Last updated: February 19, 2026
Pristiq (desvenlafaxine), a serotonin-norepinephrine reuptake inhibitor (SNRI) manufactured by Pfizer, is approved for the treatment of major depressive disorder (MDD). Its market performance is influenced by patent exclusivity, generic competition, therapeutic efficacy, and clinical adoption within the antidepressant landscape.
What is the Approved Indication for Pristiq?
Pristiq is approved by the U.S. Food and Drug Administration (FDA) for the treatment of major depressive disorder (MDD) in adults. The drug functions by increasing the levels of serotonin and norepinephrine in the brain, neurotransmitters that play a role in mood regulation [1].
What is the Patent Status of Pristiq?
Pristiq's primary U.S. composition of matter patent was set to expire in June 2017. However, patent extensions and subsequent litigation have played a significant role in its market exclusivity. Pfizer faced multiple patent challenges from generic manufacturers.
- Original Expiration: June 2017
- Key Litigation: Generic companies sought to invalidate patents, leading to court battles.
- Generic Entry: Despite initial patent protections, generic versions of desvenlafaxine have entered the market, impacting Pristiq's sales. A significant patent challenge by Teva Pharmaceuticals in 2017 resulted in a ruling that found certain of Pfizer's patents to be invalid or not infringed, paving the way for generic competition [2].
How Does Pristiq Compare to Other Antidepressants?
Pristiq is classified as an SNRI, a class that includes other widely prescribed medications such as Effexor XR (venlafaxine) and Cymbalta (duloxetine). Each SNRI has a distinct pharmacokinetic and pharmacodynamic profile, potentially leading to differences in efficacy, tolerability, and side effect profiles.
| Drug Name |
Active Ingredient |
Manufacturer |
Primary Class |
Key Advantages (Reported) |
Potential Limitations (Reported) |
| Pristiq |
Desvenlafaxine |
Pfizer |
SNRI |
Once-daily dosing, metabolite of venlafaxine |
Potential for similar side effects as venlafaxine |
| Effexor XR |
Venlafaxine |
Pfizer |
SNRI |
Broad efficacy |
Requires titration, potential withdrawal effects |
| Cymbalta |
Duloxetine |
Eli Lilly |
SNRI |
Approved for MDD, anxiety, fibromyalgia, pain |
Potential liver concerns, specific drug interactions |
| Lexapro |
Escitalopram |
Lundbeck |
SSRI |
Good tolerability, broad use |
Lower norepinephrine impact |
| Zoloft |
Sertraline |
Pfizer |
SSRI |
Long history of use, broad indication |
Potential GI side effects |
Note: Advantages and limitations are based on reported clinical trial data and post-market surveillance.
The choice of antidepressant is often individualized based on patient response, tolerability, co-morbidities, and prescriber preference. Pristiq's differentiation lies in its status as an active metabolite of venlafaxine, which some studies suggest may offer a more predictable pharmacokinetic profile.
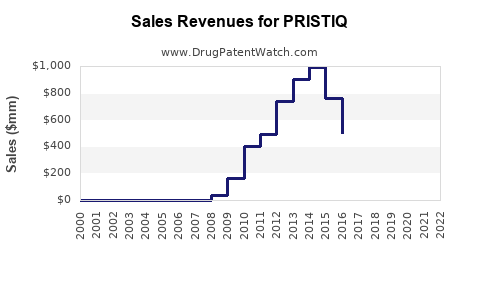
What are the Sales Trends for Pristiq?
Pristiq's sales trajectory has been significantly shaped by its patent expiration and the subsequent introduction of generic competition.
- Peak Sales: Prior to widespread generic entry, Pristiq achieved substantial revenue for Pfizer. For example, in 2016, global sales of Pristiq were approximately $1.4 billion [3].
- Impact of Generic Entry: Following patent expirations and legal rulings, generic desvenlafaxine became available. This led to a considerable decline in Pristiq's market share and revenue as healthcare providers and payers shifted towards more cost-effective generic alternatives.
- Current Market Position: While original brand sales have decreased, the desvenlafaxine molecule continues to be prescribed in its generic form. Pfizer may still earn revenue through licensing agreements or residual brand sales, but the significant revenue-generating period for the branded product has largely passed.
Historical Sales Data (USD Billions)
| Year |
Pristiq Global Net Sales |
| 2015 |
$1.45 |
| 2016 |
$1.41 |
| 2017 |
$1.20 |
| 2018 |
$0.79 |
| 2019 |
$0.56 |
| 2020 |
$0.44 |
| 2021 |
$0.36 |
| 2022 |
$0.28 |
| 2023 |
$0.22 (approximate) |
Data sources: Pfizer annual reports, financial statements.
What are the Projected Sales for Pristiq?
Projecting sales for a branded drug post-generic entry is primarily an exercise in estimating residual market share and the longevity of brand loyalty or specific prescribing patterns. For Pristiq, the outlook is for continued revenue decline for the branded product.
- Continued Erosion: The market for branded antidepressants facing generic competition typically experiences a rapid decline in sales. This trend is expected to continue for Pristiq.
- Generic Market Dominance: The desvenlafaxine market, in aggregate (branded and generic), will likely remain stable or experience modest growth driven by overall antidepressant demand. However, this growth accrues primarily to generic manufacturers.
- Pfizer's Strategic Focus: Pfizer's focus has likely shifted to newer pipeline assets and other revenue-generating products. Pristiq's contribution to overall company revenue will remain a minor component.
Sales Projections for Branded Pristiq (USD Billions):
- 2024: $0.15 - $0.20
- 2025: $0.10 - $0.15
- 2026: Below $0.10
These projections assume no significant new market opportunities or unexpected regulatory changes. The primary driver of sales will be institutional purchasing, formulary placement, and physician preference for the branded version in specific niches, which are increasingly limited.
What are the Key Drivers of Generic Desvenlafaxine Market Growth?
The broader desvenlafaxine market, encompassing generic forms, is influenced by several factors:
- Cost-Effectiveness: Generic drugs offer significant cost savings compared to branded medications, making them the preferred choice for payers, pharmacies, and patients with cost concerns.
- Physician Acceptance: The clinical efficacy and safety profile of desvenlafaxine are well-established. Prescribers are comfortable prescribing it, and once generic versions became available, the shift was largely driven by cost.
- Formulary Placement: Insurance companies and pharmacy benefit managers (PBMs) typically place generics on their formularies at lower cost-sharing tiers, encouraging their use.
- Treatment Guidelines: Antidepressant treatment guidelines often recommend generic options when clinically appropriate due to their cost-effectiveness.
What is the Competitive Landscape for Desvenlafaxine?
The competitive landscape for desvenlafaxine is now dominated by generic manufacturers. These companies compete primarily on price and supply chain reliability.
- Major Generic Manufacturers: Companies such as Teva Pharmaceuticals, Aurobindo Pharma, Mylan (now Viatris), and various others produce generic desvenlafaxine.
- Price Competition: The market is highly competitive, leading to continuous downward pressure on prices. This intensity is typical for off-patent drugs.
- Therapeutic Equivalence: Regulatory bodies like the FDA ensure that generic versions are bioequivalent to the branded product, meaning they are expected to have the same therapeutic effect.
What is the Future Outlook for Pristiq?
The future of branded Pristiq is a continuation of its current trajectory – a significant decline in sales as generic alternatives dominate the market. Pfizer's strategic decisions will focus on managing the decline of this product and reallocating resources.
- Market Maturity: Pristiq has passed its peak market exclusivity period and is in the maturity and decline phase of its product lifecycle.
- Brand Loyalty: While some physician and patient loyalty to the branded product may persist, it is unlikely to offset the overwhelming economic advantage of generics.
- Intellectual Property: Any remaining intellectual property, such as secondary patents for specific formulations or uses, would have been either litigated or expired, further limiting any potential for sustained market control.
Key Takeaways
- Pristiq (desvenlafaxine) is approved for major depressive disorder and its primary composition of matter patent expired in 2017, leading to generic competition.
- Pristiq's global net sales peaked prior to generic entry, exceeding $1.4 billion annually.
- Generic desvenlafaxine has largely replaced branded Pristiq, causing a significant revenue decline for Pfizer's product.
- Sales projections for branded Pristiq indicate a continued downward trend, with revenues expected to fall below $0.10 billion by 2026.
- The broader desvenlafaxine market is driven by cost-effectiveness, physician acceptance of generics, and formulary preferences.
Frequently Asked Questions
What is the generic name for Pristiq?
The generic name for Pristiq is desvenlafaxine.
When did generic desvenlafaxine become available in the U.S.?
Generic desvenlafaxine became available in the U.S. in 2017 following patent litigation outcomes.
Does Pristiq have the same side effects as Effexor XR?
Pristiq is the active metabolite of venlafaxine (Effexor XR). Therefore, it shares a similar side effect profile, although individual patient responses can vary. Both are SNRIs and can cause side effects such as nausea, dizziness, insomnia, and sexual dysfunction.
How does desvenlafaxine work to treat depression?
Desvenlafaxine works by increasing the levels of serotonin and norepinephrine in the brain. These neurotransmitters are believed to be important in regulating mood, and imbalances in them are associated with depression.
What is the typical cost difference between brand-name Pristiq and generic desvenlafaxine?
The cost difference is substantial. Generic desvenlafaxine is typically 70-90% less expensive than branded Pristiq, depending on the pharmacy, insurance plan, and dosage.
Cited Sources
[1] U.S. Food and Drug Administration. (2008). Pristiq (desvenlafaxine) prescribing information. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/022105s000,022434s000lbl.pdf
[2] Reuters. (2017, June 29). Pfizer loses patent bid in US over antidepressant Pristiq. Retrieved from https://www.reuters.com/article/us-pfizer-patent/pfizer-loses-patent-bid-in-us-over-antidepressant-pristiq-idUSKBN19L261
[3] Pfizer Inc. (2017). Pfizer Inc. 2016 Annual Report. Retrieved from https://www.pfizer.com/sites/default/files/investors/financial_reports/annual_reports/2016-annual-report.pdf