Share This Page
Drug Sales Trends for LOCOID
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LOCOID (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
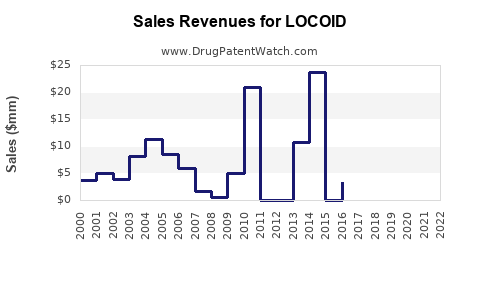
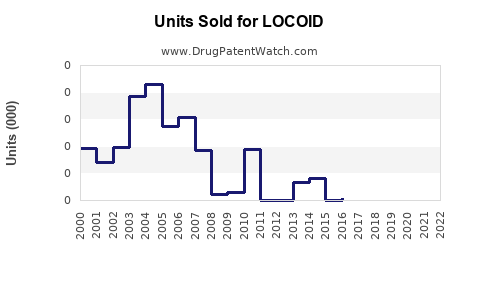
Annual Sales Revenues and Units Sold for LOCOID
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| LOCOID | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LOCOID Market Analysis and Sales Projections
LOCOID (hydrocortisone butyrate) is a topical corticosteroid used for the treatment of inflammatory skin conditions. Market performance is influenced by competition, patent landscape, and regulatory approvals in key markets.
What is the Current Market Status of LOCOID?
LOCOID is marketed by various pharmaceutical companies globally, often as a prescription topical corticosteroid. Its efficacy in treating conditions such as eczema, psoriasis, and dermatitis drives its sustained market presence. The drug is available in multiple formulations, including creams, ointments, and lotions, catering to different patient needs and application sites.
Key Market Dynamics:
- Therapeutic Area: Topical corticosteroids for dermatological inflammatory diseases.
- Formulations: Creams, ointments, lotions, and scalp solutions.
- Prescription Status: Primarily prescription-based in most major markets.
- Competitive Landscape: Faces competition from other topical corticosteroids (e.g., clobetasol propionate, betamethasone dipropionate) and newer biologic therapies for severe conditions.
What is the Patent Landscape for LOCOID?
The original patent for hydrocortisone butyrate has long expired, allowing for generic competition. However, companies may hold secondary patents related to specific formulations, delivery systems, or novel uses of the drug.
Patent Expirations and Generic Entry:
- Core Compound Patent: Expired. This has enabled the widespread availability of generic hydrocortisone butyrate products.
- Formulation Patents: Specific innovator formulations may have had or may still have patent protection. The expiration of these patents can open avenues for generic manufacturers to market bioequivalent versions of those specific formulations.
- New Indications/Delivery Systems: While less common for established drugs like LOCOID, any patents covering novel applications or advanced delivery mechanisms would extend market exclusivity for those specific innovations.
Analysis of Patent Expiration Impact:
The expiration of the primary patent for hydrocortisone butyrate has led to a fragmented market with numerous generic manufacturers. This intense competition typically results in price erosion, making branded LOCOID formulations reliant on brand recognition, specific excipient advantages, or superior clinical trial data to maintain market share and premium pricing.
What are the Sales Performance Trends for LOCOID?
Sales performance for LOCOID is a composite of branded product sales and generic hydrocortisone butyrate sales. Branded LOCOID sales are likely to exhibit a gradual decline or plateau due to generic penetration, while the overall market volume for hydrocortisone butyrate (including generics) remains substantial due to its established therapeutic value and broad applicability.
Global Sales Overview (Estimated):
Data on specific branded LOCOID sales is often aggregated within broader topical corticosteroid market reports. However, general market trends indicate:
- Branded LOCOID: Sales are likely to be in the tens of millions of U.S. dollars globally, depending on the specific brand and regional market presence. This segment is characterized by established brand loyalty and physician preference for specific formulations.
- Generic Hydrocortisone Butyrate: The total market value for generic hydrocortisone butyrate is significantly larger, likely in the hundreds of millions of U.S. dollars globally. This segment is highly price-sensitive and volume-driven.
Factors Influencing Sales:
- Prescription Volume: The number of prescriptions written for LOCOID and its generic equivalents.
- Pricing: The average selling price of branded and generic products.
- Market Access: Reimbursement policies and formulary placement by payers.
- Healthcare Provider Prescribing Habits: Physician familiarity and preference for specific brands or generics.
- Patient Adherence: Factors influencing patient compliance with treatment.
What are the Key Regulatory Considerations for LOCOID?
Regulatory approvals for LOCOID are essential for market entry and continued sales. These approvals are typically granted by national health authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Major Regulatory Approvals and Compliance:
- FDA (United States): LOCOID has been approved by the FDA for the treatment of skin conditions. Approval status and any specific labeling requirements are publicly available through the FDA's Orange Book.
- EMA (Europe): Similarly, LOCOID is registered and approved for use within the European Union. Marketing authorizations are granted for specific Member States.
- Other National Authorities: Approvals exist in numerous other countries worldwide.
- Post-Market Surveillance: Like all pharmaceuticals, LOCOID is subject to ongoing pharmacovigilance and regulatory oversight for safety and efficacy.
- Generic Drug Approvals: Generic versions of LOCOID require Abbreviated New Drug Applications (ANDAs) in the U.S. and similar abbreviated procedures in other regions, demonstrating bioequivalence to the reference listed drug.
What are the Projected Market Size and Growth for LOCOID?
Projecting future sales for LOCOID involves considering the continued influence of generics, evolving treatment paradigms for inflammatory skin diseases, and the overall growth of the dermatology market.
Market Size Projections (Hydrocortisone Butyrate, Global):
The total market for hydrocortisone butyrate, encompassing both branded and generic products, is expected to experience modest growth.
- Current Market Size (Estimated 2023-2024): $300 million - $500 million USD. This figure represents the aggregate sales value of all hydrocortisone butyrate products globally.
- Projected Compound Annual Growth Rate (CAGR) 2024-2029: 2% - 4%. This growth is primarily driven by an increasing prevalence of skin conditions and expanded access to healthcare in emerging markets, rather than significant price increases or new indications for hydrocortisone butyrate itself.
Factors Driving Future Growth:
- Increasing Incidence of Dermatological Conditions: Rising global populations and factors contributing to inflammatory skin diseases (e.g., environmental factors, stress) will sustain demand.
- Emerging Market Penetration: Improved healthcare infrastructure and increased disposable income in developing countries will expand access to prescription medications like LOCOID.
- Established Safety and Efficacy Profile: Hydrocortisone butyrate remains a first-line or second-line treatment option for many common skin ailments, ensuring continued physician and patient reliance.
Factors Limiting Future Growth:
- Intense Generic Competition: The continued dominance of generic products will limit significant price appreciation for branded LOCOID.
- Development of Advanced Therapies: The emergence of novel biologics and targeted therapies for severe and refractory dermatological conditions may shift treatment protocols for more complex cases away from traditional corticosteroids.
- Stricter Prescribing Guidelines: Efforts to minimize corticosteroid use due to potential side effects could influence prescription volumes for potent topical steroids.
What are the Competitive Threats to LOCOID?
LOCOID faces significant competitive threats from both within its therapeutic class and from emerging alternative treatments.
Key Competitive Threats:
- Other Topical Corticosteroids:
- Potency Spectrum: Competes directly with other mid-potency to high-potency topical corticosteroids (e.g., betamethasone valerate, triamcinolone acetonide) that may offer different therapeutic profiles or cost advantages.
- Formulation Innovation: Competitors may introduce novel formulations (e.g., foam, mousse) with improved patient compliance or efficacy.
- Non-Steroidal Topical Agents:
- Calcineurin Inhibitors: Tacrolimus and pimecrolimus offer steroid-sparing options, particularly for sensitive skin areas or long-term management.
- Phosphodiesterase-4 (PDE4) Inhibitors: Crisaborole provides a non-steroidal option for mild-to-moderate atopic dermatitis.
- Systemic and Biologic Therapies:
- For Moderate to Severe Disease: For patients with extensive or severe inflammatory skin conditions, systemic corticosteroids, immunosuppressants (e.g., methotrexate, cyclosporine), and biologic agents (e.g., dupilumab, secukinumab) are becoming increasingly common first-line or adjunctive treatments, potentially reducing the need for potent topical steroids.
- Generics: As previously noted, the broad availability of generic hydrocortisone butyrate significantly impacts the pricing and market share of branded LOCOID.
What are the Strengths of LOCOID?
LOCOID possesses established attributes that contribute to its continued market presence.
Key Strengths:
- Established Efficacy: Decades of clinical use have demonstrated its effectiveness in managing a wide range of inflammatory dermatoses.
- Broad Therapeutic Index: As a mid-potency corticosteroid, it offers a balance between efficacy and a generally favorable side effect profile when used appropriately.
- Multiple Formulations: Availability in various forms (cream, ointment, lotion) allows for customization based on skin type, lesion severity, and body location.
- Cost-Effectiveness (Generics): The widespread availability of generic hydrocortisone butyrate makes it a highly accessible and affordable treatment option for many patients.
- Physician Familiarity: Healthcare providers are well-acquainted with its use, prescribing habits, and expected outcomes.
What are the Weaknesses of LOCOID?
Despite its strengths, LOCOID has inherent limitations.
Key Weaknesses:
- Potential for Side Effects: Like all corticosteroids, prolonged or improper use can lead to skin atrophy, striae, telangiectasias, and systemic absorption issues.
- Limited Efficacy in Severe Disease: For severe or recalcitrant inflammatory skin conditions, LOCOID may not provide sufficient therapeutic benefit compared to stronger steroids or advanced therapies.
- Brand vs. Generic Differentiation: Branded LOCOID formulations face the challenge of differentiating themselves from lower-cost generic equivalents beyond perceived quality or specific excipients.
- Steroid Phobia: Growing awareness of corticosteroid side effects can lead to patient reluctance and physician preference for steroid-sparing alternatives.
What are the Opportunities for LOCOID?
Opportunities exist for companies manufacturing or marketing LOCOID products.
Key Opportunities:
- Targeted Marketing to Specific Dermatoses: Focusing marketing efforts on specific, well-defined inflammatory skin conditions where LOCOID remains a guideline-recommended therapy.
- Development of Novel Formulations: Investing in research and development for advanced delivery systems or formulations that enhance patient adherence, reduce side effects, or improve penetration (e.g., combination products, enhanced emollient bases).
- Emerging Market Expansion: Capitalizing on the growing demand for accessible dermatological treatments in underserved regions.
- Education and Physician Engagement: Reinforcing the appropriate use and benefits of hydrocortisone butyrate through targeted educational programs for healthcare professionals.
- Combination Therapies: Exploring the potential for fixed-dose combinations of hydrocortisone butyrate with other active ingredients (e.g., antifungals, antibacterials) to address multifactorial skin conditions.
What are the Threats to LOCOID?
External factors and market shifts pose threats to LOCOID's market position.
Key Threats:
- Increased Competition from Biologics and Novel Agents: The continued development and adoption of highly effective biologic and targeted therapies for inflammatory skin diseases, particularly for moderate-to-severe cases.
- Shifting Treatment Paradigms: A broader move towards steroid-sparing regimens as a primary approach for chronic inflammatory conditions.
- Regulatory Scrutiny and Labeling Changes: Potential for updated safety warnings or usage restrictions from regulatory bodies regarding topical corticosteroid use.
- Pricing Pressures: Continued downward pressure on pricing from payers and government health systems, especially for generic products.
- Adverse Event Reporting: An increase in reported adverse events associated with topical corticosteroids could lead to increased physician caution and patient apprehension.
Key Takeaways
LOCOID, as hydrocortisone butyrate, remains a significant topical corticosteroid, particularly its generic forms, due to its established efficacy and cost-effectiveness in treating inflammatory skin conditions. The market is characterized by intense generic competition, limiting significant price growth for branded products. Future market expansion is projected to be modest, driven by increasing disease prevalence and emerging market access, but will be constrained by the ongoing development of advanced therapies and a potential shift towards steroid-sparing treatment paradigms. Companies marketing LOCOID can leverage its established profile through targeted education, exploring novel formulations, and focusing on its role in specific dermatological indications.
Frequently Asked Questions
-
What is the difference between branded LOCOID and generic hydrocortisone butyrate? Branded LOCOID refers to specific formulations marketed under a proprietary name, while generic hydrocortisone butyrate encompasses products from multiple manufacturers that are bioequivalent to the reference product. Differences may exist in inactive ingredients, formulation specifics, and packaging, but the active ingredient and its therapeutic effect are the same.
-
How does LOCOID compare in potency to other topical corticosteroids? Hydrocortisone butyrate is classified as a mid-potency topical corticosteroid. It is less potent than very high-potency agents like clobetasol propionate but more potent than low-potency corticosteroids such as hydrocortisone (non-butyrate ester) or desonide. Its potency makes it suitable for a wide range of inflammatory skin conditions on the body, face, and intertriginous areas.
-
What are the most common side effects associated with LOCOID? Common side effects are typical of topical corticosteroids and can include burning, itching, irritation, dryness, and redness at the application site. Less common but more serious side effects with prolonged or excessive use include skin thinning (atrophy), stretch marks (striae), easy bruising, and increased risk of infection. Systemic absorption can occur with extensive application over large surface areas or under occlusion.
-
Can LOCOID be used for long-term treatment of chronic skin conditions? While LOCOID is effective for managing flare-ups of chronic inflammatory skin conditions, long-term, continuous use is generally discouraged due to the risk of side effects. Physicians typically recommend intermittent therapy, using the lowest effective potency and shortest duration necessary to control symptoms, or alternating with non-steroidal agents to minimize cumulative corticosteroid exposure.
-
What are the current trends in treating conditions typically managed by LOCOID, such as eczema or psoriasis? Current treatment trends emphasize a step-wise approach, starting with less potent agents and lifestyle modifications. For atopic dermatitis (eczema), there is a growing emphasis on emollients and steroid-sparing agents like calcineurin inhibitors and PDE4 inhibitors. For moderate to severe psoriasis and atopic dermatitis, biologic therapies and systemic immunosuppressants have become increasingly prominent, offering more targeted and sustained relief with potentially different side effect profiles.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/information-drug-pricing/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [2] European Medicines Agency. (n.d.). European Register of Medicinal Products. Retrieved from https://www.ema.europa.eu/en/medicines
More… ↓
