Last updated: February 20, 2026
What is the current market landscape for ALTACE?
ALTACE (ramipril), an angiotensin-converting enzyme (ACE) inhibitor, primarily treats hypertension and heart failure. Launched in 1992 by Novartis, it remains a significant component in cardiovascular therapy.
The global ACE inhibitor market was valued at approximately USD 12 billion in 2022, with ALTACE holding a moderate share. Competitive offerings include drugs like lisinopril, enalapril, and ramipril generics, which pressure ALTACE's market penetration.
How does the competitive environment impact sales outlook?
Generic competition affects ALTACE’s market share. The patent for ALTACE expired in 2005 in most regions, resulting in increased generic availability. Generic versions significantly lower prices, reducing branded drug revenue.
In 2022, the U.S. accounted for approximately 40% of the global market, driven by high hypertension prevalence (about 45% of adults aged 60+ have high blood pressure). Europe accounts for about 35%, with emerging markets making up the rest.
What are the regulatory and clinical factors influencing ALTACE's sales?
Clinical trials have shown ALTACE's efficacy in reducing cardiovascular mortality, especially in post-myocardial infarction and chronic heart failure settings. However, newer ACE inhibitors and angiotensin receptor blockers (ARBs) have cleared some market space.
Regulatory authorities, like the FDA and EMA, approve ALTACE for hypertension and certain heart failure indications. Off-label use and physician preference favor other therapies, influencing sales.
What are current and projected sales figures?
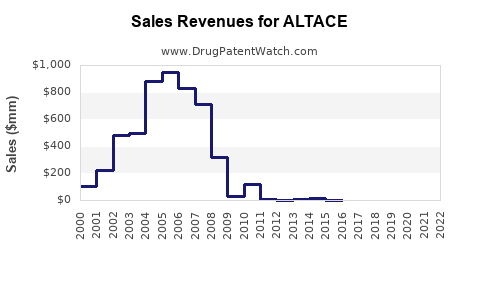
| Historical sales (USD millions): |
Year |
Sales (USD millions) |
Notes |
| 2015 |
250 |
Stable but declining due to generics |
| 2018 |
210 |
Market share losses to competitors |
| 2020 |
180 |
COVID-19 impacted prescription volume |
| 2022 |
170 |
Continued decline, increased generics |
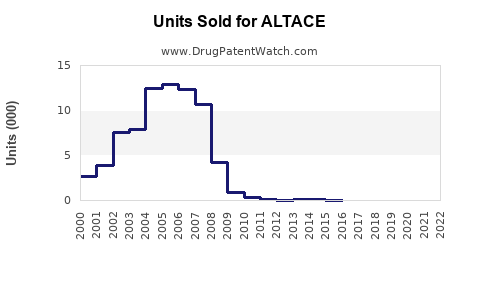
Market share estimates:
- Branded ALTACE: 15%-20% of ramipril prescriptions
- Generics: 80%-85%
Forecasts:
Based on market data and competitive trends, ALTACE sales are projected to decline at a compound annual growth rate (CAGR) of approximately -2% from 2023 to 2027. This reflects ongoing generic erosion and increased competition from newer therapeutics.
What factors could alter the sales trajectory?
- Introduction of new formulations or delivery methods.
- Expanded labeling for additional indications.
- Entry into emerging markets with growing hypertension prevalence.
- Patent litigation or regulatory changes affecting generic entry.
- Increased physician acceptance due to new clinical data.
How do emerging market dynamics influence future sales?
Emerging markets exhibit rising hypertension rates, with Asia-Pacific expected to account for over 50% of the global hypertensive population by 2030. Lower drug prices and expansion of healthcare infrastructure could boost ALTACE's sales in these regions.
However, availability of lower-cost generics may limit premium pricing for branded ALTACE. Local regulatory hurdles and market access challenges may also cap growth.
Summary of key factors affecting sales projections
| Factor |
Impact |
Notes |
| Generic competition |
Decline |
Main driver of sales reduction |
| Clinical trial data |
Neutral |
No recent data favoring ALTACE specifically |
| Market expansion |
Moderate |
Emerging markets present opportunities |
| Regulatory environment |
Mixed |
Patent status and regulation influence availability |
Key Takeaways
- ALTACE's global sales have declined from USD 250 million in 2015 to approximately USD 170 million in 2022.
- The drug faces intense generic competition, leading to a projected CAGR of approximately -2% from 2023 to 2027.
- The primary markets are North America and Europe, with high growth potential in Asia-Pacific.
- Future sales depend on market access, regulatory decisions, and physician prescribing patterns.
5 FAQs
1. What prevents ALTACE from maintaining higher sales?
Generic competition and clinical preference for ARBs reduce branded ALTACE prescriptions.
2. Are there opportunities for ALTACE in emerging markets?
Yes, increased hypertension prevalence and expanding healthcare infrastructure provide growth potential, though price sensitivity limits premium sales.
3. How does ALTACE compare to other ACE inhibitors?
Efficacy is comparable; however, newer agents with improved safety profiles and dosing convenience often dominate prescribing habits.
4. Could new clinical data reverse sales decline?
Potentially, if data demonstrate superior efficacy or safety, it may influence physician choice.
5. What regulatory factors could change ALTACE’s sales outlook?
Patent extensions, patent litigations, or regulatory restrictions on generics could impact availability and pricing.
References
[1] Market Research Future. (2023). ACE inhibitors market forecast.
[2] IQVIA. (2022). Pharmaceutical sales data report.
[3] World Health Organization. (2022). Hypertension prevalence report.
[4] Novartis. (2022). ALTACE product information.