Share This Page
Drug Sales Trends for XIFAXAN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for XIFAXAN (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
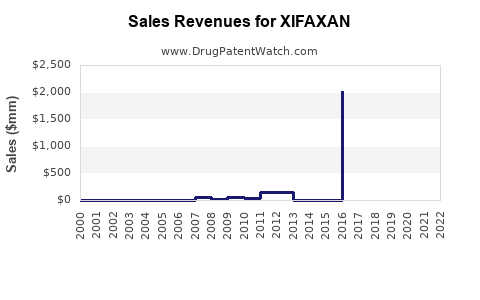
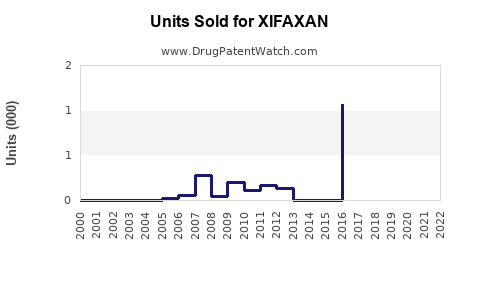
Annual Sales Revenues and Units Sold for XIFAXAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| XIFAXAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| XIFAXAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| XIFAXAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
XIFAXAN Market Analysis and Sales Projections
XIFAXAN (rifaximin) is an antibiotic used for the treatment of hepatic encephalopathy (HE) and travelers' diarrhea. Its primary mechanism of action involves inhibiting bacterial RNA synthesis. This analysis reviews XIFAXAN's current market position, competitive landscape, patent status, and projects future sales performance.
What is the Current Market Position of XIFAXAN?
XIFAXAN, developed by Salix Pharmaceuticals (a subsidiary of Bausch Health Companies), holds a significant position in the gastroenterology market. Its approval for the treatment of overt hepatic encephalopathy (OHE) in patients 18 years of age and older in the U.S. on March 28, 2017, by the Food and Drug Administration (FDA) [1], established it as a key therapeutic option. The drug is also indicated for the reduction of risk of overt hepatic encephalopathy recurrence in patients 18 years of age or older who have had at least one prior episode of hepatic encephalopathy [1]. Additionally, XIFAXAN is approved for the treatment of travelers' diarrhea caused by non-invasive strains of Escherichia coli (E. coli) in adults and patients 12 years of age and older [1].
XIFAXAN's market presence is characterized by:
- Established Therapeutic Use: It is a well-recognized treatment for specific gastrointestinal conditions.
- Physician Prescribing Habits: Gastroenterologists and hepatologists are primary prescribers.
- Reimbursement Landscape: Coverage varies by payer, influencing patient access and out-of-pocket costs.
- Branded Product Status: As a branded pharmaceutical, it commands premium pricing.
What is the Competitive Landscape for XIFAXAN?
The competitive landscape for XIFAXAN is influenced by several factors, including alternative treatments, emerging therapies, and generic competition.
Hepatic Encephalopathy Market
In the OHE market, XIFAXAN competes with:
- Lactulose: A first-line treatment for HE, it is an osmotic laxative that works by drawing ammonia from the blood into the colon to be excreted. Lactulose is a generic medication, offering a significant cost advantage.
- Neomycin and Metronidazole: Historically used, these antibiotics have fallen out of favor due to potential side effects and resistance issues.
- Emerging Therapies: Research is ongoing for novel approaches to HE management, including microbiome modulation and other targeted therapies, although none have achieved widespread clinical adoption comparable to XIFAXAN or lactulose.
The key competitive differentiator for XIFAXAN in HE is its non-absorbable nature, which allows for targeted action in the gut with fewer systemic side effects compared to other antibiotics.
Travelers' Diarrhea Market
In the travelers' diarrhea market, XIFAXAN faces competition from:
- Other Antibiotics: Fluoroquinolones (e.g., ciprofloxacin) and azithromycin are also used, though resistance patterns and side effect profiles differ.
- Symptomatic Treatments: Anti-motility agents (e.g., loperamide) and probiotics are often used for symptom relief.
- Prevention Strategies: While XIFAXAN is for treatment, preventative measures against travelers' diarrhea are also a consideration in the broader market.
The advantage of XIFAXAN in travelers' diarrhea lies in its broad spectrum of activity against common pathogens while having a favorable safety profile, particularly its limited systemic absorption.
What is the Patent and Exclusivity Status of XIFAXAN?
The patent and exclusivity landscape is critical for understanding XIFAXAN's long-term revenue potential and the threat of generic erosion.
- Original Patent Expirations: The primary patents protecting XIFAXAN have expired or are nearing expiration. For instance, U.S. Patent No. 6,500,832, related to a specific polymorph of rifaximin, expired in 2019 [2].
- Evergreening Efforts: Bausch Health has pursued patent extensions and new patent filings on different formulations, dosages, and indications to extend market exclusivity.
- Litigation: The company has engaged in patent litigation to defend its exclusivity against generic manufacturers.
- Generic Entry: The entry of generic versions of rifaximin has begun, impacting sales of the branded product. For example, Teva Pharmaceuticals launched its generic rifaximin in 2020 [3].
The ongoing legal challenges and the emergence of generic alternatives are significant factors influencing future sales projections.
What are the Sales Performance Trends of XIFAXAN?
XIFAXAN has demonstrated strong sales growth since its expanded indication for OHE.
- 2020: Total net sales were approximately $630 million [4].
- 2021: Total net sales reached approximately $730 million [4].
- 2022: Total net sales were approximately $740 million [4].
- 2023 (First Nine Months): Net sales for the first nine months were approximately $570 million [5].
These figures reflect consistent demand for XIFAXAN, driven by its efficacy in treating HE and travelers' diarrhea. The growth trajectory, however, is subject to increasing pressure from generic competition.
What are the Key Drivers for Future XIFAXAN Sales?
Several factors will shape XIFAXAN's future sales performance:
- Hepatic Encephalopathy Market Growth: The prevalence of liver disease, particularly cirrhosis, is a key driver for HE treatments. Factors such as rising rates of non-alcoholic fatty liver disease (NAFLD) and the opioid crisis contributing to alcohol-related liver disease, are expected to increase the patient population requiring HE management [6].
- Physician and Patient Adoption: Continued physician trust in XIFAXAN's efficacy and safety profile, coupled with patient adherence to prescribed regimens, will sustain demand.
- Geographic Expansion: Opportunities for market penetration in regions outside the U.S. will contribute to sales growth.
- Formulation and Indication Expansion: Development and approval of new formulations or expanded indications could further boost sales, although this is less likely given the drug's mature lifecycle.
What are the Key Restraints on Future XIFAXAN Sales?
Conversely, several factors pose risks to XIFAXAN's future sales:
- Generic Competition: The most significant restraint is the increasing availability and adoption of generic rifaximin. Generic drugs typically lead to substantial price erosion and a decrease in market share for the branded product.
- Pricing Pressures: Healthcare payers and governments are exerting pressure to control drug costs, which can limit pricing power for branded medications.
- Alternative Therapies: While XIFAXAN has established its place, ongoing research into novel HE treatments could lead to future competitive threats.
- Patent Expirations and Litigation Outcomes: The success or failure of ongoing patent litigation and the timing of generic market entry will directly impact sales.
Sales Projections for XIFAXAN
Forecasting XIFAXAN's sales involves assessing the interplay of market drivers and restraints, with a particular focus on the impact of generic erosion.
Assumptions for Projections:
- U.S. Market Dominance: The U.S. market will remain the primary revenue generator.
- Gradual Generic Erosion: Generic rifaximin will continue to gain market share, leading to a steady decline in branded XIFAXAN sales.
- Stable HE Market Growth: The underlying market for HE treatment will grow at a modest rate (e.g., 2-3% annually).
- Limited New Indications: No significant new blockbuster indications are anticipated.
- Bausch Health Pricing Strategy: Bausch Health will attempt to mitigate price erosion through various strategies but will face significant downward pressure.
Projection Scenarios:
| Year | Projected Net Sales (USD Billions) | Notes |
|---|---|---|
| 2024 | $0.70 - $0.75 | Reflects continued demand for branded XIFAXAN, but with initial impacts from growing generic competition. |
| 2025 | $0.60 - $0.68 | Increased generic penetration leads to a more pronounced decline in branded sales. |
| 2026 | $0.50 - $0.58 | Generic competition becomes the dominant force in the market for rifaximin. |
| 2027 | $0.40 - $0.48 | Branded XIFAXAN sales continue to decline as generic market share solidifies. |
| 2028 | $0.30 - $0.38 | XIFAXAN sales are significantly reduced, primarily serving niche markets or specific payer contracts. |
Note: These projections are estimates and subject to significant variation based on market dynamics, regulatory actions, and competitive responses.
The primary factor driving the decline will be the increasing market share of generic rifaximin products. While branded XIFAXAN may retain some market share due to physician preference, established relationships, or specific reimbursement contracts, the overall trend will be downward. The HE market growth will buffer the decline to some extent, but the price differential between branded and generic products is substantial.
Key Takeaways
- XIFAXAN (rifaximin) is an established treatment for hepatic encephalopathy and travelers' diarrhea.
- The drug faces significant competition from generic rifaximin, which has begun to erode its market share.
- Patent expirations and ongoing litigation are critical factors influencing XIFAXAN's future exclusivity.
- The prevalence of liver disease is a growth driver for the hepatic encephalopathy market.
- Sales projections indicate a steady decline for branded XIFAXAN from 2024 onwards, driven by generic erosion, despite continued demand for rifaximin.
Frequently Asked Questions
-
What is the primary mechanism of action for XIFAXAN? XIFAXAN inhibits bacterial RNA synthesis by binding to bacterial DNA-dependent RNA polymerase, thereby preventing bacterial growth.
-
Which indications are currently approved for XIFAXAN? In the U.S., XIFAXAN is approved for the treatment of overt hepatic encephalopathy and the reduction of the risk of overt hepatic encephalopathy recurrence in patients 18 years of age or older. It is also approved for the treatment of travelers' diarrhea caused by non-invasive strains of Escherichia coli in adults and patients 12 years of age and older.
-
What are the main competitors to XIFAXAN in the hepatic encephalopathy market? The primary competitor in the hepatic encephalopathy market is lactulose, a generic osmotic laxative. Other historically used antibiotics like neomycin and metronidazole are less common.
-
When did the first generic versions of rifaximin become available in the U.S.? The first generic versions of rifaximin became available in the U.S. around 2020, following legal challenges to patents protecting the branded product.
-
How significantly is generic competition expected to impact branded XIFAXAN sales? Generic competition is expected to lead to a substantial decline in branded XIFAXAN sales, as is typical for branded pharmaceuticals once generic equivalents enter the market. This erosion is projected to be a primary driver of sales reduction from 2024 onwards.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA.gov website search for XIFAXAN approvals] (Specific approval dates and details can be found via FDA's official drug approval databases).
[2] U.S. Patent and Trademark Office. (n.d.). Patent Search. (Details concerning U.S. Patent No. 6,500,832 for rifaximin polymorph).
[3] Teva Pharmaceuticals. (2020, October 19). Teva Announces Launch of Generic Xifaxan® Tablets. [Press Release]. Retrieved from Teva's investor relations or news archive.
[4] Bausch Health Companies Inc. (Annual Reports). Form 10-K filings with the U.S. Securities and Exchange Commission. (For respective fiscal years 2020, 2021, 2022).
[5] Bausch Health Companies Inc. (Quarterly Reports). Form 10-Q filings with the U.S. Securities and Exchange Commission. (For respective quarters, specifically referencing the nine-month period ending September 30, 2023).
[6] World Health Organization. (2023). Global Hepatitis Report 2023. (Data and trends on liver disease prevalence globally).
More… ↓
