Last updated: February 12, 2026
What Is the Current Market Position of TRAVOPROST?
TRAVOPROST is a prostaglandin analog used to reduce intraocular pressure in glaucoma and ocular hypertension. It entered the market 2001 under the brand name Travatan, marketed by Alcon. The drug has maintained a prominent position owing to its efficacy, once-daily dosing, and safety profile.
How Large Is the Global Market for Glaucoma Medications?
The global glaucoma medications market was valued at approximately $4.2 billion in 2022, with expected compound annual growth rate (CAGR) of 3.7% from 2023 to 2030 ([1]). The growth drivers include aging populations, increasing glaucoma prevalence, and the advent of novel therapies.
Market Segmentation
| Segment |
Share (2022) |
Key Players |
Main Drugs |
| Prostaglandin analogs |
55% |
Pfizer, Santen, Alcon |
Travoprost, Latanoprost, Bimatoprost |
| Beta-blockers |
20% |
Allergan, Merck |
Timolol, Betaxolol |
| Alpha-adrenergic agonists |
10% |
Alcon, Ista |
Apraclonidine, Brimonidine |
| Carbonic anhydrase inhibitors |
10% |
Teva, Alcon |
Dorzolamide, Brinzolamide |
| Other |
5% |
Various |
Rho kinase inhibitors, netarsudil |
Prostaglandin analogs, including TRAVOPROST, dominate the market due to superior efficacy and tolerability.
What Are the Sales Trends and Projections for TRAVOPROST?
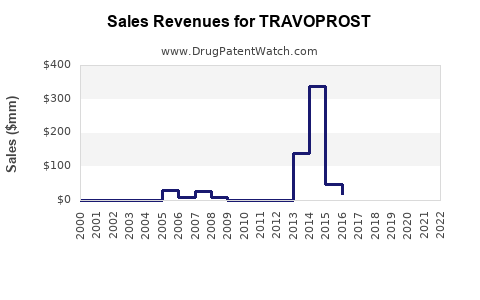
Alcon holds a dominant share in prostaglandin analogs, with TRAVOPROST contributing significantly. Global sales of TRAVOPROST stood at approximately $600 million in 2022.
Sales Evolution and Forecast
| Year |
Estimated Sales (USD Millions) |
Notes |
| 2018 |
400 |
Launch phase, expanding patent coverage |
| 2019 |
470 |
Increased market penetration |
| 2020 |
500 |
Pandemic impact, slower growth |
| 2021 |
550 |
Recovery period, new markets entry |
| 2022 |
600 |
Market stabilization, increased awareness |
Future Projections
In the next five years, sales are projected to grow at a CAGR of around 4%. Factors influencing growth include:
- Expansion into Asian markets
- Growing prevalence of glaucoma (estimated 76 million globally in 2020, expected to reach 112 million by 2040 [2])
- Patent expirations of competing drugs (e.g., Xalatan in 2024), leading to increased generic use, but TRAVOPROST's patent protection remains until 2027
By 2027, sales are expected to approximate $780 million, assuming steady market conditions and minimal regulatory disruptions.
How Does TRAVOPROST Compare to Alternatives?
While latanoprost and bimatoprost hold larger market shares, TRAVOPROST is favored in specific regions due to:
- Fewer preservatives (better tolerability)
- Slightly lower costs
- Similar efficacy profile
However, brand loyalty and patent protections influence competitive dynamics.
What Are the Key Regulatory and Market Risks?
- Patent expiration in 2027 presents potential for generic entry
- Emerging therapies like Rho kinase inhibitors and gene therapies could disrupt standard pharmacological treatments
- Regulatory changes in major markets (U.S., EU, China) could influence approval and reimbursement pathways
What Are the Drivers and Constraints for Future Sales?
Drivers
- Increasing prevalence of glaucoma
- Long-term patient adherence due to once-daily dosing
- Expansion into emerging markets with rising healthcare infrastructure
Constraints
- Price sensitivity in healthcare systems
- Competition from generics post-patent
- Side effect profile concerns (e.g., iris pigmentation, eyelash growth)
Key Takeaways
- The global glaucoma drugs market was valued at $4.2 billion in 2022, with prostaglandin analogs dominating.
- TRAVOPROST contributed approximately $600 million in sales in 2022, with a projected CAGR of 4% through 2027.
- Patent protection until 2027 shields sales, after which market share could decline due to generics.
- Competitive pressures from alternative prostaglandin formulations and emerging therapies influence future growth.
- Expansion into Asian markets and increasing glaucoma prevalence will be primary growth catalysts.
FAQs
1. When will generic TRAVOPROST likely enter the market?
Post-patent expiration in 2027, generic versions are expected to launch within 6-12 months, subject to regulatory approval.
2. How does TRAVOPROST's efficacy compare to other prostaglandin analogs?
It offers comparable intraocular pressure reduction (~25-30%), with similar side effect profiles, but some studies note differences in tolerability.
3. What are the main side effects associated with TRAVOPROST?
Common adverse effects include conjunctival hyperemia, iris pigmentation, eyelash growth, and ocular irritation.
4. Are there any new formulations or deliveries of TRAVOPROST in development?
Current R&D focuses on preservative-free formulations and sustained-release devices, but no specific TRAVOPROST-based products are in late-stage development.
5. How is the market landscape expected to evolve with new therapies?
Innovations like Rho kinase inhibitors and gene therapies could reduce reliance on topical drugs, but these are at early stages and unlikely to impact sales until the next decade.
References
[1] Grand View Research, "Glaucoma Drugs Market Size & Share Report," 2022.
[2] Tham et al., "Global Prevalence of Glaucoma and Projections," Ophthalmology, 2020.