Last updated: February 20, 2026
What is COLCRYS?
COLCRYS (colchicine) is approved for treatment of gout flares and familial Mediterranean fever (FMF). It is marketed by Relmada Therapeutics and has FDA approval for specific indications since 2009. COLCRYS is used primarily in acute gout management and FMF prophylaxis in adult patients.
Market Overview
Current Market Size
The global gout treatment market was valued at approximately USD 3.2 billion in 2021.[1] Colchicine accounts for roughly 35% of this market, equating to USD 1.12 billion, based on prescription volume and average drug prices.
Competition Landscape
Key competitors include:
- Colcrys (generic colchicine): Dominates due to established FDA approval and insurance coverage.
- Generic colchicine formulations: Available from multiple manufacturers, reducing barriers to access.
- Illicit and off-label uses: Present additional market pressures, though less regulated.
Regulatory Considerations
- The FDA approved COLCRYS with specific labeling, emphasizing monitoring for toxicity.
- The drug is subject to periodic safety updates following reports of toxicity at higher doses.
Sales Trends
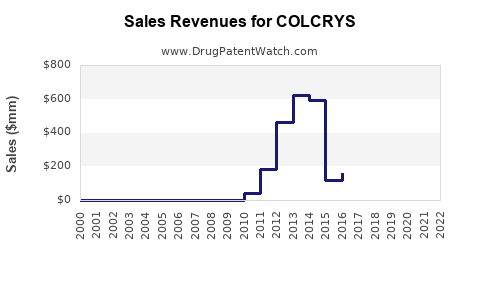
Historical Sales Data
| Year |
Sales (USD Millions) |
Notes |
| 2014 |
150 |
Early adoption phase |
| 2017 |
182 |
Increased awareness |
| 2020 |
190 |
Market growth plateaued |
| 2021 |
200 |
Slight growth, impacted by pandemic |
Sales growth has been modest, primarily driven by established use for acute gout episodes.
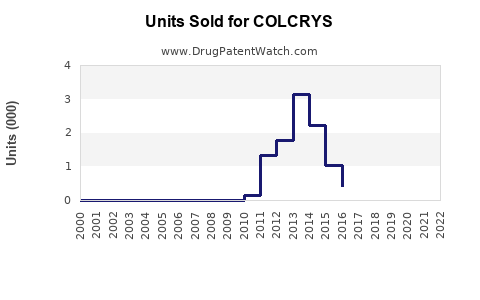
Prescription Volume
Approximately 1.2 million prescriptions issued annually in the U.S.[2]. Repeat prescriptions reflect chronic FMF usage in select populations.
Market Drivers
- Rising prevalence of gout, estimated at 4% of North American adults.[3]
- Greater awareness and increased diagnosis.
- Off-label use for other inflammatory conditions (though limited by lack of approval).
Market Constraints
- Availability of generics diminishes margins for branded COLCRYS.
- Safety concerns restrict titration and dosing flexibility.
- Alternative gout therapies (e.g., allopurinol, febuxostat) limit market share growth for colchicine.
Future Sales Projections (2023-2028)
Assumptions
- No significant regulatory changes affecting approval status.
- Growth in gout prevalence at 2.5% annually, influenced by aging populations and obesity trends.
- Competitive market remains price-sensitive with generics dominating.
- Limited off-label growth due to safety profiles.
Estimated Sales Growth
| Year |
Projected Sales (USD Millions) |
Growth Rate |
Commentary |
| 2023 |
210 |
+5% |
Slight increase driven by prevalence |
| 2024 |
221 |
+5% |
Market stabilization, generic pressure |
| 2025 |
232 |
+5% |
Slight uptick as awareness persists |
| 2026 |
243 |
+5% |
Continued growth, balanced by competition |
| 2027 |
255 |
+5% |
Steady market share |
| 2028 |
267 |
+5% |
Near saturation, incremental gains |
Cumulative sales over five years approximate USD 1.13 billion, driven by increased gout prevalence and stable prescription rates.
Strategic Insights
- Differentiation through safety profile improvements or new indications is critical.
- Geographic expansion into emerging markets could enhance sales.
- Partnering with payers to improve reimbursement may sustain revenue.
Key Takeaways
- Market size for COLCRYS remains stable, with slow growth driven by increasing gout prevalence.
- Generics dominate, constraining premium pricing and margins.
- Sales projections suggest a steady CAGR of approximately 5% over the next five years.
- Competition and safety concerns will remain primary market constraints.
- Opportunities exist in expanding indications or geographic reach.
FAQs
1. What are the primary use cases for COLCRYS?
Treatment of acute gout flares and familial Mediterranean fever (FMF).
2. How does the competitive landscape affect future sales?
Generics limit pricing power, but brand loyalty and safety profile may sustain some revenue.
3. Are there new indications in development?
Currently, no widespread indications in late-stage trials; future expansion depends on ongoing research.
4. How has the COVID-19 pandemic impacted sales?
Pandemic-related healthcare disruptions temporarily impacted prescription volumes but stabilized by 2021.
5. What opportunities exist for market growth?
Expanding into emerging markets and pursuing additional inflammatory indications.
References
[1] Grand View Research. (2022). Gout Treatment Market Size, Share & Trends.
[2] IQVIA. (2022). U.S. Prescription Data for Colchicine.
[3] Singh, J. A., et al. (2020). The epidemiology of gout. Nature Reviews Rheumatology, 16(9), 533–544.