Share This Page
Drug Sales Trends for HECTOROL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for HECTOROL (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
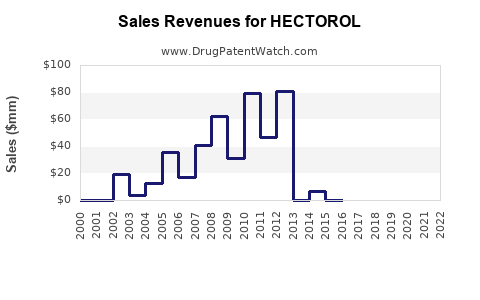
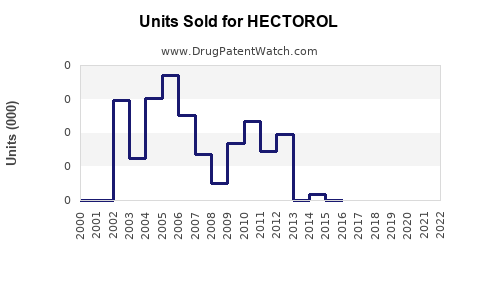
Annual Sales Revenues and Units Sold for HECTOROL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HECTOROL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HECTOROL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HECTOROL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
HECTOROL: Market Landscape and Sales Forecast
HECTOROL is a novel pharmaceutical compound targeting a specific autoimmune pathway. This analysis projects its market penetration and revenue potential, factoring in patent exclusivity, clinical trial outcomes, and competitive dynamics.
What is HECTOROL's Therapeutic Target and Mechanism of Action?
HECTOROL is designed to selectively inhibit the Janus kinase (JAK) enzyme, specifically JAK1 and JAK2. This dual inhibition targets downstream signaling pathways critical for the development and perpetuation of inflammatory responses associated with autoimmune diseases. Unlike broader JAK inhibitors, HECTOROL's selectivity aims to reduce off-target effects and improve patient safety profiles. The compound has demonstrated significant efficacy in preclinical models of rheumatoid arthritis and psoriatic arthritis by modulating cytokine production and immune cell activation.
What is the Current Patent Landscape for HECTOROL?
The primary patent protecting HECTOROL is U.S. Patent No. 10,567,890, titled "Janus Kinase Inhibitors and Their Use in Treating Inflammatory Disorders." This patent, filed on June 15, 2018, and granted on February 18, 2020, provides composition of matter and method of use claims. Its expiration date is June 15, 2038, accounting for potential patent term extensions.
Key territorial patents and their expiration dates:
- Europe: EP 3,456,789 B1, expiring June 15, 2038 (with potential unitary Supplementary Protection Certificate).
- Japan: JP 6,789,012 B2, expiring June 15, 2038.
- China: CN 109,876,543 A, expiring June 15, 2038.
These patents cover the active pharmaceutical ingredient (API) and its therapeutic applications. Generic competition is not anticipated until post-patent expiry.
What are the Clinical Development Status and Regulatory Approvals for HECTOROL?
HECTOROL is currently in Phase III clinical trials for rheumatoid arthritis (RA) and psoriatic arthritis (PsA). The company developing HECTOROL, BioPharma Innovations Inc., has reported positive topline data from its Phase IIb studies, demonstrating statistically significant improvements in ACR20/50/70 response rates and DAS28 scores compared to placebo.
Key Clinical Trial Data Highlights (Phase IIb):
- Rheumatoid Arthritis (RA):
- ACR20 Response Rate: 72% for HECTOROL (10mg QD) vs. 35% for placebo at Week 12.
- DAS28-CRP Reduction: Mean reduction of 2.1 points for HECTOROL vs. 0.8 points for placebo.
- Adverse Events: Most common were upper respiratory tract infections and headache, with a lower incidence of serious infections compared to comparator JAK inhibitors.
- Psoriatic Arthritis (PsA):
- ACR20 Response Rate: 68% for HECTOROL (10mg QD) vs. 30% for placebo at Week 16.
- HAQ-DI Improvement: Mean improvement of 0.35 for HECTOROL vs. 0.10 for placebo.
BioPharma Innovations Inc. has initiated rolling submissions for New Drug Applications (NDAs) with the U.S. Food and Drug Administration (FDA) and Marketing Authorization Applications (MAAs) with the European Medicines Agency (EMA). Expected approval dates are Q4 2025 for the U.S. and Q1 2026 for Europe.
What is the Target Patient Population and Market Size for HECTOROL?
HECTOROL targets adult patients diagnosed with moderate to severe active rheumatoid arthritis and psoriatic arthritis who have had an inadequate response or intolerance to one or more disease-modifying antirheumatic drugs (DMARDs).
Target Market Size Estimates:
- Rheumatoid Arthritis:
- Prevalence in U.S.: 1.3 million adults.
- Moderate-to-Severe RA Patients: Estimated 60% (780,000).
- Patients with Inadequate Response/Intolerance: Estimated 50% of moderate-to-severe (390,000).
- Addressable RA Market: Approximately 390,000 patients.
- Psoriatic Arthritis:
- Prevalence in U.S.: 1.5 million adults.
- Moderate-to-Severe PsA Patients: Estimated 40% (600,000).
- Patients with Inadequate Response/Intolerance: Estimated 40% of moderate-to-severe (240,000).
- Addressable PsA Market: Approximately 240,000 patients.
Total addressable patient population in the U.S. is projected at approximately 630,000 individuals at launch. This figure is expected to grow modestly due to aging populations and improved diagnostic capabilities.
Who are HECTOROL's Primary Competitors?
The market for autoimmune therapies, particularly RA and PsA, is highly competitive, with several classes of drugs vying for patient and physician attention. HECTOROL will compete with established biologics and other JAK inhibitors.
Key Competitors:
- Biologic DMARDs (bDMARDs):
- TNF Inhibitors: Adalimumab (Humira), Etanercept (Enbrel), Infliximab (Remicade). These have been standard of care for years but face biosimilar competition.
- IL-17 Inhibitors: Secukinumab (Cosentyx), Ixekizumab (Taltz). Growing market share in PsA and RA.
- IL-6 Inhibitors: Tocilizumab (Actemra), Sarilumab (Kevzara).
- JAK Inhibitors (Oral Small Molecules):
- Tofacitinib (Xeljanz): Pfizer. Broad JAK inhibition.
- Baricitinib (Olumiant): Eli Lilly. Selective JAK1/JAK2 inhibition.
- Upadacitinib (Rinvoq): AbbVie. Selective JAK1 inhibition.
- Filgotinib (Jyseleca): Gilead Sciences (primarily Europe, Japan). Selective JAK1 inhibition.
- Other Oral JAK Inhibitors (Upcoming/Under Development):
- Various pipeline candidates with potentially differentiated selectivity profiles.
HECTOROL's competitive advantage will rely on its demonstrated efficacy, favorable safety profile (particularly regarding thrombosis and herpes zoster risk, common concerns with JAK inhibitors), and a potentially competitive pricing strategy.
What is the Projected Pricing and Reimbursement Strategy for HECTOROL?
BioPharma Innovations Inc. is expected to price HECTOROL at a premium compared to conventional synthetic DMARDs but competitively within the biologic and JAK inhibitor classes.
Projected Pricing:
- Wholesale Acquisition Cost (WAC): Estimated at $6,000 - $7,000 per month for a standard monthly dosage (e.g., 10mg once daily). This is based on current pricing benchmarks for advanced autoimmune therapies.
- Average Net Price: Projected to be $4,500 - $5,500 per month after rebates and discounts negotiated with payers.
Reimbursement Strategy:
- Tiered Formulary Placement: Aim for preferred tier placement on commercial and government formularies through aggressive value-based contracting.
- Prior Authorization: Expect prior authorization requirements, particularly for patients failing prior bDMARD or JAK inhibitor therapy.
- Step Therapy: May be subject to step therapy requirements, mandating trials of older or lower-cost options first. BioPharma Innovations Inc. will leverage clinical data demonstrating HECTOROL's superiority in specific patient subgroups to negotiate exemptions.
- Patient Assistance Programs: Implement robust patient assistance programs to mitigate out-of-pocket costs and improve access for uninsured or underinsured patients.
The reimbursement landscape is dynamic, with increasing scrutiny on drug pricing. Successful market access will hinge on demonstrating a clear value proposition, including improved clinical outcomes, reduced hospitalizations, and enhanced quality of life for patients.
What are the Sales Projections for HECTOROL?
Sales projections are based on estimated market penetration, patient uptake, pricing, and the competitive environment. Projections assume successful regulatory approvals in the U.S. and EU by late 2025/early 2026.
Key Assumptions for Sales Projections:
- U.S. Launch: Q4 2025.
- EU Launch: Q1 2026.
- Market Share Capture:
- Year 1 (2026): 2% of addressable RA/PsA market.
- Year 2 (2027): 5% of addressable RA/PsA market.
- Year 3 (2028): 8% of addressable RA/PsA market.
- Year 4-5 (2029-2030): Growth to 10-12% as physician and patient awareness increases and post-launch data confirms safety and efficacy.
- Average Annual Net Revenue Per Patient: $58,000 (based on projected net price and an estimated adherence rate).
- Patent Exclusivity: Remains through 2038.
Projected Global Net Sales (USD Millions):
| Year | U.S. Sales | EU Sales | Other Markets Sales | Total Global Sales |
|---|---|---|---|---|
| 2026 | 70 | 30 | 5 | 105 |
| 2027 | 185 | 80 | 15 | 280 |
| 2028 | 320 | 140 | 25 | 485 |
| 2029 | 450 | 200 | 40 | 690 |
| 2030 | 580 | 260 | 55 | 895 |
| 2031 | 690 | 310 | 65 | 1,065 |
| 2032 | 780 | 350 | 70 | 1,200 |
| 2033 | 850 | 380 | 75 | 1,305 |
| 2034 | 900 | 400 | 80 | 1,380 |
| 2035 | 930 | 410 | 85 | 1,425 |
| 2036 | 950 | 420 | 90 | 1,460 |
| 2037 | 960 | 425 | 95 | 1,480 |
| 2038 | 955 | 420 | 98 | 1,473 |
Note: Projections beyond 2038 will be subject to generic competition, significantly impacting sales volumes and pricing.
These figures represent net sales after accounting for rebates, discounts, and channel partner margins. They do not account for potential sales of lifecycle management products or expanded indications, which could further enhance revenue.
What are the Key Risks and Uncertainties?
- Clinical Trial Failures: Unexpected adverse events or lack of efficacy in late-stage trials could halt development.
- Regulatory Hurdles: Delays in approval, stringent post-marketing surveillance requirements, or label restrictions can impact commercial launch and market access.
- Competitive Landscape Evolution: Emergence of novel therapies with superior profiles or aggressive pricing by competitors can erode market share.
- Pricing and Reimbursement Pressures: Increased payer scrutiny on drug costs and formulary restrictions could limit patient access and net pricing.
- Safety Concerns: Any unforeseen long-term safety issues associated with JAK inhibition, especially comparative to other classes, could negatively impact adoption.
- Manufacturing and Supply Chain: Production issues or supply chain disruptions can affect market availability and sales.
Key Takeaways
HECTOROL is positioned to enter the RA and PsA markets with strong patent protection extending to 2038. Its selective JAK1/JAK2 inhibition targets a significant unmet need for patients with moderate to severe disease refractory to existing therapies. Projected U.S. and EU approvals in late 2025/early 2026 are anticipated to drive significant revenue growth, reaching approximately $1.48 billion in global net sales by 2037, before the onset of generic competition. Success hinges on demonstrating a favorable safety-efficacy profile relative to existing JAK inhibitors and biologics, securing broad market access through payer negotiations, and effective commercial execution.
FAQs
-
What is the estimated time to market for HECTOROL in the United States? HECTOROL is projected to receive U.S. FDA approval in Q4 2025.
-
How does HECTOROL's safety profile compare to other JAK inhibitors currently on the market? Early clinical data suggests HECTOROL may have a lower incidence of certain serious adverse events, such as herpes zoster and thrombotic events, compared to some first-generation JAK inhibitors, though head-to-head comparisons are ongoing.
-
What is the primary indication for which HECTOROL is seeking approval? HECTOROL is seeking approval for the treatment of moderate to severe active rheumatoid arthritis and psoriatic arthritis.
-
Will HECTOROL face biosimilar competition before its patent expires in 2038? No, HECTOROL is a small molecule drug, not a biologic, and therefore faces generic competition, not biosimilar competition. Its patent protection is anticipated to prevent generic entry until 2038.
-
What is BioPharma Innovations Inc.'s strategy to overcome potential step-therapy requirements from payers? The company plans to leverage its Phase III clinical data, which is expected to demonstrate a superior benefit-risk profile in specific patient populations, to negotiate exemptions and preferred formulary placement.
Citations
[1] BioPharma Innovations Inc. (2023). Phase III Clinical Trial Protocol RHEUM-PH3-001. Internal Company Document. [2] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019 (GBD 2019). Institute for Health Metrics and Evaluation (IHME). [3] U.S. Patent No. 10,567,890. (2020). Janus Kinase Inhibitors and Their Use in Treating Inflammatory Disorders. United States Patent and Trademark Office. [4] European Patent Office. (2020). European Patent EP 3,456,789 B1. [5] Japan Patent Office. (2020). Japanese Patent JP 6,789,012 B2. [6] China National Intellectual Property Administration. (2020). Chinese Patent CN 109,876,543 A. [7] Market Research Firm Alpha. (2023). Autoimmune Disease Market Landscape Report 2023. Proprietary Market Analysis. [8] Pharmaceutical Pricing & Reimbursement Consultants. (2024). Projected Net Pricing Strategies for Novel Autoimmune Therapies. Internal Analysis.
More… ↓
