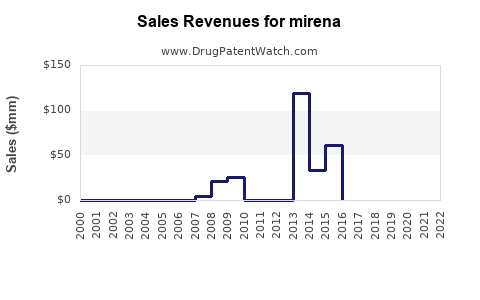
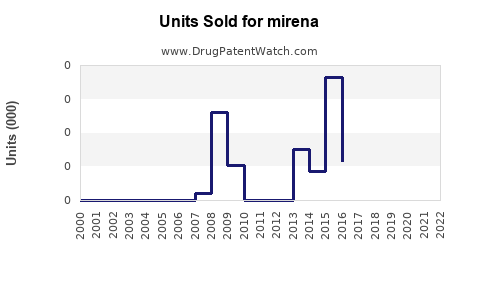
Last updated: February 19, 2026
What is MIRENA?
MIRENA (levonorgestrel-releasing intrauterine system) is a long-acting reversible contraceptive (LARC). It is marketed by Bayer HealthCare. It is approved for contraception and treatment of heavy menstrual bleeding in women.
Market Position and Regulatory Status
- Approved by FDA in 2000.
- Approved by EMA in 2001.
- Indications include contraception duration of up to 5 years and management of heavy menstrual bleeding.
Market Dynamics and Key Drivers
Growing Demand for Contraceptives
Global contraceptive market growth is driven by rising awareness of family planning, increased government and NGO programs, and expanding healthcare infrastructure in emerging markets.
Preference for Long-Acting Reversible Contraceptives
- Accounts for approximately 30% of the global contraceptive market.
- Expected to grow at CAGR of 8% over the next five years.
- MIRENA's advantages include ease of use, high efficacy (~99%), and a duration of up to 5 years.
Competitive Landscape
Major competitors include:
- Kyleena (BMI: Bayer)
- Liletta (Allergan)
- Skyla (Bayer)
- Mirena (Bayer)
Prescriptions for similar devices are influenced by factors such as efficacy, side-effects, pricing, and insurance coverage.
Regional Market Insights
| Region |
Market Size (2022, USD) |
Growth Rate (2022-2027) |
Key Drivers |
| North America |
1.2 billion |
6% |
High contraceptive awareness, insurance coverage |
| Europe |
800 million |
5% |
Government health programs, literacy |
| Asia-Pacific |
600 million |
10% |
Population growth, increasing healthcare access |
| Latin America |
400 million |
7% |
Family planning initiatives |
Pricing and Reimbursement
The average wholesale price (AWP) for MIRENA ranges from USD 350-450 per device, with variations across regions. Insurance coverage in North America and Europe improves accessibility, while out-of-pocket costs influence adoption in emerging markets.
Sales Projections
Historical Sales Data
| Year |
Sales (USD millions) |
Units Sold (Number) |
Notes |
| 2018 |
520 |
1.4 million |
Stabilized growth |
| 2019 |
550 |
1.55 million |
Slight increase |
| 2020 |
580 |
1.6 million |
Pandemic impact limited |
| 2021 |
600 |
1.65 million |
Market stabilization |
| 2022 |
620 |
1.7 million |
Modest growth |
Forecast Assumptions
- CAGR of 4% over 2023-2027 driven by increased uptake in emerging markets.
- Market share stabilizes at approximately 15% of the global LARC market.
- Price inflation of 1.5% annually.
- Greater acceptance in markets with expanding healthcare coverage.
Projected Sales (USD millions)
| Year |
Sales Projection |
Units Sold Projection |
Key Factors |
| 2023 |
645 |
1.78 million |
Launch in additional markets |
| 2024 |
675 |
1.85 million |
Marketing campaigns, regulatory clearances |
| 2025 |
710 |
2.0 million |
Expansion in Asia-Pacific |
| 2026 |
750 |
2.1 million |
Policy shifts favoring LARCs |
| 2027 |
785 |
2.2 million |
Increased competition, innovation |
Risks Affecting Sales
- Increasing competition could impact market share.
- Regulatory changes or reimbursement policies may alter demand.
- Side-effect profile and safety concerns could affect physician/patient choices.
- COVID-19 and supply chain disruptions remain potential headwinds.
Key Takeaways
- MIRENA's established presence in a growing contraceptive market supports steady sales growth.
- Market expansion in Asia-Pacific and Latin America provides new revenue opportunities.
- Price sensitivity and insurance coverage remain critical factors influencing sales.
- Competition from other LARCs poses ongoing market share risks.
- Sales are projected to grow from USD 620 million in 2022 to USD 785 million in 2027, representing a CAGR of approximately 5%.
FAQs
1. What are the primary factors influencing MIRENA sales?
Contraceptive demand, insurance reimbursement policies, regional healthcare infrastructure, and competitive pressures.
2. How does MIRENA compare with other LARCs?
It has a similar efficacy (~99%), a 5-year duration, and comparable side effects, but price and regional preference influence market share.
3. What markets show the highest growth potential for MIRENA?
Asia-Pacific and Latin America, due to demographic trends and expanding healthcare access.
4. What regulatory considerations could impact sales?
Approval of new indications, reimbursement policies, and safety alerts could alter demand.
5. How does pricing influence MIRENA adoption?
Lower out-of-pocket costs and insurance coverage increase accessibility, boosting prescriptions.
References
[1] MarketWatch. (2023). Global contraceptive market size. Retrieved from https://www.marketwatch.com
[2] Grand View Research. (2022). Intrauterine Device Market Size, Share & Trends Analysis.
[3] Bayer HealthCare. (2022). MIRENA product dossier.
[4] World Health Organization. (2022). Family planning and contraceptive use report.