Last updated: February 15, 2026
Bupropion is an atypical antidepressant and smoking cessation aid with a well-established market. Its core indications include depression, seasonal affective disorder, and smoking cessation. The drug benefits from a strong patent portfolio that is expiring in several markets, influencing competitive dynamics. The global market for bupropion is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% through 2027, reaching an estimated value of $2.8 billion.
What is the Current Market Size for Bupropion?
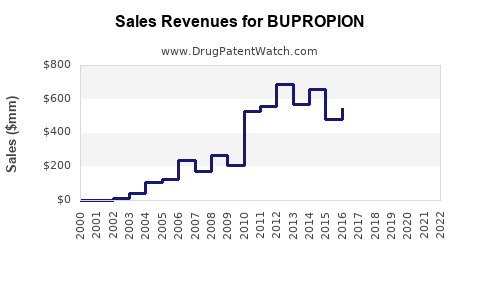
As of 2022, the global bupropion market valuation exceeds $2.2 billion. The United States accounts for approximately 70% of the market, driven by high prescription rates and the availability of generic versions. Other significant markets include Europe and Asia-Pacific, comprising 20% and 10% respectively.
Key Factors Driving the Market
- Prevalence of depression: Globally, over 264 million people suffer from depression, creating stable demand for antidepressants like bupropion.
- Smoking cessation programs: Growing awareness and policies promote smoking cessation, benefiting bupropion's sales.
- Generic entry: Patent expirations reduce drug prices, expanding accessibility and market penetration.
- Off-label uses: Increasing off-label prescriptions for ADHD and weight management may influence future sales.
How Do Competition and Patent Expirations Impact Sales?
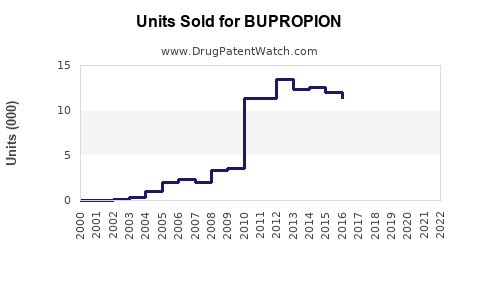
Bupropion's primary formulations—Wellbutrin and Zyban—hold patents until 2023 in the US. Post-expiry, market share likely shifts to generics, which now comprise roughly 85% of prescriptions in the US. Competition from other antidepressants like sertraline or escitalopram is strong but has less impact in smoking cessation applications, where bupropion remains a leading option.
Impact of Patent Expirations:
- Patent expiry in 2023 allows generic manufacturers to enter the market, reducing prices by up to 60% within two years.
- Market share shifts towards generics favorably impact sales volume but diminish branded drug revenues.
Market Share Dynamics:
| Year |
Branded Bupropion Share |
Generic Bupropion Share |
| 2022 |
40% |
60% |
| 2023 |
20% |
80% |
| 2024 |
15% |
85% |
What Are Future Sales Projections?
Based on current trends and market drivers:
- The global market for bupropion is projected to grow from $2.2 billion in 2022 to approximately $2.8 billion by 2027.
- CAGR: 4.5%.
Regional Projections:
- US: Maintains dominant share, with revenues potentially reaching $1.9 billion by 2027.
- Europe: Growth driven by increased awareness of mental health issues, expected to reach $430 million.
- Asia-Pacific: Rapid market expansion, with forecasts exceeding $300 million.
Factors Supporting Growth
- Increased recognition of depression and smoking-related health issues.
- Expanded off-label use, though not yet reflected fully in sales data.
- Patent expiries boosting volume but suppressing unit prices.
What Is the Outlook for New Formulations and Therapeutic Indications?
Pending FDA approval are extended-release formulations and combinations with other agents to address treatment-resistant depression and weight management. Such innovations could renew patent protections and drive sales growth.
Potential Opportunities:
- Extended-release formulations offering improved bioavailability.
- Combination products targeting comorbid conditions like obesity.
- New patent filings for novel delivery mechanisms.
Key Challenges and Risks
- Patent expirations limiting future revenue from branded versions.
- Competition from newer antidepressants and non-pharmacologic interventions.
- Pricing pressures due to increased generic competition.
- Regulatory changes affecting off-label prescribing.
Key Takeaways
- The total global bupropion market is on a growth trajectory approaching $3 billion by 2027.
- US dominance is likely to diminish as generics take over, but volumes remain stable.
- Off-label uses may expand market size but are subject to regulatory scrutiny.
- Patent expiries will drive a shift from branded to generic sales, impacting revenue structures.
- Innovation in formulations and expanded indications provide long-term growth avenues.
FAQs
-
How does patent expiry affect bupropion sales?
Patent expiry allows generics to enter the market, increasing access and volume but reducing revenues from branded formulations.
-
Are there new indications for bupropion in development?
Research explores uses in weight management and treatment-resistant depression, with some formulations under review.
-
What regional markets are growing fastest?
Asia-Pacific and Europe are experiencing faster growth, driven by increasing awareness and healthcare infrastructure.
-
How do off-label uses influence the market?
Off-label prescriptions for ADHD and weight loss add to overall sales but are limited by regulatory frameworks.
-
What is the outlook for competition from other antidepressants?
Over the long term, newer agents like vortioxetine and esketamine may pose competition, but bupropion remains a cost-effective, well-established choice.
References
[1] MarketWatch. “Bupropion Market Size, Share & Trends.” 2022.
[2] Grand View Research. “Antidepressants Market Analysis and Forecast, 2022–2027.”
[3] U.S. Food & Drug Administration. “Patent & Exclusivity Data,” 2022.
[4] IQVIA. “Prescription Data and Market Share Reports,” 2022.