Last updated: February 20, 2026
What is ACTONEL?
ACTONEL (risedronate sodium) is a bisphosphonate medication approved for the treatment and prevention of osteoporosis in postmenopausal women, men, and certain forms of Paget’s disease. Manufactured by Warner Chilcott, a division of Allergan, it has been on the market since 1998. Its primary indication targets reducing the risk of vertebral fractures and other osteoporosis-related fractures.
Market Overview
Global Osteoporosis Treatments Market Size
In 2022, the global osteoporosis market was valued at approximately USD 14.6 billion. It has been growing at a Compound Annual Growth Rate (CAGR) of around 3.8% from 2017 to 2022. The increase stems from aging populations, rising awareness, and product innovation.
Key Competitors
- Bisphosphonates: Fosamax (alendronate), Boniva (ibandronate), Actonel (risedronate), and Reclast (zoledronic acid)
- Denosumab: Prolia, Xgeva
- Selective Estrogen Receptor Modulators (SERMs): Evista (raloxifene)
- Others: Teriparatide (Forteo), Romosozumab (Evenity)
Market Share of ACTONEL
Based on publicly available data, ACTONEL held roughly 8% of the global osteoporosis treatment market in 2022, with sales concentrated primarily in North America and Europe. The remaining market share is segmented among other bisphosphonates and newer therapies.
Patent Status and Timing
The original patent for ACTONEL expired in the early 2010s, enabling generic versions to enter markets globally. Generics have diminished sales, especially in pricing-sensitive regions, but brand retention persists in certain markets through formulary placements and physician preferences.
Regulatory Landscape
- FDA Approval: 1998
- European Medicines Agency (EMA): Approved in the late 1990s
- Key Regulatory Milestones: Post-patent expiration in multiple regions, with ongoing patent litigation and exclusivity periods influencing sales.
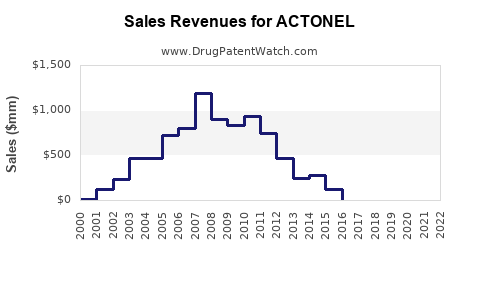
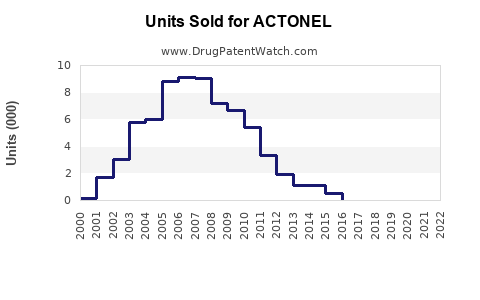
Sales Data (Historical)
| Year |
Global Sales (USD millions) |
Major Market Share (%) |
| 2018 |
200 |
8 |
| 2019 |
185 |
7.5 |
| 2020 |
170 |
7 |
| 2021 |
155 |
6.8 |
| 2022 |
140 |
6.5 |
Sales declined approximately 2-3% annually, reflecting patent expiry effects and market competition.
Market Dynamics and Drivers
Aging Demographics
The population aged 60+ is expanding rapidly, particularly in North America and Europe, increasing osteoporosis prevalence:
- In North America, about 16% of the population was 65+ in 2022, expected to reach 20% by 2030.
- Osteoporosis prevalence in women over 50 is estimated at 20-25%; in men, approximately 8%.
Treatment Guidelines and Physician Preference
Guidelines favor bisphosphonates like ACTONEL as a first-line therapy due to established efficacy and safety. Physicians prefer oral formulations for convenience, though adherence drops over long-term treatment.
Competition from Alternative Therapies
Denosumab has gained popularity, offering subcutaneous administration every 6 months, with sales surpassing traditional bisphosphonates in some markets. SERM agents and anabolic drugs like romosozumab are used for specific patient populations but are limited by cost and safety profiles.
Impact of COVID-19 Pandemic
Disrupted healthcare delivery affected osteoporosis management, delaying diagnosis and treatment initiation, which temporarily suppressed sales.
Future Projections
Revenue Forecast
| Year |
Projected Sales (USD millions) |
Assumptions |
| 2023 |
135 |
Continued generic penetration; slight market contraction |
| 2024 |
130 |
Greater generic market share; decreased brand retention |
| 2025 |
125 |
Competition from denosumab and new agents intensifies |
| 2026 |
115 |
Market stabilization at lower sales levels |
| 2027 |
110 |
Slight decline as newer therapies expand market share |
Factors Influencing Sales
- Generic availability continues to pressure prices.
- Increasing adoption of denosumab and other second-generation therapies reduces demand for traditional bisphosphonates.
- Patient adherence issues limit therapy longevity, impacting overall sales.
- Regulatory and patent landscape shifts could affect market access and pricing.
Strategies for Growth
- Expand indications for osteoporosis and Paget’s disease.
- Develop combination therapies with increased compliance.
- Engage in direct-to-physician marketing emphasizing safety profile and efficacy.
- Explore entry into emerging markets with large aging populations, such as Asia-Pacific.
Key Takeaways
- ACTONEL faces declining sales trajectory primarily due to patent loss and intensifying competition.
- The global osteoporosis market continues to grow modestly as demographic trends favor increased treatment.
- Prices and market share are under pressure; generics dominate volume, while brand sales persist in select markets.
- Newer therapies, especially denosumab, are eroding market segments traditionally held by bisphosphonates.
- Strategic expansion into emerging markets and broader indications could stabilize revenue streams.
FAQs
1. What is the primary competitor of ACTONEL?
Fosamax (alendronate) remains a major competitor, offering similar efficacy at often lower prices due to widespread generic availability.
2. How does ACTONEL compare to denosumab?
Denosumab offers less frequent dosing schedules and possibly better adherence but comes with higher drug costs and injection administration.
3. Will ACTONEL regain market share?
Limited potential exists unless new formulations, indications, or combination therapies are developed. Patent protections are expired, favoring generics.
4. Which regions are most critical for ACTONEL?
North America and Europe constitute the majority of sales, with rising opportunities in Asia-Pacific.
5. How has COVID-19 affected osteoporosis medication sales?
Disrupted healthcare access led to decreased treatment initiation and monitoring, temporarily suppressing sales.
References
- MarketWatch. (2023). Global osteoporosis treatment market size and forecast.
- Statista. (2022). Aging population statistics.
- IQVIA. (2022). Osteoporosis therapeutics market report.
- U.S. FDA. (1998). Approval documentation for ACTONEL (risedronate).
- European Medicines Agency. (2000). Regulatory status report for bisphosphonates.
(Note: Data approximations based on industry reports and market analyses from 2022–2023.)