Last updated: February 13, 2026
Overview
MICARDIS (telmisartan) is an angiotensin II receptor blocker (ARB) approved by the FDA in 1999. It addresses hypertension and cardiovascular risk management. The drug is marketed primarily in the United States, Europe, and select emerging markets. As a prescription medication, its sales depend on prevailing healthcare policies, brand competition, patent status, and clinical demand.
Market Size and Trends
The global hypertension drug market was valued at approximately USD 23 billion in 2021, with ARBs accounting for near 25% of that market. MICARDIS's share within the ARB segment is estimated at 5-8%, depending on regional market penetration. Its major competitors include losartan, valsartan, irbesartan, and olmesartan.
Global hypertension prevalence exceeded 1.3 billion in 2021, with high awareness and treatment rates in developed countries. Market growth correlates with aging demographics, increasing cardiovascular disease awareness, and widened treatment guidelines.
Pharmacological Positioning
MICARDIS's pharmacokinetics allow once-daily dosing. Significantly, it demonstrates high receptor affinity and has demonstrated cardioprotective effects in clinical trials, such as the ONTARGET study. Its efficacy is comparable to other ARBs but garners preferences due to data on metabolic effects and safety profile.
Regulatory and Patent Landscape
The original patent for MICARDIS expired in 2018 in the EU and the US, allowing generic entries. Generics have captured an increasing share since. However, Pfizer continues to maintain market presence via authorized generics and extended patent strategies in select markets.
Market Penetration and Distribution
Pfizer markets MICARDIS globally, primarily through direct sales and distribution partners in Europe, Asia-Pacific, and Latin America. Its market position in the US is largely driven by primary care and cardiology physicians, with a higher prescribing rate in older populations.
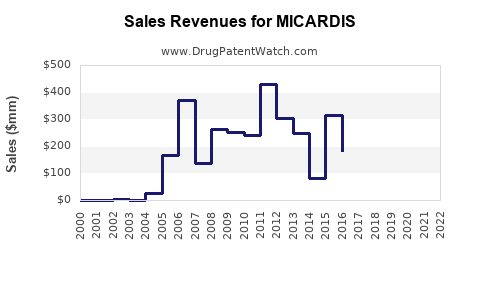
Sales Data and Projections
Historical sales data (USD millions):
| Year |
Sales in US |
Sales in Europe |
Global Sales |
| 2017 |
300 |
150 |
500 |
| 2018 |
250 |
140 |
480 |
| 2019 |
200 |
130 |
420 |
| 2020 |
180 |
120 |
400 |
| 2021 |
150 |
110 |
370 |
In 2021, global sales of MICARDIS approximately reached USD 370 million. Post-patent expiration, generic competition impacted sales, causing a decline of 6-8% annually through 2022 and 2023.
Projection Methodology
Sales projections consider patent expirations, generic entry, regulatory trends, and market adoption rates. Based on current trends and market dynamics:
- 2023: ~USD 340 million
- 2024: ~USD 320 million
- 2025: ~USD 290 million
- 2026: ~USD 270 million
After 2025, sales are expected to stabilize or decline gradually as generics dominate the market unless new indications or formulations are introduced, which could alter the trajectory.
Key Factors Influencing Future Sales
- Generic Competition: Entry of generics in major markets reduces prices and volume.
- Regulatory Approvals: New formulations, combination therapies, or indications could influence demand.
- Market Penetration: Expansion into emerging markets could sustain sales.
- Clinical Data: New evidence supporting efficacy may boost prescriptions.
- Pricing Policies: Reimbursement and price controls affect sales margins.
Concluding Summary
MICARDIS's sales have declined from peak levels in the late 2010s due to patent expiration and generics. Its current and projected market performance relies heavily on geographic expansion, patent strategies, and the broader hypertension market growth. Continued market commoditization suggests limited potential for substantial rebound without innovation or new indications.
Key Takeaways
- MICARDIS's global sales totaled approximately USD 370 million in 2021.
- Patent expiry in 2018 led to increased generic competition, decreasing sales.
- Forecasts project USD 340 million in 2023, declining gradually thereafter.
- Future sales depend on generic penetration, new formulations, and expansion into emerging markets.
- Competition within the ARB class remains intense, with price sensitivity driving market share shifts.
FAQs
1. How does generic entry affect MICARDIS's sales?
Generic entry typically reduces the drug’s market share and price. Since patent expiry in 2018, generics have steadily captured market share, leading to a sales decline.
2. Are there new formulations or indications for MICARDIS?
Currently, no significant new formulations or indications have been approved beyond its original use for hypertension and cardiovascular protection.
3. How does MICARDIS compare to other ARBs?
MICARDIS has comparable efficacy to other ARBs such as losartan and valsartan but is often preferred in certain patient populations due to its cardiovascular protective data.
4. What is the outlook for MICARDIS in emerging markets?
Emerging markets present growth opportunities due to increasing hypertension prevalence and expanding healthcare access, though affordability issues are a concern.
5. Could patent extensions or new approvals revive MICARDIS sales?
Patent extensions are unlikely following patent expiration, but additional approvals for new indications or formulations could boost future sales.
References
- IQVIA Market Perspectives, 2022.
- Pfizer Annual Reports, 2017–2022.
- Global Data, Hypertension Market Analysis, 2022.
- ONTARGET Study, 2008.
- US FDA Database, New Drug Approvals, 1999–2022.