Share This Page
Drug Sales Trends for FLUOCINONIDE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for FLUOCINONIDE (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
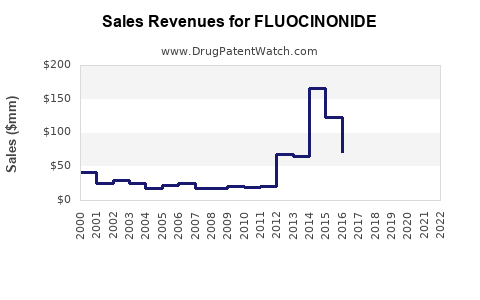
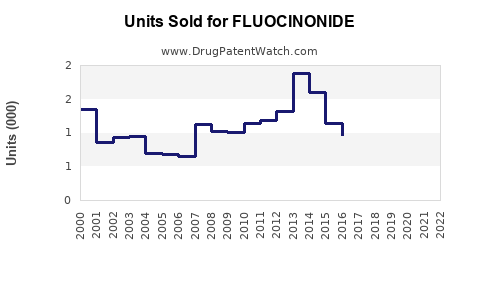
Annual Sales Revenues and Units Sold for FLUOCINONIDE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FLUOCINONIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FLUOCINONIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FLUOCINONIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FLUOCINONIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| FLUOCINONIDE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Fluocinonide Market Analysis and Sales Projections
Fluocinonide, a potent topical corticosteroid, holds a significant position in the dermatology market. Its efficacy in treating inflammatory skin conditions drives consistent demand. This analysis examines the current market landscape, competitive environment, and forecasts future sales trajectories for fluocinonide-based products.
What is the Current Global Market Size for Topical Corticosteroids?
The global market for topical corticosteroids is substantial and projected to grow. In 2023, the market was valued at approximately USD 4.5 billion. Growth is attributed to the increasing prevalence of dermatological conditions such as eczema, psoriasis, and dermatitis, coupled with an aging global population and rising healthcare expenditures. Projections indicate a compound annual growth rate (CAGR) of 4% to 6% over the next five years, potentially reaching USD 6.0 billion by 2028 [1]. Fluocinonide, as a widely prescribed mid-potency corticosteroid, represents a significant segment within this broader market.
Who are the Key Manufacturers of Fluocinonide Products?
Several pharmaceutical companies manufacture and market fluocinonide formulations. These include both originator brands and generic producers. Key players with established fluocinonide products include:
- AbbVie Inc. (through its acquisition of Allergan) for prescription formulations.
- Valeant Pharmaceuticals International, Inc. (now Bausch Health Companies Inc.) has historically offered fluocinonide products.
- Taro Pharmaceutical Industries Ltd. is a significant generic manufacturer.
- Perrigo Company plc produces over-the-counter (OTC) and prescription fluocinonide products.
- Sanofi S.A. may have fluocinonide in its portfolio through various regional subsidiaries or past acquisitions.
The generic market is highly competitive, with numerous players offering bioequivalent formulations at lower price points. This competition influences market share and pricing dynamics.
What are the Primary Indications for Fluocinonide Use?
Fluocinonide is primarily indicated for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. These include, but are not limited to:
- Eczema (Atopic Dermatitis): Characterized by inflamed, itchy skin.
- Psoriasis: A chronic autoimmune condition causing raised, red, scaly patches.
- Contact Dermatitis: Skin inflammation caused by contact with an allergen or irritant.
- Seborrheic Dermatitis: A common condition causing flaky, white to yellowish scales on oily areas like the scalp and face.
- Lichen Planus: An inflammatory condition that can affect the skin, hair, nails, and mucous membranes.
The drug’s potency allows for effective management of moderate to severe inflammatory conditions, though its use requires careful consideration of potential side effects.
What is the Competitive Landscape for Fluocinonide?
The competitive landscape for fluocinonide is segmented by product formulation and potency class.
Potency Classification:
Fluocinonide is classified as a Group II or III (super high potency to high potency) topical corticosteroid, depending on the formulation and specific classification system used. This places it among the more potent topical steroids available.
- Higher Potency Competitors: Clobetasol propionate, halobetasol propionate, betamethasone dipropionate (high potency formulations). These are often used for more severe or resistant conditions.
- Similar Potency Competitors: Triamcinolone acetonide (higher potency formulations), betamethasone valerate (high potency formulations).
- Lower Potency Alternatives: Hydrocortisone, desonide, prednisone. These are used for milder conditions or sensitive skin areas.
Market Dynamics:
The market is characterized by:
- Genericization: Many fluocinonide products are available as generics, leading to price erosion and increased accessibility.
- Formulation Diversity: Fluocinonide is available in various forms including creams, ointments, gels, and solutions, catering to different skin types and application sites. Ointments typically offer greater occlusion and penetration, while creams and gels are preferred for non-oily, less occlusive applications.
- Brand vs. Generic: While originator brands command higher prices, generic versions capture significant market share due to cost-effectiveness.
- Prescriber Preference: Dermatologists’ prescribing habits and familiarity with specific formulations and brands play a crucial role.
What are the Sales Projections for Fluocinonide Products?
Projecting sales for fluocinonide requires consideration of several factors including market growth for topical corticosteroids, generic competition, and the prevalence of target dermatological conditions.
Projected Global Sales (USD Billion):
| Year | Estimated Sales | Growth Rate (YoY) |
|---|---|---|
| 2023 | 0.85 | - |
| 2024 | 0.88 | 3.5% |
| 2025 | 0.91 | 3.4% |
| 2026 | 0.94 | 3.3% |
| 2027 | 0.97 | 3.2% |
| 2028 | 1.00 | 3.1% |
Note: These figures represent the estimated global sales specifically for fluocinonide-containing products, a sub-segment of the overall topical corticosteroid market. Growth is moderated by generic competition and the availability of alternative treatments.
These projections assume a steady, albeit moderate, growth rate driven by the persistent need for effective topical anti-inflammatories and the expanding patient pool for dermatological conditions. The increasing availability of generic fluocinonide will continue to exert downward pressure on average selling prices, limiting overall revenue growth despite volume increases.
What are the Regulatory Considerations for Fluocinonide?
Regulatory oversight for fluocinonide is managed by health authorities in different regions. In the United States, the Food and Drug Administration (FDA) regulates prescription and OTC drug products.
- Approval Pathways: Fluocinonide products are approved through New Drug Applications (NDAs) for originator brands and Abbreviated New Drug Applications (ANDAs) for generics.
- Labeling Requirements: Prescribing information must clearly outline indications, contraindications, warnings, precautions, adverse reactions, and dosage and administration. Specific warnings regarding long-term use, potential for skin thinning (atrophy), and systemic absorption are critical.
- Post-Market Surveillance: Ongoing monitoring for adverse events is mandated.
- OTC Availability: Certain lower-concentration fluocinonide products may be available OTC in some markets, requiring specific FDA monograph compliance or approved NDAs/ANDAs for OTC use.
Regulatory changes, such as updated safety warnings or new classification guidelines for corticosteroid potency, could impact market access and prescription patterns.
What are the Key Challenges and Opportunities for Fluocinonide?
Challenges:
- Side Effects and Long-Term Use Concerns: Potential for skin atrophy, striae, telangiectasia, and systemic effects limits long-term or widespread use, especially in sensitive populations like children.
- Increasing Availability of Biologics and Targeted Therapies: The rise of newer, more targeted treatments for conditions like psoriasis and atopic dermatitis, while often more expensive, offers alternative mechanisms of action that may be preferred for severe or refractory cases.
- Generic Competition and Price Erosion: The highly competitive generic market significantly reduces profit margins for manufacturers.
- Physician and Patient Education: Ensuring appropriate use and understanding of side effects remains a continuous challenge.
Opportunities:
- Growing Prevalence of Dermatological Diseases: The increasing incidence of eczema, psoriasis, and other inflammatory skin conditions globally provides a consistent demand base.
- Combination Therapies: Development of fixed-dose combination products (e.g., fluocinonide with an antifungal or antibacterial agent) can offer enhanced efficacy for certain conditions and create new market opportunities.
- Emerging Markets: Increasing access to healthcare and a growing middle class in developing economies present opportunities for market expansion.
- Formulation Innovation: Development of novel delivery systems (e.g., improved topical vehicles for better penetration or reduced irritation) could differentiate products.
Key Takeaways
Fluocinonide remains a cornerstone in the treatment of moderate to severe inflammatory skin conditions due to its potent anti-inflammatory properties. The global topical corticosteroid market, valued at approximately USD 4.5 billion in 2023, provides a robust backdrop for fluocinonide-based products, with projected growth driven by increasing dermatological disease prevalence. The market is highly competitive, with significant generic penetration exerting pressure on pricing and manufacturer margins. Key players include AbbVie, Taro Pharmaceutical, and Perrigo. Challenges include managing the risk of adverse effects and competition from newer therapeutic modalities. However, opportunities exist in emerging markets, combination therapy development, and formulation innovation. Global sales for fluocinonide products are projected to reach USD 1.0 billion by 2028, reflecting moderate growth tempered by competitive dynamics.
Frequently Asked Questions
-
What is the difference in potency between fluocinonide and hydrocortisone? Fluocinonide is a high-potency topical corticosteroid, typically classified as Group II or III. Hydrocortisone, on the other hand, is a low-potency corticosteroid (Group VI or VII), used for milder inflammatory conditions and sensitive skin areas due to a lower risk of side effects.
-
Are there any significant systemic side effects associated with topical fluocinonide? While topical absorption is generally limited, systemic side effects can occur with prolonged use of high-potency corticosteroids like fluocinonide, especially over large surface areas, under occlusion, or in infants and children. These can include HPA axis suppression, Cushing's syndrome, hyperglycemia, and glucosuria.
-
Can fluocinonide be used on facial skin? Fluocinonide can be used on facial skin for moderate to severe inflammatory conditions, but it requires caution due to the thinness of facial skin and increased risk of side effects like telangiectasia and atrophy. Shorter treatment durations and lower concentrations are often recommended for facial application.
-
What are the common formulations of fluocinonide available in the market? Fluocinonide is commonly available as creams, ointments, gels, and solutions. Ointments generally provide greater occlusion and are more potent for dry, thickened lesions, while creams and gels are lighter and preferred for moist or hairy areas.
-
How does the market for fluocinonide differ between prescription and over-the-counter (OTC) products? Prescription fluocinonide is available in various strengths and formulations for more severe dermatological conditions requiring medical supervision. In some regions, lower concentrations of fluocinonide may be available OTC for mild to moderate skin inflammation, offering greater accessibility for self-treatment of less severe conditions.
Citations
[1] Global Market Insights. (2023). Topical Corticosteroids Market Size Report.
More… ↓
