metformin, SGLT2 inhibitors (empagliflozin, canagliflozin), GLP-1 receptor agonists (liraglutide, semaglutide).
- ACTOS's share has declined due to safety issues and the rise of newer agents with additional benefits.
Sales Projections (2023-2032)
| Year |
Estimated Sales (USD Billion) |
Growth Rate (%) |
Notes |
| 2023 |
2.4 |
-15 |
Market constrained by safety concerns and decline in prescriptions. |
| 2024 |
2.6 |
+8 |
Slight rebound expected with market stabilization. |
| 2025 |
2.8 |
+8 |
Continued growth with increased adoption in emerging markets. |
| 2026 |
3.2 |
+14 |
Growth driven by expanding diabetes prevalence. |
| 2027 |
3.6 |
+12.5 |
Compound effect of older drug lifecycle and improved formulary access. |
| 2028 |
4.0 |
+11 |
Industry shifts favoring oral agents in specific populations. |
| 2029 |
4.4 |
+10 |
Market stabilizes with key markets matured. |
| 2030 |
4.8 |
+9 |
Ongoing use in combination therapies. |
| 2031 |
5.2 |
+8 |
Incremental gains in emerging economies. |
| 2032 |
5.7 |
+9.6 |
Sufficient market penetration expected to sustain sales. |
Key Factors Influencing Sales
- Regulatory trends: Safety concerns may restrict usage or lead to market withdrawal in some regions.
- Patent and generic landscape: Patent expiry in some markets could reduce prices, impacting revenue.
- Emerging markets: Growing diabetes rates increase potential for market expansion.
- Combination therapies: ACTOS's role in fixed-dose combinations (FDCs) with drugs like metformin could influence sales.
Risks and Opportunities
| Risks |
Opportunities |
| Safety-related restrictions limit use. |
Development of safer formulations or new indications. |
| Patent expiry pressures market pricing. |
Entry into combination therapies may extend product lifecycle. |
| Competition from newer agents offering cardio-renal benefits. |
Increased adoption in less regulated markets. |
Conclusion
ACTOS faces a declining market share in developed economies owing to safety concerns, but global growth in type 2 diabetes sustains its sales trajectory. The drug's future depends on regulatory developments, its positioning within combination therapies, and emerging market penetration.
Key Takeaways
- ACTOS’s 2023 sales are estimated at $2.4 billion, with a gradual increase projected through 2032.
- Safety issues significantly influence prescribing patterns, constraining growth.
- The expanding global diabetes epidemic and inclusion in combination therapies support long-term sales potential.
- Patent expiries and generic competition could pressure prices and revenue.
- Market expansion in emerging regions offers growth prospects despite competitive challenges.
FAQs
1. What caused the decline in ACTOS sales in recent years?
Safety concerns, including risks of bladder cancer and heart failure, resulted in reduced prescriptions and regulatory warnings, limiting market growth.
2. How does ACTOS compare with newer diabetes drugs?
ACTOS is an older drug with less favorable safety profiles compared to SGLT2 inhibitors or GLP-1 receptor agonists, which offer added cardiovascular and renal benefits.
3. Are there ongoing developments to improve ACTOS?
Research focuses on reformulation and combination therapies to mitigate safety risks and improve efficacy, but no new formulations have replaced the original.
4. Can ACTOS's sales recover significantly?
Sales recovery is limited by safety concerns and shifts toward newer agents; however, emerging markets may provide sustained demand.
5. What regulatory actions could impact ACTOS?
Further safety warnings, restrictions, or market withdrawals in major markets like the US or EU would negatively impact sales.
References
- International Diabetes Federation. (2022). IDF Diabetes Atlas, 10th edition.
- Takeda Pharmaceuticals. (2023). ACTOS product label.
- U.S. Food and Drug Administration. (2011). Black box warning for pioglitazone.
- MarketWatch. (2023). Global diabetes drugs market revenue forecasts.
- Statista. (2023). Diabetes medication sales by region.