Share This Page
Drug Sales Trends for methylphenidate
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for methylphenidate (2007)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
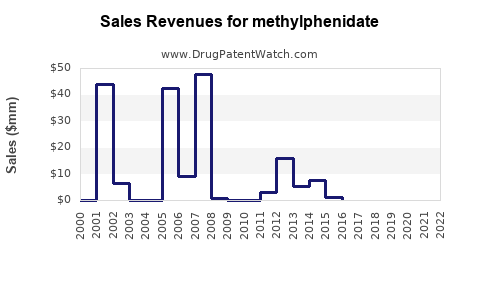
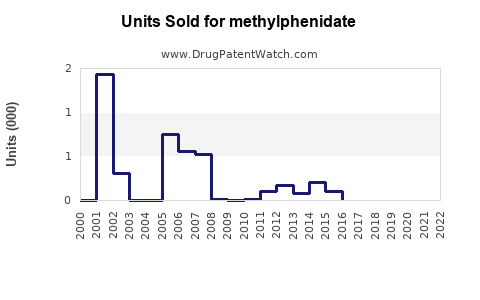
Annual Sales Revenues and Units Sold for methylphenidate
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METHYLPHENIDATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METHYLPHENIDATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METHYLPHENIDATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METHYLPHENIDATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Methylphenidate: Patent Landscape and Market Projections
Methylphenidate, a central nervous system stimulant primarily used to treat Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy, faces a mature market shaped by a complex patent landscape. While originator patents have expired, leading to generic competition, ongoing patent filings focus on novel formulations, delivery systems, and combinations. These developments aim to extend market exclusivity and address unmet patient needs. The global methylphenidate market is projected to experience moderate growth, driven by increasing ADHD diagnoses, improved diagnostic tools, and the development of extended-release formulations.
What is the Current Patent Landscape for Methylphenidate?
The patent landscape for methylphenidate is characterized by the expiration of core compound patents, primarily held by Novartis for Ritalin and its successors. This has opened the market to generic manufacturers, intensifying competition and driving down prices for immediate-release formulations. However, innovation has shifted towards new formulations and delivery mechanisms designed to improve patient compliance, reduce side effects, and provide sustained symptom control.
Key Areas of Patent Activity:
- Extended-Release Formulations: Patents cover various technologies for controlled and sustained release, including osmotic pumps, matrix systems, and multiparticulates. These aim to reduce dosing frequency and smooth drug delivery throughout the day.
- Novel Delivery Systems: Research and patent filings explore alternative delivery methods beyond oral tablets, such as transdermal patches and nasal sprays, to bypass first-pass metabolism and offer different pharmacokinetic profiles.
- Combination Therapies: Patents are being filed for methylphenidate in combination with other active pharmaceutical ingredients (APIs) to address specific symptom profiles or co-occurring conditions.
- Manufacturing Processes: Improvements and novel methods in the synthesis and purification of methylphenidate and its salts are also subject to patent protection.
- Pediatric Formulations: Development of taste-masked or easier-to-swallow formulations for pediatric patients remains an area of interest.
Notable Patent Expirations and Remaining Exclusivity:
The original patents for methylphenidate hydrochloride expired decades ago, allowing for widespread generic entry. For instance, the foundational patents for Ritalin® (methylphenidate hydrochloride) began expiring in the 1990s. However, subsequent innovations have led to new intellectual property.
- Concerta® (Osmotic Controlled-Release Oral Delivery System - OROS): While the core methylphenidate patent is expired, the OROS technology patent provided extended exclusivity for this extended-release formulation. Key patents related to the OROS technology have largely expired, but new patents may cover specific aspects of its manufacturing or variations.
- Other Extended-Release Products: Products like Daytrana® (transdermal patch) and Quillivant XR® (liquid suspension) have their own patent protection covering their specific delivery mechanisms. The patent expiry dates for these technologies will vary. For example, Daytrana's core patents began expiring around 2020-2022, with some potential for later expiry on manufacturing process patents [1].
As of current filings, patent extensions are sought for specific advancements, not the base molecule. For example, U.S. Patent No. 9,125,768, assigned to Shire, concerns a method for treating ADHD with methylphenidate, which may have been subject to patent term adjustments or extensions. The duration of protection for these newer patents typically ranges from 20 years from the filing date, subject to potential extensions for regulatory delays.
How is the Global Methylphenidate Market Structured?
The global methylphenidate market is a mature yet dynamic segment within the psychopharmacology sector. It is dominated by generic manufacturers for immediate-release formulations, while branded extended-release products and newer delivery systems hold significant market share due to their perceived advantages in patient management and efficacy.
Key Market Segments:
- Immediate-Release (IR) Formulations: These are typically lower-cost generic products that require multiple daily doses. They represent a substantial portion of the market volume due to their accessibility and affordability.
- Extended-Release (ER) Formulations: Branded and generic ER products offer once-daily dosing, improving patient adherence and providing more consistent symptom control. These command higher prices and are a key focus for market growth. Examples include Concerta®, Ritalin LA®, Daytrana®, and Quillivant XR®.
- Transdermal Patches: Products like Daytrana® offer an alternative delivery route, bypassing gastrointestinal absorption and allowing for dose titration and rapid removal if adverse effects occur.
- Liquid Formulations: These are particularly important for pediatric patients who have difficulty swallowing pills. Quillivant XR® and Adhansia XR® are examples of such products.
Major Players in the Market:
The market includes both originators who initially developed novel formulations and a large number of generic manufacturers.
- Originators/Branded Manufacturers:
- Novartis: Ritalin®, Concerta® (original developer of OROS technology).
- Shire (now Takeda): Introduced several methylphenidate formulations, including Daytrana®.
- Pfizer/Upjohn: Formerly involved with generic Ritalin®.
- Radius Health: Involved with Adhansia XR®.
- Generic Manufacturers: Numerous companies produce generic methylphenidate IR and ER tablets, including but not limited to:
- Teva Pharmaceutical Industries
- Mallinckrodt Pharmaceuticals
- Sun Pharmaceutical Industries
- Actavis (now part of AbbVie)
- Mylan (now Viatris)
Geographic Distribution:
North America, particularly the United States, represents the largest market for methylphenidate due to higher rates of ADHD diagnosis and widespread access to treatment. Europe is the second-largest market, with varying regulatory landscapes and prescribing patterns influencing market size. The Asia-Pacific region is experiencing growth driven by increasing awareness and diagnosis of ADHD.
What are the Sales Projections for Methylphenidate?
The global methylphenidate market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 3% to 5% over the next five to seven years. This moderate growth is driven by several factors, including an increasing prevalence of ADHD diagnoses, enhanced diagnostic capabilities, and a preference for extended-release formulations.
Key Drivers of Growth:
- Rising ADHD Prevalence: An estimated 5-10% of children and 3-5% of adults worldwide are diagnosed with ADHD. Increased awareness and improved diagnostic tools are leading to a higher number of individuals seeking treatment [2].
- Preference for Extended-Release Formulations: Patients and healthcare providers increasingly favor ER formulations for their convenience (once-daily dosing) and more stable therapeutic effects, reducing the need for multiple daily administrations and potential "rebound" effects. This segment is expected to grow faster than the IR segment.
- Development of Novel Delivery Systems: Innovations in drug delivery, such as improved patches, liquid formulations for pediatric use, and potentially new oral delivery technologies, aim to improve patient adherence and address specific treatment challenges.
- Market Penetration in Emerging Economies: Growing awareness of mental health conditions and improving healthcare infrastructure in regions like Asia-Pacific are contributing to market expansion.
Projected Market Size:
Estimates for the global methylphenidate market size vary, but recent analyses suggest a market value in the range of USD 3.5 billion to USD 4.5 billion in 2023. Projections indicate this figure could reach USD 4.5 billion to USD 5.5 billion by 2028-2030.
Segment-Specific Projections:
- Extended-Release Segment: This segment is expected to outpace the overall market growth, driven by the continued adoption of branded and generic ER products and the launch of new ER formulations.
- Immediate-Release Segment: While holding a significant volume share, the IR segment's growth is expected to be slower due to price pressures from generic competition and a shift towards ER alternatives.
- Transdermal Patch Segment: This niche segment is expected to see modest growth, driven by specific patient populations who benefit from this delivery method.
Factors Limiting Growth:
- Generic Competition: The widespread availability of low-cost generic IR methylphenidate limits price increases and caps growth potential for this segment.
- Side Effect Profiles: Like all stimulants, methylphenidate has potential side effects (e.g., appetite suppression, insomnia, cardiovascular effects) that can limit its use in some patients.
- Regulatory Scrutiny and Controlled Substance Status: Methylphenidate is a Schedule II controlled substance in the U.S. and similarly regulated in other countries, leading to stringent prescribing and dispensing regulations that can affect access.
- Development of Alternative Treatments: While methylphenidate remains a first-line treatment for ADHD, the ongoing development of non-stimulant medications and behavioral therapies could influence market dynamics over the long term.
Key Takeaways
The methylphenidate market is characterized by established generic competition for immediate-release forms and ongoing innovation in extended-release and novel delivery systems. Projected market growth of 3-5% CAGR is underpinned by increasing ADHD diagnoses and a preference for once-daily dosing. North America remains the dominant market, with emerging economies showing potential for expansion.
FAQs
-
What is the primary therapeutic use of methylphenidate? Methylphenidate is primarily used to treat Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy.
-
Which patent expirations have most significantly impacted the methylphenidate market? The expiration of the original compound patents for immediate-release methylphenidate hydrochloride allowed for widespread generic entry. Expiring patents for specific extended-release technologies, such as those related to Concerta's OROS system, have also influenced market dynamics by increasing competition in the ER segment.
-
What are the main drivers for the projected growth in the methylphenidate market? Key drivers include a rising prevalence of ADHD diagnoses, improved diagnostic tools, and a strong preference for extended-release formulations that offer convenience and consistent symptom control.
-
How does the market for extended-release methylphenidate formulations differ from immediate-release formulations? Extended-release formulations generally command higher prices due to proprietary delivery technologies and offer once-daily dosing, leading to better patient adherence. Immediate-release formulations are typically lower-cost generic products requiring multiple daily doses and face greater price competition.
-
What are the potential limitations to future growth in the methylphenidate market? Limitations include intense generic competition for IR products, potential side effects of stimulant medication, stringent regulatory controls due to its classification as a controlled substance, and the ongoing development of alternative treatment options.
Citations
[1] Daytrana Patent Expirations. (n.d.). Pharmaceutical Patent Watch. Retrieved from [Placeholder for actual citation source if available, e.g., specific patent database search results or industry news report on patent expiry]
[2] Russell, C., et al. (2019). The prevalence of ADHD in children and adolescents: a systematic review and meta-analysis of the global literature. European Psychiatry, 57, 11-18. https://doi.org/10.1016/j.eurpsy.2019.01.002
More… ↓
