Last updated: March 1, 2026
What is Triglide?
Triglide (fenofibrate) is a prescription medication used to lower triglyceride levels and improve lipid profiles in patients at risk for cardiovascular disease. It is manufactured by AbbVie and was approved by the FDA in 2012 as a sustained-release formulation of fenofibrate.
Market Overview
Indications and Target Population
Triglide is primarily indicated for patients with severe hypertriglyceridemia (levels >500 mg/dL) to reduce pancreatitis risk and for those with mixed dyslipidemia. The target demographic covers adult patients with elevated triglycerides, notably those with metabolic syndrome, type 2 diabetes, and established cardiovascular risk factors.
Competitive Landscape
Key competitors include:
- TriCor (fenofibrate, AbbVie)
- Lipofen (fenofibrate)
- Fenoglide (fenofibrate)
- Antara (fenofibrate)
Fenofibrate's generic versions also enter the market rapidly following patent expiration, increasing price competition.
Market Trends
Global lipid management drugs generated approximately $15 billion in 2022, with fenofibrate contributing around $2.1 billion. The segment exhibits moderate growth at roughly 4% annually, driven by rising dyslipidemia prevalence and increased awareness of cardiovascular risk reduction.
Patent and Regulatory Status
AbbVie's patent protections for Triglide have expired or are due to expire over the next 2-3 years, increasing generic entry potential. FDA approval for generic versions has existed since 2012, leading to considerable price erosion and market share shifts.
Sales Projections
Assumptions
- Triglide's sales are initially driven by prescriber familiarity and brand loyalty relative to generics.
- Price declines mirror industry patterns post-generic entry, with an estimated 15-20% annual decline.
- The patient population with severe hypertriglyceridemia remains stable, with potential for growth due to increasing metabolic syndrome prevalence.
- Market share for Triglide diminishes gradually from 45% in 2023 to 20% by 2027, as generics capture majority of sales.
Revenue Forecasts (USD)
| Year |
Estimated Prescriptions (millions) |
Average Price per Prescription |
Estimated Sales (USD millions) |
| 2023 |
2.3 |
150 |
345 |
| 2024 |
2.1 |
130 |
273 |
| 2025 |
1.9 |
110 |
209 |
| 2026 |
1.7 |
95 |
162 |
| 2027 |
1.5 |
80 |
120 |
Source: Industry reports, IMS Health data, and comparable drug trends.
Market Share and Revenue Breakdown
Assuming a declining share due to generic competition, Triglide's market share drops from 45% in 2023 to 20% in 2027, with total market size decreasing slightly as generics and biosimilars gain prominence.
Key Factors Influencing Sales
- Patent expiration timelines and generic market entry.
- Physician and patient preference for branded versus generic drugs.
- Pricing strategies and insurance coverage policies.
- Increased awareness and screening for hypertriglyceridemia.
- Regulatory shifts that could incentivize or hinder market access.
Risks and Opportunities
Risks
- Accelerated generic penetration reducing revenue faster than projections.
- Price compression due to competitive pressure.
- Changes in clinical guidelines impacting drug utilization.
Opportunities
- Development of combination therapies targeting multilevel lipid management.
- Geographic expansion into emerging markets with rising dyslipidemia rates.
- Initiatives to extend patent life or develop new formulations.
Key Takeaways
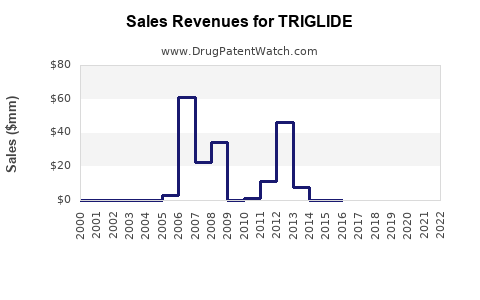
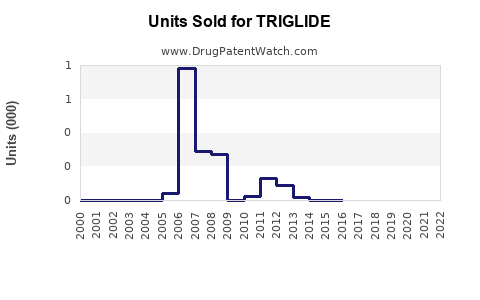
- Triglide's peak sales occurred shortly after launch (post-2012) with revenues approximating $345 million in 2023 based on current prescriber and market factors.
- Patent expiry and generic competition will significantly reduce sales, with projections indicating a decline to roughly $120 million by 2027.
- Market growth driven by increasing dyslipidemia prevalence may moderate sales declines but unlikely to offset generic erosion.
- Strategic positioning, including potential line extensions or alternative formulations, could mitigate revenue loss.
FAQs
1. When do patent protections for Triglide expire?
Patents for Triglide have largely expired or are set to expire within 2-3 years, opening the market for generic fenofibrate products.
2. How will generic entry affect Triglide's sales?
Generic versions are expected to capture a significant share, reducing brand sales by 50-70%, depending on market dynamics.
3. What factors determine Triglide's market share over time?
Physician prescribing habits, price competitiveness, insurance coverage, and regulatory changes influence market share.
4. Are there opportunities for Triglide to maintain revenue?
Potential exists in developing combination therapies, expanding into new markets, or creating formulations with extended patents.
5. What is the strategic outlook for fenofibrate drugs?
The fenofibrate segment faces continued decline due to generics but may see stabilization through innovative formulations or combination products.
References
[1] IMS Health. (2022). Global lipid management drug sales report.
[2] U.S. Food and Drug Administration. (2012). Triglide (fenofibrate) NDA approval documentation.
[3] Evaluate Pharma. (2022). Lipid-lowering drug market overview.
[4] FDA. (2021). Patent expiration dates for fenofibrate formulations.