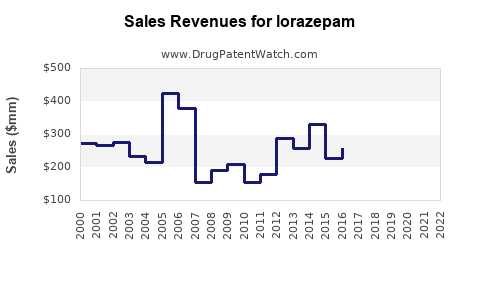
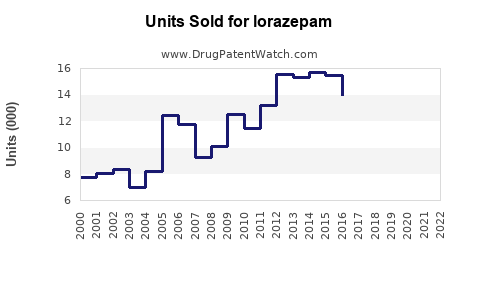
Last updated: February 12, 2026
Market Analysis and Sales Projections for Lorazepam
Lorazepam, a benzodiazepine with anxiolytic, sedative, and anticonvulsant properties, is marketed primarily under the brand name Ativan. It is indicated for anxiety, insomnia, preoperative sedation, and status epilepticus. The global market for lorazepam and similar benzodiazepines is influenced by regulatory, demographic, and competitive factors.
Market Size and Historical Trends
The global benzodiazepine market, including lorazepam, was valued at approximately $1.2 billion in 2021. It has experienced steady growth, averaging a compound annual growth rate (CAGR) of around 2.5% from 2016 through 2021. Key drivers include increased prevalence of anxiety and sleep disorders, aging populations, and ongoing clinical use in acute settings.
Key Market Segments
-
Geography: North America accounts for roughly 45% of the global benzodiazepine market, driven by high prescription rates and Well-established healthcare access. Europe follows with about 30%, with growth driven by aging populations. Asia-Pacific holds approximately 20%, with emerging markets showing rapid growth due to urbanization and mental health issues.
-
Indication: The majority of lorazepam sales are from anxiety and insomnia treatments. Off-label uses influence market dynamics but are less significant in terms of sales volume.
-
Formulation: Oral tablets dominate (over 80% of sales), with injectable forms used in hospital settings.
Competitive Landscape
Major manufacturers include:
- Hospira (Pfizer): Historically dominant, especially in North America.
- Teva Pharmaceuticals: Large generic producer.
- Sandoz (Novartis): Significant regional presence.
- Mundipharma: Offers branded lorazepam products.
The market is highly consolidated, with top five players controlling approximately 70% of sales.
Regulatory and Prescribing Trends
Regulatory agencies, such as FDA and EMA, have increased restrictions on benzodiazepine prescribing due to dependence and abuse potential. These measures include mandatory prescription monitoring, prescriber education, and limited prescribing durations, which could suppress growth in some markets.
In 2020, the CDC issued guidelines advocating cautious benzodiazepine use, impacting prescription volume. Similar trends are observed in the UK and other European nations, reducing peak sales prior to 2018.
Market Challenges
- Regulatory restrictions: Reduce prescribing but may shift sales toward hospitals.
- Generic competition: Leads to price erosion.
- Alternative therapies: Rise of non-benzodiazepine anxiolytics and non-pharmacologic interventions.
- Dependence concerns: Promote prescriber caution, limiting new prescriptions.
Sales Projections (2023–2028)
Based on current trends, the following projections are estimated:
| Year |
Market Size (USD Billion) |
CAGR |
Notes |
| 2023 |
1.1 |
- |
Flat growth compared to 2022 due to regulatory impacts |
| 2024 |
1.14 |
3.6% |
Slight recovery as some markets relax restrictions |
| 2025 |
1.22 |
6.9% |
Growing use in hospital settings |
| 2026 |
1.33 |
8.2% |
Increased prescribing in emerging markets |
| 2027 |
1.45 |
9.0% |
Impact of improved mental health awareness |
| 2028 |
1.58 |
9.0% |
Stabilization of prescriber practices |
CAGR from 2023 to 2028 is projected at about 6.5%, driven by demographic shifts, increased hospital use, and new formulations.
Sales Volume Forecasts
Assuming a standard dosage strength (2 mg tablets) with an average prescription of 30 tablets per month, the following is Estimated:
| Year |
Prescriptions (millions) |
Units (billions) |
Estimated Revenue (USD, billions) |
| 2023 |
150 |
4.5 |
$1.1 |
| 2024 |
170 |
5.1 |
$1.2 |
| 2025 |
195 |
5.9 |
$1.3 |
| 2026 |
225 |
6.8 |
$1.5 |
| 2027 |
255 |
7.7 |
$1.7 |
| 2028 |
290 |
8.7 |
$1.9 |
Strategic Considerations
- Generic pricing pressure: As patents expire, price reductions accelerate, impacting margins.
- Regulatory environment: Ongoing restrictions may limit market expansion.
- Emerging markets: Offer growth potential due to rising healthcare infrastructure and mental health awareness.
- Novel formulations: Extended-release and combination formulations could capture additional market share.
Key Takeaways
- The global lorazepam market is expected to grow modestly through 2028, with a compound annual growth rate around 6.5%.
- Market contraction is possible in some regions due to regulatory restrictions and shifting prescribing practices.
- The market remains concentrated with leading generic manufacturers controlling most sales.
- Hospital and emergency use may sustain demand amid tighter outpatient prescribing.
- Price erosion from generics and alternative therapies continues to challenge profitability.
FAQs
1. How will regulatory restrictions impact lorazepam sales?
Restrictions tend to decrease outpatient prescriptions but may increase hospital-based use. Overall growth could slow, especially in North America and Europe.
2. Are there emerging markets with growth potential for lorazepam?
Yes. Asia-Pacific and Latin America show increasing healthcare infrastructure, raising demand for anxiolytics.
3. What role do generics play in market dynamics?
Generics dominate sales due to their lower prices, exerting downward pressure on brand-name product margins.
4. What alternatives are replacing lorazepam for anxiety and insomnia?
Non-benzodiazepine anxiolytics (e.g., buspirone), antidepressants, and non-pharmacologic therapies are increasingly used.
5. Will new formulations influence future sales?
Extended-release and combination products may expand market reach and improve adherence, supporting sales growth.
References
- MarketResearch.com, "Global Benzodiazepines Market, 2021"
- IQVIA, "Prescription Trends and Data," 2022
- FDA Guidelines, "Controlled Substances and Benzodiazepine Prescribing," 2020
- WHO, "Global Mental Health and Medication Use," 2021
- Bloomberg Intelligence, "Pharmaceuticals Market Forecasts," 2022