Share This Page
Drug Sales Trends for ADDERALL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ADDERALL (2008)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
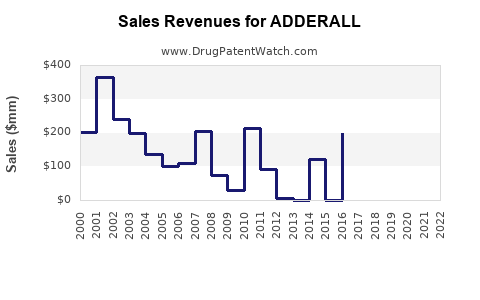
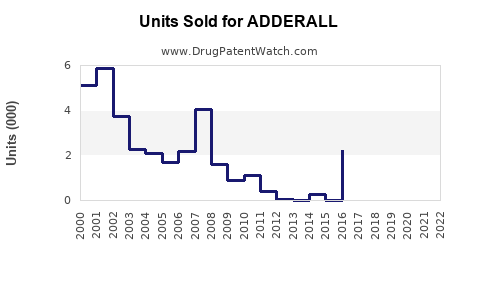
Annual Sales Revenues and Units Sold for ADDERALL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ADDERALL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ADDERALL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ADDERALL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Adderall Market Analysis and Sales Projections
Adderall, a combination of amphetamine and dextroamphetamine, is a Schedule II controlled substance prescribed for Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy. Its market presence is characterized by established brand loyalty, a broad patient population, and ongoing patent landscape considerations.
What is the Current Market Share and Revenue for Adderall?
The Adderall market is dominated by its branded version, Shire's Adderall XR (extended-release), and a significant generic segment. In 2022, the global market for ADHD medications, which Adderall is a major component of, was valued at approximately $22.7 billion and is projected to reach $36.1 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.9% [1].
While specific, up-to-the-minute revenue figures for Adderall alone are often consolidated within broader ADHD medication market reports, the brand has historically been a significant revenue driver. For instance, Shire (now Takeda Pharmaceutical Company) reported substantial sales from Adderall XR prior to patent expirations, consistently ranking it among the top ADHD treatments [2]. The generic market, while fragmenting revenue, ensures continued widespread access and volume sales.
Key Revenue Contributors and Market Dynamics:
- Branded Adderall XR: Maintained premium pricing and significant market share for years due to its extended-release formulation and established efficacy.
- Generic Adderall: Offers cost-effective alternatives, driving patient access and prescription volume, though at lower price points per unit.
- Regional Variations: Market penetration and sales performance are influenced by healthcare systems, regulatory approvals, and prescriber habits in different countries.
What is the Patent Landscape for Adderall?
The original patents for Adderall have long expired, allowing for the widespread production of generic versions. However, the patent landscape is dynamic, with new patent filings related to manufacturing processes, formulations, and new therapeutic uses.
Timeline of Key Patent Expirations and Developments:
- Original Composition of Matter Patents: Expired in the early 2000s [3].
- Adderall XR Patents: Key patents for the extended-release formulation began expiring in the late 2000s and early 2010s, paving the way for generic competition [4].
- Process Patents and Formulation Improvements: Manufacturers may hold patents on specific manufacturing methods or novel delivery systems that can extend market exclusivity for certain formulations.
Current Patent Considerations:
- Leva-Methylphenidate Patents: While not directly Adderall, the patent landscape for related ADHD medications, such as the extended-release formulation of methylphenidate (e.g., Quillivant XR), can influence market dynamics and competitive strategies.
- Patent Litigation: Disputes over existing patents or the validity of new patents can impact market entry timelines for new generic or improved brand formulations.
What are the Projected Sales for Adderall and its Generics?
Projecting sales for a drug with a mature branded product and a substantial generic presence involves analyzing the overall ADHD market growth, pricing trends, and the impact of competition.
Factors Influencing Future Sales:
- Increasing ADHD Diagnosis Rates: An upward trend in ADHD diagnoses, particularly in adults, is a primary driver of market growth [5].
- Market Saturation and Competition: The presence of numerous branded and generic ADHD medications creates intense competition, moderating price increases and potentially capping individual product sales growth.
- Controlled Substance Regulations: Stricter regulations and dispensing limitations for amphetamines can influence prescription volumes and market access.
- Emergence of New Treatments: Development of novel ADHD therapies, including non-stimulant options or innovative delivery systems, could impact Adderall's market share.
Sales Projection Scenarios (2024-2028):
Given the established nature of Adderall and the broad ADHD market growth, the combined sales of branded Adderall (primarily Adderall XR, where still marketed by Takeda) and its numerous generic equivalents are expected to see modest, steady growth.
- Base Case: Assuming current diagnostic trends and regulatory frameworks persist, the total market value attributed to Adderall and its generics is projected to grow at a CAGR of 4-6% over the next five years. This translates to a combined annual revenue increase of approximately $500 million to $1 billion globally within this segment.
- Growth Drivers: Increased awareness, improved diagnostic tools, and expanded access to treatment in developing markets will underpin this growth.
- Challenges: Pricing pressures from payers, potential shifts towards non-stimulant alternatives for specific patient profiles, and supply chain disruptions for controlled substances could temper growth.
What is the Competitive Landscape for Adderall?
Adderall competes within a crowded therapeutic space for ADHD medications, primarily against other stimulant and non-stimulant drugs.
Key Competitors:
- Methylphenidate-based Stimulants:
- Ritalin (novartis) / Concerta (Janssen Pharmaceuticals): Extended-release methylphenidate formulations.
- Daytrana (Novartis): Transdermal methylphenidate patch.
- Quillivant XR (Viatris): Liquid extended-release methylphenidate.
- Other Amphetamine-based Stimulants:
- Vyvanse (Takeda Pharmaceutical Company): Lisdexamfetamine dimesylate, a prodrug of dextroamphetamine, known for its longer duration of action and lower abuse potential.
- Evekeo (Mallinckrodt Pharmaceuticals): Amphetamine sulfate tablets.
- Adzenys XR-ODT (Neos Therapeutics): Orally disintegrating tablet formulation of amphetamine.
- Non-Stimulant Medications:
- Strattera (Eli Lilly and Company): Atomoxetine, a selective norepinephrine reuptake inhibitor.
- Intuniv (Shire/Takeda): Extended-release guanfacine.
- Kapvay (Supernus Pharmaceuticals): Extended-release clonidine.
Competitive Advantages and Disadvantages of Adderall:
- Advantages:
- Established Efficacy: Decades of clinical use and broad physician familiarity.
- Dual Mechanism: Combination of amphetamine and dextroamphetamine offers a specific pharmacokinetic profile.
- Formulation Options: Availability of both immediate-release and extended-release (XR) formulations.
- Disadvantages:
- Controlled Substance Status: Subject to strict prescribing and dispensing regulations, potentially limiting access and increasing administrative burden.
- Abuse Potential: Higher risk of misuse and diversion compared to non-stimulants.
- Side Effect Profile: Common side effects include appetite suppression, insomnia, and cardiovascular effects.
- Generic Erosion: Significant price erosion due to generic competition.
What are the Regulatory and Supply Chain Considerations for Adderall?
As a Schedule II controlled substance, Adderall is subject to stringent regulations by agencies like the U.S. Drug Enforcement Administration (DEA) and equivalent bodies internationally.
DEA Quotas and Supply:
The DEA sets annual aggregate production quotas for Schedule II substances, including amphetamine and its derivatives, to prevent diversion and abuse. These quotas directly impact the manufacturing volume of Adderall and its generic equivalents.
- Impact of Quotas: Fluctuations in DEA quotas can lead to supply shortages, affecting patient access and market availability. Recent years have seen reports of widespread Adderall shortages [6].
- Manufacturing Challenges: Pharmaceutical companies producing Adderall must navigate complex manufacturing processes and comply with strict security and tracking requirements.
International Regulations:
While the U.S. market is the largest, Adderall and similar amphetamine-based stimulants are regulated differently in other countries regarding prescribing practices, import/export controls, and availability.
Recent Developments:
- FDA and DEA Actions: In response to reported shortages, both the Food and Drug Administration (FDA) and the DEA have taken steps to address manufacturing and supply chain issues, including potentially increasing quotas and working with manufacturers to ramp up production [7].
- Formulation-Specific Regulations: While the active pharmaceutical ingredients are regulated under controlled substance laws, specific dosage forms or extended-release technologies may be subject to additional FDA scrutiny and approval processes.
Key Takeaways
- The global ADHD medication market, a primary segment for Adderall, is projected for continued growth, driven by increasing diagnoses.
- Original Adderall patents have expired, leading to a competitive generic market. New patent filings focus on manufacturing processes and formulations.
- Combined sales of branded Adderall and its generics are anticipated to grow modestly, influenced by diagnostic trends, competition, and regulatory controls.
- Adderall faces intense competition from other stimulant and non-stimulant ADHD medications.
- As a Schedule II controlled substance, Adderall is subject to strict DEA quotas and regulations, which have historically led to supply shortages and impact manufacturing volumes.
Frequently Asked Questions
-
What is the primary indication for Adderall? Adderall is primarily prescribed for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy.
-
Has the patent exclusivity for Adderall expired? Yes, the core patents for the original composition of matter and the extended-release formulation (Adderall XR) have expired, allowing for generic production.
-
What regulatory body controls the supply of Adderall in the United States? The U.S. Drug Enforcement Administration (DEA) sets annual production quotas for Schedule II controlled substances like Adderall.
-
What are the main competitive stimulants to Adderall? Key competitors include Ritalin, Concerta, and Vyvanse.
-
Are there any significant ongoing shortages of Adderall? Yes, reports of Adderall shortages have been recurrent in recent years, impacting availability for patients.
Citations
[1] Grand View Research. (2023). ADHD Medications Market Size, Share & Trends Analysis Report By Drug Class (Stimulants, Non-stimulants), By Disease Type (ADHD in Children, ADHD in Adults), By Distribution Channel, By Region, And Segment Forecasts, 2023-2030.
[2] Shire plc. (2010-2016). Annual Reports. (Specific figures vary by year and are publicly available in SEC filings).
[3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
[4] Skuza, R. (2009, April 13). Generic Adderall XR Launched. Drug Topics.
[5] Centers for Disease Control and Prevention. (2020, September 29). Attention-Deficit/Hyperactivity Disorder (ADHD) Data & Statistics.
[6] U.S. Food & Drug Administration. (2022, October 19). FDA Addresses Drug Shortages.
[7] U.S. Drug Enforcement Administration. (2023, January 26). DEA Statement on Amphetamine Shortage.
More… ↓
