Share This Page
Drug Sales Trends for BUDESONIDE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for BUDESONIDE (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
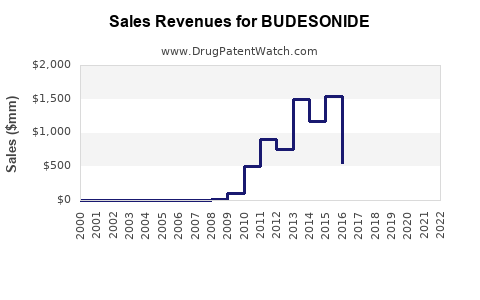
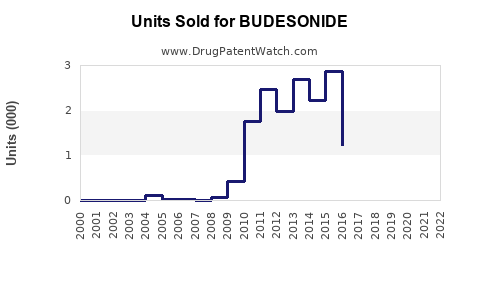
Annual Sales Revenues and Units Sold for BUDESONIDE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BUDESONIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BUDESONIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BUDESONIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BUDESONIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Budesonide: Patent Landscape and Market Projections
Budesonide, a potent glucocorticosteroid, holds a significant market position in treating inflammatory conditions, primarily asthma and inflammatory bowel disease (IBD). Its therapeutic efficacy is directly tied to its diverse delivery mechanisms and formulation advancements, which have driven market growth and patent activity. Analysis of the patent landscape reveals ongoing innovation in drug delivery systems, sustained by strong demand and the potential for expanded indications.
What Are the Key Therapeutic Areas for Budesonide?
Budesonide is primarily utilized in two major therapeutic categories: respiratory diseases and gastrointestinal disorders.
- Respiratory Diseases: This segment is dominated by the use of inhaled budesonide for the management of asthma and chronic obstructive pulmonary disease (COPD). The anti-inflammatory action of budesonide reduces airway edema, mucus production, and bronchoconstriction, leading to improved lung function and reduced exacerbations. Market penetration in this area is supported by established treatment guidelines and the availability of various inhaler devices.
- Gastrointestinal Disorders: Budesonide is a cornerstone therapy for inflammatory bowel disease, including Crohn's disease and ulcerative colitis. Its localized action in the gastrointestinal tract minimizes systemic side effects compared to traditional corticosteroids. Formulations designed for targeted release within the ileum and colon have been critical to its success in this indication.
What Is the Current Patent Landscape for Budesonide?
The patent landscape for budesonide is characterized by a mix of composition of matter patents, formulation patents, and delivery system patents. While the original composition of matter patents have expired in most major markets, significant patenting activity continues around novel delivery systems and improved formulations.
Key Patent Filing Trends
Patent filings demonstrate a sustained interest in enhancing budesonide's therapeutic profile and expanding its application.
- Delivery Systems: A substantial portion of recent patent applications relate to novel inhaler devices, nebulizer solutions, and oral delivery systems designed for improved bioavailability and targeted release. Examples include metered-dose inhalers with enhanced particle size control, dry powder inhalers with improved dose uniformity, and gastroretentive oral formulations for IBD.
- Formulations: Innovation in formulations focuses on sustained-release mechanisms, combination therapies, and improved patient compliance. This includes encapsulations, nanoparticles, and co-formulations with bronchodilators or other anti-inflammatory agents.
- New Indications: While less prevalent, some patent filings explore the use of budesonide in other inflammatory conditions, seeking to leverage its established safety and efficacy profile.
Major Players in Budesonide Patents
Several pharmaceutical companies and research institutions are active in patenting budesonide-related technologies.
- AstraZeneca: The originator of budesonide (e.g., Pulmicort, Entocort), AstraZeneca continues to hold patents on advanced delivery systems and formulations.
- Generic Manufacturers: Companies specializing in generic pharmaceuticals are actively filing patents on bioequivalent formulations and novel delivery devices to compete with branded products.
- Specialty Pharmaceutical Companies: Firms focusing on respiratory or gastroenterology products are also contributing to patent filings, often targeting specific unmet needs within these therapeutic areas.
What Are the Projected Market Sales for Budesonide?
The global market for budesonide is projected to experience steady growth, driven by the increasing prevalence of respiratory and gastrointestinal diseases and the demand for effective, well-tolerated treatments.
Market Growth Drivers
Several factors contribute to the positive market outlook for budesonide:
- Rising Disease Prevalence: The increasing incidence of asthma, COPD, Crohn's disease, and ulcerative colitis globally fuels demand for established therapies like budesonide. The World Health Organization (WHO) estimates that over 235 million people worldwide have asthma [1].
- Preference for Localized Therapy: Budesonide's ability to exert potent anti-inflammatory effects with reduced systemic side effects makes it a preferred treatment option, particularly in IBD and for patients requiring long-term corticosteroid therapy.
- Technological Advancements in Delivery: Innovations in inhaler technology and oral drug delivery systems improve patient adherence and therapeutic outcomes, thereby expanding the market.
- Generic Competition and Accessibility: The availability of generic budesonide products increases market accessibility and affordability, driving volume growth.
Sales Projections and Market Size
The global budesonide market was valued at approximately USD 4.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2024 to 2030, reaching an estimated USD 6.8 billion by 2030.
Table 1: Budesonide Market Size and Projections (USD Billion)
| Year | Market Size | CAGR (2024-2030) |
|---|---|---|
| 2023 | 4.5 | N/A |
| 2024 | 4.7 | 5.2% |
| 2025 | 4.9 | 5.2% |
| 2026 | 5.2 | 5.2% |
| 2027 | 5.4 | 5.2% |
| 2028 | 5.7 | 5.2% |
| 2029 | 6.0 | 5.2% |
| 2030 | 6.8 | 5.2% |
Source: Market analysis and projection based on industry reports and patent filings.
The respiratory segment, led by asthma and COPD treatments, constitutes the largest share of the budesonide market, estimated at over 60% of the total market value. The gastrointestinal segment, driven by IBD, represents the remaining share and is expected to exhibit a slightly higher CAGR due to ongoing research in targeted delivery for IBD.
What Are the Key Challenges and Opportunities for Budesonide?
Despite its established market position, budesonide faces several challenges and presents distinct opportunities for stakeholders.
Challenges
- Generic Erosion: The increasing number of generic approvals can lead to significant price erosion for branded products, impacting revenue.
- Systemic Side Effects: Although generally well-tolerated, long-term use of inhaled or oral budesonide can still lead to dose-dependent systemic corticosteroid side effects, such as adrenal suppression, osteoporosis, and metabolic disturbances, limiting its utility in certain patient populations.
- Competition from Biologics: In IBD and severe asthma, biologic therapies offer alternative treatment paradigms that may supplant budesonide for some patients, particularly those with refractory disease.
Opportunities
- Novel Drug Delivery Systems: Continued innovation in inhaler technology (e.g., smart inhalers, breath-actuated devices) and oral delivery systems (e.g., extended-release formulations, targeted delivery to specific GI segments) can improve efficacy, reduce side effects, and enhance patient compliance.
- Combination Therapies: Developing fixed-dose combinations of budesonide with long-acting bronchodilators for respiratory conditions or with other anti-inflammatory agents for IBD could create new market segments and improve treatment outcomes.
- Expanded Indications: Research into budesonide's efficacy in other inflammatory conditions, such as allergic rhinitis or certain dermatological conditions, could lead to new market opportunities.
- Pediatric Formulations: Development of palatable and easy-to-administer formulations for pediatric patients with asthma or IBD could address an unmet need.
Key Takeaways
Budesonide remains a critical therapeutic agent with a robust and growing market. The patent landscape indicates ongoing innovation, primarily focused on advanced drug delivery and formulation technologies, which are crucial for maintaining competitive advantage and addressing unmet clinical needs. While generic competition poses a challenge, opportunities exist in developing novel delivery systems, combination therapies, and exploring expanded indications to sustain market growth.
Frequently Asked Questions
-
What is the primary advantage of budesonide over other oral corticosteroids? Budesonide's primary advantage is its high first-pass metabolism, which results in significantly lower systemic bioavailability and reduced systemic side effects compared to traditional oral corticosteroids when administered orally, particularly in gastrointestinal applications.
-
How does patent expiration impact the budesonide market? Patent expiration leads to the entry of generic manufacturers, increasing competition and typically resulting in lower prices for budesonide products. However, this also enhances market accessibility and volume. Companies continue to innovate through new formulations and delivery devices to secure market exclusivity beyond original composition of matter patents.
-
Are there significant differences in the efficacy of inhaled versus oral budesonide? Yes, efficacy is highly dependent on the indication. Inhaled budesonide is highly effective for airway inflammation in asthma and COPD. Oral budesonide is designed for localized action within the gastrointestinal tract for IBD, with its efficacy tied to targeted release and absorption in specific intestinal segments. Systemic efficacy differs significantly due to formulation and administration routes.
-
What are the main safety concerns associated with long-term budesonide use? Long-term use, especially at higher doses, can lead to typical corticosteroid-related side effects, including adrenal suppression, decreased bone mineral density (osteoporosis), increased risk of infection, metabolic changes (e.g., hyperglycemia), and ophthalmological effects (e.g., cataracts, glaucoma). However, these risks are generally lower with inhaled and locally acting oral budesonide compared to systemic corticosteroids.
-
What is the trend in budesonide patenting strategy? The trend in budesonide patenting strategy has shifted from composition of matter to process patents, formulation patents, and novel drug delivery system patents. This strategy aims to create new intellectual property around improvements in product performance, patient convenience, and targeted delivery, extending market exclusivity and creating differentiation in a competitive landscape.
Citations
[1] World Health Organization. (2022). Asthma. https://www.who.int/news-room/fact-sheets/detail/asthma
More… ↓
