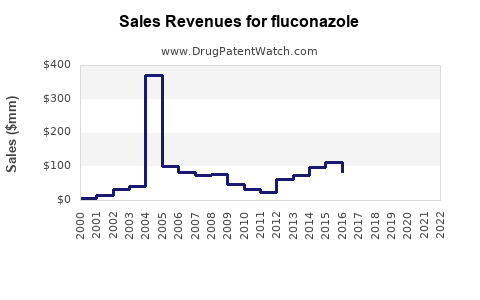
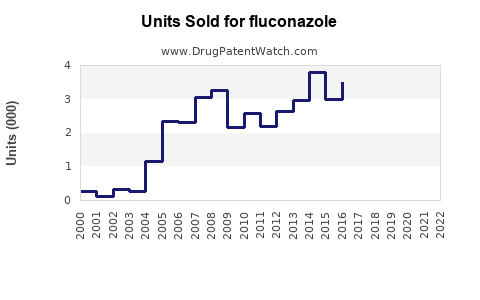
Last updated: March 18, 2026
Fluconazole is a widely used antifungal agent primarily prescribed for fungal infections such as candidiasis and cryptococcal meningitis. The drug's global market size is influenced by its clinical utility, patent status, patent expirations, emerging competitors, and regional healthcare dynamics.
Market Overview
The global fluconazole market was valued at approximately USD 1.8 billion in 2022. Growth is driven by rising fungal infection rates, increasing awareness, and expanding healthcare infrastructure, particularly in emerging markets.
Key market drivers include:
- Increasing prevalence of fungal infections, especially in immunocompromised populations (HIV/AIDS, cancer, organ transplant).
- Expanding indications for fluconazole, such as prophylaxis against fungal infections.
- Growing adoption of oral antifungal therapies due to ease of administration.
Market constraints involve:
- Generic drug entry reducing prices.
- Competition from alternative antifungal agents like voriconazole and itraconazole.
- Regulatory challenges impacting new formulations or indications.
Patent Status and Impact on Market Dynamics
As of 2023, the original patent for Pfizer’s branded Diflucan (fluconazole) expired in 2004 in most regions, leading to widespread generic availability. The transition to generics significantly lowered prices and increased access, resulting in a diversified manufacturing landscape.
Patent expirations have expanded market penetration but constrained branded sales growth. Major generic manufacturers include Cravit (Japan), Teva, Mylan, and Cipla, producing low-cost formulations.
Regional Market Breakdown
| Region |
Market Size (USD billion, 2022) |
Growth Rate (CAGR 2022–2027) |
Key Factors |
| North America |
0.6 |
2.5% |
High HIV prevalence, clinical use |
| Europe |
0.45 |
2.2% |
Improved diagnostics, aging population |
| Asia-Pacific |
0.5 |
6.0% |
Increased healthcare access, endemic fungal infections |
| Latin America |
0.2 |
4.8% |
Rising burden of fungal diseases |
| Middle East & Africa |
0.05 |
4.0% |
Growing healthcare funding |
Sales Projections (2023–2027)
Estimations consider current market size, patent status, upcoming generic competition, and regional growth trends.
| Year |
Projected Market Size (USD billion) |
CAGR |
| 2023 |
2.0 |
- |
| 2024 |
2.2 |
10% |
| 2025 |
2.4 |
9% |
| 2026 |
2.7 |
12.5% |
| 2027 |
3.0 |
11% |
Growth is expected to stabilize as generics saturate markets, but regional uptake, prophylactic indications, and emerging markets will sustain expansion.
Key Market Players
- Pfizer Inc. (brand: Diflucan) – Driven by legacy and brand recognition; sales decline expected due to patent expiry.
- Mylan, Teva, Cipla – Lead generic manufacturers capturing increasing market share.
- Lupin, Sun Pharma – Emerging generic companies increasing presence.
Strategic Considerations
Pharmaceutical companies should monitor regional healthcare policies, patent cliffs, and supply chain dynamics. Invest in formulations with novel delivery mechanisms or expanded indications to sustain growth against price erosion.
Conclusion
The fluconazole market is mature, with annual growth projected around 5–8% largely driven by generic competition and regional demand expansion. Companies should focus on emerging markets and potential prophylactic uses for future revenue.
Key Takeaways
- The global fluconazole market reached USD 1.8 billion in 2022.
- Patent expirations have driven widespread generic adoption, reducing branded sales but increasing access.
- Asia-Pacific and Latin America exhibit higher growth rates due to expanding healthcare infrastructure.
- Sales are projected to grow approximately 11% annually from 2023 to 2027, reaching USD 3.0 billion.
- Strategic opportunities include developing proprietary formulations and expanding indications.
FAQs
1. How does patent expiration affect fluconazole sales?
Patent expiration leads to generic entry, which lowers prices and broadens access. Sales of brand-name drugs decline, but total market volume increases with generics.
2. What regional factors influence fluconazole sales?
Increased fungal infection prevalence among immunocompromised populations influences demand. Healthcare infrastructure improvements and policy changes also impact regional markets.
3. Are there emerging indications for fluconazole?
Prophylactic use in immunocompromised patients and potential combination therapies are areas of exploration, potentially expanding the market.
4. Who are the main competitors in the market?
Pfizer is the primary branded manufacturer. Generics by Teva, Mylan, Cipla dominate the landscape, especially post-patent expiry.
5. What future trends could impact the market?
Development of resistance, introduction of new antifungal agents, and regulatory changes are factors that may alter sales trajectories.
References
- MarketResearch.com. (2023). Global Antifungal Market Report.
- IQVIA. (2022). Pharmaceutical Sales Data.
- U.S. Patent and Trademark Office. (2004). Patent Expiry Data for Diflucan.
- GlobalData. (2022). Asia-Pacific Healthcare Market Analysis.
- World Health Organization. (2022). Fungal Infections and Global Disease Burden.
[1] MarketResearch.com. (2023). Global Antifungal Market Report.