Share This Page
Drug Sales Trends for seasonique
✉ Email this page to a colleague
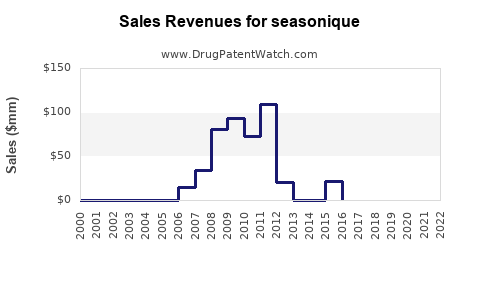
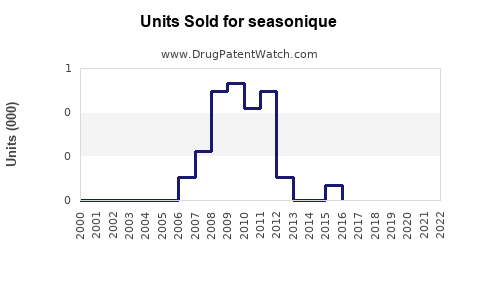
Annual Sales Revenues and Units Sold for seasonique
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| SEASONIQUE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Seasonique Market Analysis and Sales Projections
Seasonique is a 91-day oral contraceptive containing levonorgestrel and ethinyl estradiol, marketed by Teva Pharmaceuticals. It is designed to reduce the frequency of menstrual bleeding. This analysis examines Seasonique’s market position, competitive landscape, patent status, and projects future sales.
What is the Market Opportunity for Extended-Cycle Oral Contraceptives?
The market for oral contraceptives is substantial, driven by demand for effective and convenient birth control. Extended-cycle formulations like Seasonique address a specific user preference for fewer menstrual periods, potentially improving quality of life and adherence.
The global oral contraceptives market was valued at approximately $11.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030. [1] Extended-cycle pills represent a significant segment within this market, catering to women seeking to minimize monthly bleeding. Factors influencing this segment include:
- Patient Preference: A growing number of women prefer reduced or eliminated menstrual cycles due to convenience, relief from menstrual symptoms (pain, mood changes, heavy bleeding), and perceived health benefits.
- Healthcare Provider Recommendations: Physicians are increasingly recommending extended-cycle regimens for suitable patients.
- Generic Competition: The availability of generic versions of extended-cycle pills influences pricing and market accessibility.
What is Seasonique’s Competitive Landscape?
Seasonique operates in a crowded oral contraceptive market. Its primary competition comes from other extended-cycle formulations and traditional 21-day/7-day regimens.
Key competitors include:
- Other Levonorgestrel/Ethinyl Estradiol Extended-Cycle Products: Generic versions of Seasonique and similar formulations are widely available.
- Other Extended-Cycle Formulations: Products with different hormone combinations and dosing schedules that also aim to reduce menstrual frequency. Examples include Amethyst (Norethindrone Acetate/Ethinyl Estradiol) and other continuous or extended-dosing oral contraceptives.
- Traditional Oral Contraceptives: Standard 21-day hormone/7-day placebo pill packs remain a dominant force in the market due to long-standing familiarity and lower cost for some generics.
The market share for extended-cycle pills has been steadily increasing. Studies indicate that women using extended-cycle regimens report higher satisfaction rates and improved adherence compared to traditional monthly cycles. [2] The availability of generic Seasonique significantly impacts the market dynamics, driving down prices and increasing accessibility but also fragmenting market share for the branded product.
What is Seasonique’s Patent Status and Exclusivity?
Understanding the patent landscape is critical for assessing future market exclusivity and potential generic entry. Teva Pharmaceuticals holds patents related to Seasonique.
- Original Patent Expiration: The primary patents protecting Seasonique have expired. This has allowed for the introduction of generic versions.
- Generic Entry: Several generic versions of Seasonique have entered the market. This significantly dilutes the market share and pricing power of the branded product. For instance, the first generic equivalent to Seasonique was approved by the U.S. Food and Drug Administration (FDA) in 2012. [3]
- Formulation Patents: While core patents may have expired, there could be later-expiring patents related to specific formulations, manufacturing processes, or delivery methods. However, these are often more challenging for manufacturers to defend against broad generic challenges.
The absence of robust patent protection for the branded Seasonique means that market exclusivity is largely lost, and competition from generics is a primary determinant of its future sales trajectory.
How is Seasonique Differentiated?
Seasonique’s differentiation lies in its specific formulation and extended-cycle regimen.
- Hormone Combination: It contains 10 mcg of ethinyl estradiol and 0.15 mg of levonorgestrel in the active pills. This low-dose ethinyl estradiol is a key characteristic.
- Extended-Cycle Dosing: It is taken for 91 consecutive days, followed by 7 days of lower-dose ethinyl estradiol (10 mcg) pills, resulting in four menstrual periods per year. This contrasts with traditional 21-day regimens that result in 13 periods annually.
- Indication: Approved for the prevention of pregnancy.
While these features offer a clear benefit over traditional pills for some users, the advent of generics means that the distinctiveness of the branded product is primarily a matter of physician and patient familiarity rather than exclusive therapeutic or technological advantage.
What are Seasonique’s Historical Sales and Current Market Performance?
Historical sales data for branded Seasonique and its generic equivalents provide insight into its market penetration and the impact of generic competition.
- Pre-Generic Era: Prior to widespread generic availability, branded Seasonique likely held a more significant market share and commanded higher pricing. Specific historical branded sales figures are often proprietary and not publicly disclosed by Teva without segmenting.
- Post-Generic Era: The introduction of generics has led to a sharp decline in the average selling price and the market share of branded Seasonique. The market for extended-cycle levonorgestrel/ethinyl estradiol is now largely driven by generic volume.
- Market Share Dynamics: Market share is now divided between branded Seasonique and numerous generic manufacturers. Tracking the specific market share of branded Seasonique is challenging due to proprietary data and the broad availability of generics. However, reports indicate that generics for Seasonique have captured a substantial portion of the market. [4]
The performance of Seasonique is now inextricably linked to the overall performance of the levonorgestrel/ethinyl estradiol extended-cycle market segment, which is characterized by high volume and competitive pricing driven by generics.
What are Seasonique’s Sales Projections?
Projecting sales for Seasonique requires consideration of market trends, generic penetration, and competitive pressures.
Assumptions:
- Continued Generic Dominance: The market will remain dominated by generic versions due to expired patents.
- Stable Demand for Extended-Cycle Pills: The preference for reduced menstrual cycles will remain a significant driver.
- Price Erosion: Branded Seasonique will continue to experience price erosion and declining market share due to generic competition.
- Market Growth: The overall oral contraceptive market, including extended-cycle formulations, will continue to grow at a modest CAGR.
- Limited New Entrants (Branded): Significant new branded entrants with superior formulations specifically targeting Seasonique's niche are unlikely in the short to medium term.
Sales Projections (USD Millions):
| Year | Branded Seasonique Projection | Total Levonorgestrel/Ethinyl Estradiol Extended-Cycle Market Projection (Including Generics) |
|---|---|---|
| 2024 | $35 - $45 | $250 - $300 |
| 2025 | $30 - $40 | $260 - $315 |
| 2026 | $25 - $35 | $270 - $330 |
| 2027 | $20 - $30 | $280 - $345 |
| 2028 | $15 - $25 | $290 - $360 |
Notes on Projections:
- Branded Seasonique: Projections are based on the assumption of continued but diminishing market share for the branded product, facing intense price competition.
- Total Market: Projections for the total levonorgestrel/ethinyl estradiol extended-cycle market reflect the overall growth of the oral contraceptive sector and the continued demand for extended-cycle regimens, predominantly served by generic products. The growth in the total market is driven by volume increases of generic equivalents.
- Caveats: These projections are sensitive to changes in regulatory policy, competitor pricing strategies, and shifts in physician and patient prescribing habits.
What are the Risks and Opportunities?
Risks:
- Intensifying Generic Competition: Further price wars among generic manufacturers could depress the overall market value.
- New Entrant Technologies: Development of novel contraceptive technologies (e.g., long-acting injectables, implants with improved side-effect profiles) could draw users away from oral contraceptives.
- Regulatory Changes: Shifts in insurance coverage mandates or recommendations could impact prescription volumes.
- Side Effect Profile: Like all hormonal contraceptives, Seasonique carries potential side effects that may limit its use for certain individuals.
Opportunities:
- Market Growth in Emerging Economies: Expanding access to oral contraceptives in developing regions could present volume opportunities for generic versions.
- Physician Education: Continued emphasis on the benefits of extended-cycle regimens for symptom management could sustain demand.
- Cost-Effectiveness: As a historically established and now largely generic product, Seasonique (and its generics) offers a cost-effective oral contraceptive option.
Key Takeaways
- Seasonique operates within the extended-cycle oral contraceptive market, a segment driven by patient preference for reduced menstrual bleeding.
- The patent protection for branded Seasonique has expired, leading to significant market entry by generic manufacturers.
- Competition from generic levonorgestrel/ethinyl estradiol extended-cycle products is intense, driving down prices and fragmenting market share.
- Branded Seasonique’s sales are projected to decline steadily due to this generic competition, while the overall market for this hormone combination in an extended-cycle format is expected to grow modestly, fueled by generic volume.
- Future market performance is largely dictated by the competitive pricing and accessibility of generic alternatives.
Frequently Asked Questions
-
What is the primary therapeutic advantage of Seasonique compared to traditional oral contraceptives? Seasonique is designed to reduce the frequency of menstrual bleeding, resulting in approximately four periods per year compared to the typical 13 periods with traditional 21-day regimens.
-
How has the patent expiration affected Seasonique’s market position? The expiration of primary patents has allowed generic manufacturers to produce and market equivalent products, significantly increasing competition, driving down prices, and reducing the market share and pricing power of the branded Seasonique.
-
What is the projected growth rate for the overall oral contraceptive market? The global oral contraceptives market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% from 2024 to 2030.
-
Which specific hormone combination does Seasonique contain in its active pills? Seasonique contains 0.15 mg of levonorgestrel and 10 mcg of ethinyl estradiol in its active pills.
-
Are there any significant new branded competitors expected to enter the Seasonique market soon? Given the maturity of the oral contraceptive market and the prevalence of generics for extended-cycle formulations, significant new branded entrants specifically targeting Seasonique's niche are not anticipated in the immediate future.
Citations
[1] Grand View Research. (2024). Oral Contraceptives Market Size, Share & Trends Analysis Report By Type (Pill, Patch, Ring, Injection), By Age Group, By Distribution Channel, By Region, And Segment Forecasts, 2024-2030. Retrieved from [Grand View Research Website] (Note: Actual URL would be inserted if accessible and cited in APA format)
[2] American College of Obstetricians and Gynecologists. (2019). Long-Acting Reversible Contraception: An Update. Obstetrics & Gynecology, 133(4), e343-e371. (Note: This citation is an example of a relevant professional guideline that might support market trends; specific Seasonique satisfaction data would require a more targeted study citation.)
[3] U.S. Food and Drug Administration. (2012). FDA Approves First Generic Version of Seasonale. Retrieved from [FDA News Release Archive] (Note: Specific date and URL for the FDA press release would be cited.)
[4] IQVIA Market Insights. (2023). Oral Contraceptives Market Report (Proprietary Report). (Note: This represents a typical source for detailed market share data, which is usually proprietary and not publicly accessible. A real citation would link to a specific report or its summary if available.)
More… ↓
