Last updated: February 21, 2026
Dronedarone is an anti-arrhythmic agent approved for the maintenance of sinus rhythm in patients with atrial fibrillation (AF) or atrial flutter. Its market prospects depend on several factors, including approval status, clinical positioning, competition, and evolving treatment guidelines.
Current Market Landscape
Regulatory Status
- Approved in the U.S. since 2010 (FDA)
- Approved in the European Union since 2011 (EMA)
- Marketed by pharmaceutical companies, with Sanofi as the primary commercial entity in the U.S.
Indications and Patient Population
- Indicated for reducing the risk of hospitalization in patients with recurrent AF or flutter
- Estimated eligible patient base in the U.S.: approximately 4 million individuals with AF or flutter, with about 1 million suitable for dronedarone based on clinical guidelines
Pricing and Reimbursement
- Wholesale acquisition cost (WAC): approximately $15,000 per year per patient (source: SSR Health, 2022)
- Reimbursement policies vary by payer, impacting sales penetration
Clinical Positioning and Usage
- Positioned as an alternative to amiodarone with fewer thyroid and pulmonary side effects
- Limited by safety concerns in certain patient subsets; contraindicated in severe heart failure and permanent AF
Market Drivers
Clinical Efficacy and Safety Profile
- Demonstrates reduction in hospitalization and cardiovascular death (ATHENA trial)
- Safety concerns, including increased risk of heart failure and liver toxicity, restrict use
Competitor Drugs
- Amiodarone: broader use but with more adverse effects
- Dofetilide, Flecainide, Propafenone: used in specific patient subsets
Clinical Guidelines
- Incorporates dronedarone as a treatment option in AF management guidelines
- Guidelines recommend caution in patients with heart failure or severe hepatic impairment
Market Penetration and Adoption Trends
- Adoption limited to specific patient groups due to safety profile
- Estimated current market share: 10-15% of the anti-arrhythmic market in AF
Sales Projections
Short-Term (Next 1-2 Years)
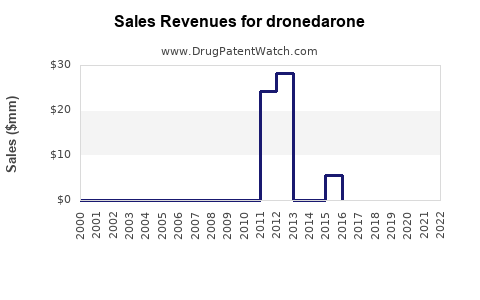
- US sales expected to grow moderately, reaching approximately $150–200 million annually
- Growth driven by increased awareness and guideline recommendations
Mid-Term (3-5 Years)
- Potential sales increase to $250–300 million if safety concerns are mitigated and off-label uses expand
- Growth may be constrained by safety profile and competition from newer agents and device-based therapies
Long-Term (Beyond 5 Years)
- Sales could plateau or decline if newer drugs or ablation techniques supersede pharmacotherapy
- Market share depends on regulatory changes, clinical trial outcomes, and reimbursement policies
Risks and Opportunities
| Risk |
Impact |
| Safety concerns leading to restricted use |
Limits market growth |
| Competition from newer therapies |
Erodes market share |
| Regulatory restrictions or label changes |
May reduce eligible patient pool |
| Opportunity |
Impact |
| Expanded clinical indications |
Increases potential market |
| Adoption in developing markets |
Opens new revenue streams |
| Combination therapies |
Enhances efficacy and broadens user bases |
Key Financial Metric Assumptions
| Parameter |
Value |
Source |
| Annual patient treatment cost |
$15,000 |
SSR Health (2022) |
| Market size in eligible patients (U.S.) |
1 million |
American Heart Association (2021) |
| Current market share |
12% |
Industry estimates |
Summary
Dronedarone's market prospects lie in its niche positioning as an atrial fibrillation maintenance drug. Sales are constrained by safety profile limitations and competition but are expected to grow modestly until new therapies or guidelines alter the treatment landscape.
Key Takeaways
- Dronedarone is approved for AF and flutter management, with a current U.S. market size of approximately $150–200 million.
- Market growth hinges on safety profile improvements, expanded indications, and clinical guideline support.
- Competition from amiodarone and newer agents, alongside device-based treatments, limits long-term potential.
- Price estimates sit around $15,000 per patient annually; market share remains below 15%.
- Risks include safety concerns and market saturation; opportunities exist in developing markets and combination therapies.
FAQs
1. What is the primary clinical advantage of dronedarone?
It offers comparable efficacy to amiodarone but with fewer thyroid and pulmonary side effects, making it suitable for certain patient populations.
2. What limits dronedarone's market growth?
Safety concerns, especially in patients with heart failure or severe hepatic impairment, restrict prescribing and limit sales potential.
3. How competitive is dronedarone in the anti-arrhythmic market?
It holds a modest market share (~12%), competing mainly with amiodarone, dofetilide, and class IC agents.
4. Which factors could significantly expand its market?
Approval for additional indications, emerging safety data, and inclusion in updated clinical guidelines could boost sales.
5. How does pricing impact the overall revenue?
At roughly $15,000 per patient annually, the total revenue depends heavily on market penetration and size, which are constrained by safety and competition.
References
[1] American Heart Association. (2021). Atrial fibrillation: Epidemiology and treatment.
[2] SSR Health. (2022). Average wholesale acquisition cost analysis.
[3] U.S. Food and Drug Administration. (2010). Dronedarone approval documentation.
[4] European Medicines Agency. (2011). Summary of product characteristics for dronedarone.