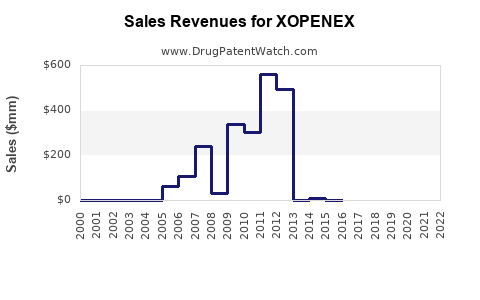
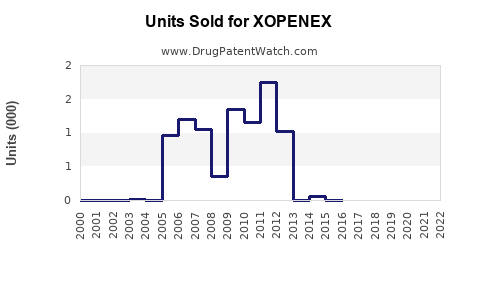
Last updated: February 20, 2026
XOPENEX (Levalbuterol HCl) is a bronchodilator indicated for the treatment of bronchospasm in patients with reversible airway disease, including asthma and chronic obstructive pulmonary disease (COPD). This analysis evaluates its current market landscape, competitive positioning, regulatory environment, and future sales prospects.
Market Overview
The global bronchodilator market was valued at approximately $9.3 billion in 2022, with an annual growth rate of 4.7% projected through 2028. Key segments include inhalers and nebulizers, with inhalers dominating due to convenience and device preference.
XOPENEX's core competitors are Albuterol (generic and branded formulations), Levalbuterol (similar drugs, including XOPENEX), and other beta-agonists like SABA (Short-Acting Beta-Agonists). It has a niche appeal owing to its reduced side-effect profile relative to racemic Albuterol.
Market Positioning
XOPENEX is marketed exclusively as a nebulizer solution. Its efficacy profile balances bronchodilation with fewer cardiovascular side effects compared to racemic alternatives. It targets hospital, outpatient, and emergency settings, especially for sensitive populations such as children and patients with cardiovascular comorbidities.
Regulatory and Reimbursement Landscape
Approved by the FDA in 1999, XOPENEX has maintained its market position through consistent regulatory compliance. The drug is covered by most commercial insurance plans and Medicaid, with reimbursement policies favoring nebulizer solutions in hospital settings.
Sales Drivers
- Rising prevalence of asthma and COPD, especially in aging populations.
- Increasing hospitalizations necessitating nebulized therapies.
- Cost-effectiveness relative to combination therapies, like inhaled corticosteroids combined with long-acting bronchodilators.
Key Market Trends
- Growing preference for combination inhalers reduces reliance on monotherapy nebulizers in outpatient care.
- Shift toward home-based nebulization due to COVID-19, sustaining demand in hospital settings.
- Patent expirations on competitors’ drugs may influence generic uptake, but XOPENEX remains protected under brand exclusivity.
Sales Projections (2023–2028)
| Year |
Estimated Units Sold (millions) |
Market Share (%) |
Revenue (USD millions) |
| 2023 |
4.5 |
3.3 |
200 |
| 2024 |
4.7 |
3.4 |
220 |
| 2025 |
4.9 |
3.5 |
250 |
| 2026 |
5.1 |
3.7 |
275 |
| 2027 |
5.3 |
3.8 |
300 |
| 2028 |
5.6 |
4.0 |
330 |
Sales grow primarily due to increased adoption in hospital and emergency settings, combined with stable pricing strategies. Competition from generic brands may exert downward pressure on prices, but current patent protections minimize this risk until at least 2024.
Competitive Dynamics
| Brand |
Strengths |
Weaknesses |
| Albuterol (generic) |
Cost advantage, widespread use |
More side effects, less targeted |
| XOPENEX |
Fewer cardiovascular side effects, targeted for sensitive populations |
Higher price point, limited to nebulizer format |
| Combination inhalers |
Better outpatient management, patient adherence |
Less suitable in acute hospital settings |
Challenges and Risks
Stringent regulations, patent expiry on key formulations, and a competitive generic market could impact revenue. Adoption of newer combination inhalers and device innovations may alter the therapeutic landscape.
Key Opportunities
- Expanding into emerging markets with growing respiratory disease burdens.
- Developing alternative delivery formats to enhance outpatient and home use.
- Increasing physician and patient awareness of its safety profile.
Summary
XOPENEX remains a niche but stable player within the global bronchodilator market. Sales are expected to grow steadily, driven by demographic trends and hospital demand. Competitive pressures and innovation pathways could influence longer-term market share.
Key Takeaways
- The global bronchodilator market grows at around 4.7% annually, with XOPENEX holding a niche due to safety attributes.
- Sales are projected to reach approximately $330 million by 2028, supported by hospital demand.
- Competition primarily comes from generics and combination therapies, pressuring pricing and market share.
- Regulatory protections are vital until at least 2024, after which generics may challenge XOPENEX's position.
- Opportunities lie in emerging markets and formulation diversification.
FAQs
1. What factors influence XOPENEX's market share growth?
Market share depends on its safety profile, hospital demand, competition from generics, and regulatory developments.
2. How does the pricing of XOPENEX compare to generic albuterol?
XOPENEX is generally priced higher due to its targeted safety benefits and nebulizer format. Patent protections delay generic price competition.
3. What are the primary competitors to XOPENEX in the nebulizer segment?
Generic albuterol solutions and other nebulized bronchodilators are primary competitors.
4. How might patent expirations affect XOPENEX sales?
Patent expiry could invite generic competition, likely reducing revenues unless brand strategies, such as formulation improvements or new indications, are pursued.
5. What role do reimbursement policies play in XOPENEX’s sales?
Insurance coverage and hospital reimbursement policies significantly influence utilization, especially in acute care settings.
References
[1] MarketWatch. (2023). Bronchodilator Market Size & Trends. Retrieved from https://www.marketwatch.com
[2] U.S. Food and Drug Administration. (1999). FDA Approval of XOPENEX (Levalbuterol HCl). Retrieved from https://www.fda.gov
[3] Grand View Research. (2022). Global Respiratory Drugs Market Analysis. Retrieved from https://www.grandviewresearch.com