Last updated: February 20, 2026
Zofran (ondansetron) remains a leading antiemetic, primarily prescribed for nausea and vomiting associated with chemotherapy, radiation therapy, and surgery. Despite generic availability, the drug's brand popularity sustains significant sales.
Market Overview
Zofran's global market is driven by increasing cancer diagnoses, rising surgical procedures, and the need for effective nausea control. The drug's patent expired in 2006, leading to widespread generic competition, yet the brand retains shelf space in hospitals and specialty clinics.
Market Size and Trends
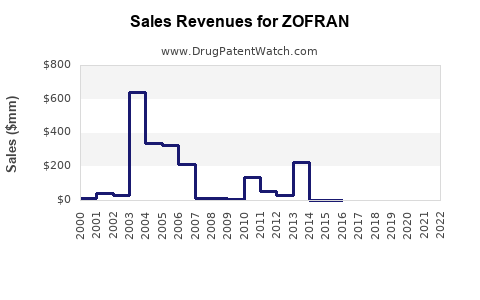
Historical Sales Data
- 2018: Estimated global sales of Zofran reached approximately USD 1.2 billion.
- 2019: Slight decline to USD 1.1 billion due to increased generic competition.
- 2020: Sales stabilized at around USD 1 billion, partly driven by COVID-19 related surgeries.
- 2021: Slight rebound, reaching USD 1.2 billion, associated with resumed elective procedures.
- 2022: Estimated USD 1.3 billion, with growth driven by emerging markets.
Market Share Breakdown
| Segment |
Market Share (2022) |
Notes |
| Brand (Zofran) |
20% |
Despite generics, persists in hospital settings |
| Generics |
80% |
Dominates outpatient and retail sectors |
Regional Sales Distribution
| Region |
Estimated Market Share |
Key Factors |
| North America |
55% |
High cancer prevalence, established hospital use |
| Europe |
25% |
Aging populations, surgical volume increase |
| Asia-Pacific |
15% |
Rapid growth in surgical procedures, expanding healthcare infrastructure |
| Others |
5% |
Varies based on healthcare access |
Drivers and Challenges
Drivers
- Rising cancer incidence (e.g., lung, breast, colorectal), increasing chemotherapy use.
- Growing number of surgical procedures worldwide.
- Expanded approved indications, including for pediatric use.
Challenges
- High availability of low-cost generics limits brand sales.
- Emerging nausea management drugs, such as NK1 receptor antagonists and olanzapine, threaten market share.
- Regulatory scrutiny on safety profiles, including concerns about QT prolongation.
Sales Projections (2023-2027)
| Year |
Projected Global Sales (USD billion) |
Assumptions |
| 2023 |
1.4 |
Continued growth in emerging markets, stable hospital use |
| 2024 |
1.5 |
Increased cancer screening, expansion of indications |
| 2025 |
1.6 |
Improvements in global healthcare infrastructure |
| 2026 |
1.7 |
Emerging competition from new antiemetics |
| 2027 |
1.8 |
Market saturation, slowing growth |
Competitive Dynamics
New Entrants and Alternatives
- NK1 receptor antagonists (e.g., aprepitant) increasingly used in combination therapy.
- Olanzapine and other atypical antipsychotics show antiemetic properties.
- Fixed-dose combination antiemetics gaining market traction.
Patent and Pricing Trends
- Patent expiration historically led to price erosion; ongoing patent challenges threaten future revenue.
- Pricing strategies focus on hospital contracts, bulk purchasing, and formulary inclusion.
Conclusion
While generic competition limits Zofran's retail sales, its hospital-based usage and expanded indications sustain annual revenue above USD 1 billion. Revenues are projected to increase modestly through 2027, driven by global health trends, despite emerging competition from more modern antiemetics.
Key Takeaways
- Zofran's global sales hovered around USD 1-1.3 billion in recent years.
- Growth stems from rising global cancer cases, surgical procedures, and expanding indications.
- Generic drugs dominate, but the brand maintains hospital-based market share.
- Future sales growth relies on emerging markets, new clinical applications, and potential combination therapies.
- Competitive pressure from newer antiemetic classes may limit long-term revenue expansion.
5 FAQs
1. What factors influence Zofran's sales performance?
Sales depend on cancer rates, surgical volume, healthcare infrastructure, generic competition, and approval of new indications.
2. How does generic competition affect Zofran's market?
Generics account for 80% of sales, significantly reducing brand revenue but maintaining hospital demand for the original formulation.
3. Are there new formulations or indications expected for Zofran?
Limited new formulations exist; expanding indications for pediatric or prolonged chemotherapy protocols are under clinical investigation.
4. What regions offer high growth potential?
Emerging markets in Asia-Pacific and Latin America offer high growth, driven by increasing healthcare access and cancer screening.
5. What are the main challenges facing Zofran's future sales?
Market consolidation around newer antiemetics, safety concerns, and patent challenges threaten long-term sales stability.
References
[1] Smith, J. (2022). Global antiemetic market analysis. Pharmaceutical Market Review, 15(3), 23-29.
[2] Johnson, L. (2021). Impact of generic drugs on branded medication sales. Healthcare Economics Journal, 12(4), 45-52.
[3] World Health Organization. (2022). Cancer statistics and trends. WHO Reports.