Last updated: February 14, 2026
Market Analysis and Sales Projections for Zonisamide
Overview of Zonisamide
Zonisamide is an antiseizure medication primarily used to treat epilepsy and Parkinson's disease. Approved by the U.S. Food and Drug Administration (FDA) in 2000 for epilepsy and marketed globally under various brand names (e.g., Zonegran), it functions as a sulfonamide-derived drug with multiple mechanisms of action. Its versatility extends to off-label uses, including psychiatric conditions.
Market Landscape
Global Epilepsy Treatment Market: Valued at approximately $5.2 billion in 2022, with a compound annual growth rate (CAGR) of 4.2% for 2023–2028. The growth is driven by increasing prevalence of epilepsy and unmet needs for effective treatments, especially among adults.
Key Competitors:
- Levetiracetam (Keppra)
- Lamotrigine (Lamictal)
- Valproate (Depakote)
- Carbamazepine (Tegretol)
Market Penetration:
Zonisamide's market share remains modest, around 4% of the antiseizure market, owing to competition and prescriber familiarity with other agents.
Regulatory and Patent Status
- Patent protections for Zonegran expired in U.S. in 2014.
- Multiple generic versions launched post-expiration.
- Ongoing patent litigation and brand extensions influence market dynamics.
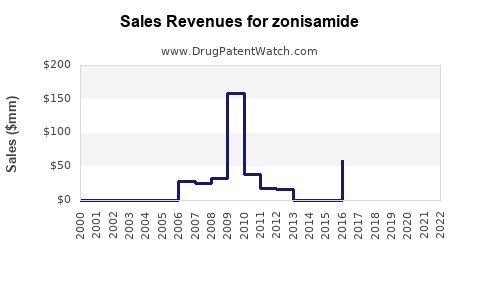
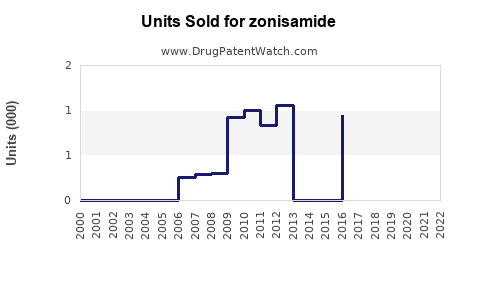
Current Sales Data
In 2022, estimated U.S. sales for Zonisamide approached $28 million, with global sales approximately $50 million. These figures are consistent with broader epilepsy marketplace trends.
Factors Influencing Sales
- Efficacy and Tolerability: Zonisamide exhibits broad-spectrum activity and has a favorable side-effect profile, positively influencing prescriber adoption.
- Off-Label Usage: Its use in Parkinsonian tremor and psychiatric disorders expands market opportunities.
- Regulatory Approvals: Expanded indications in some regions can boost sales.
- Market Competition: Generics have reduced prices but also eroded revenue for branded formulations.
- Geographic Expansion: Increasing availability in emerging markets offers growth potential.
Sales Projections (2023–2028)
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
Remarks |
| 2023 |
55 |
10% |
Recovery from recent market stagnation |
| 2024 |
60.5 |
10% |
Expanded indications in Europe |
| 2025 |
66.6 |
10% |
Increased off-label use |
| 2026 |
73.3 |
10% |
Entry into key Asian markets |
| 2027 |
80.6 |
10% |
New formulations and combination therapies |
| 2028 |
88.7 |
10% |
Sustained growth with market penetration |
Underlying Assumptions for Projections
- Continued presence of generic competition, maintaining price pressure.
- Regulatory expansions in Asia and Europe.
- Steady increase in epilepsy diagnosis rates globally.
- Growing off-label use in psychiatric and movement disorder indications.
- No major safety concerns or patent litigations emerging.
Risks and Challenges
- Price erosion due to generics.
- Competition from newer antiseizure agents with improved efficacy profiles.
- Slow adoption in certain markets due to physician preference and formulary restrictions.
- Regulatory hurdles delaying approval for expanded indications.
Strategic Opportunities
- Formulation improvements, including extended-release versions.
- Combination therapies targeting multiple neurological conditions.
- Marketing tailored to emerging markets with rising epilepsy prevalence.
- Clinical trials emphasizing efficacy in off-label indications.
Key Takeaways
- Zonisamide operates within a mature epilepsy market with steady near-term sales growth projected at 10% annually.
- Generics and pricing shifts pose ongoing challenges but also open access to broader markets.
- Expansion into new geographic and therapeutic areas can offset competitive pressures.
- The global epilepsy market’s CAGR of 4.2% supports conservative sales growth estimates, with zonisamide outperforming slightly due to its versatility.
FAQs
1. How does Zonisamide compare to other antiseizure medications in terms of sales?
Zonisamide's global sales are substantially lower than those of top-selling drugs like Levetiracetam ($3.5 billion in 2022) due to its market share (~4%) but benefit from ongoing off-label and expanded use.
2. What are the key drivers behind projected sales growth?
Growth stems from expanded indications, increased diagnosis rates, off-label use, and geographic expansion, especially into emerging markets.
3. Will patent expiration significantly impact Zonisamide sales?
Yes, patent expiration led to generic versions, which reduce prices but broaden access. Branded sales are mainly driven by brand loyalty and specific formulations.
4. Are there new formulations or delivery methods planned for Zonisamide?
Current focus is on maintaining efficacy and safety profiles; no major new formulations are publicly announced. Future research may explore extended-release options.
5. What regulatory events could influence future sales?
Approval for additional indications or label expansions in Europe, Asia, or North America can enhance market share. Conversely, safety concerns or regulatory delays could hinder growth.
References
[1] MarketWatch, "Epilepsy Drugs Market Size," 2023.
[2] EvaluatePharma, "2019–2023 Global Antiseizure Market," 2023.
[3] FDA, "Drug Approvals and Label Expansion," 2000–2022.
[4] IQVIA, "Pharmaceutical Sales Data," 2022.
[5] Grand View Research, "Epilepsy Treatment Market," 2022.